Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and cost-effective pathways for constructing complex heterocyclic scaffolds, particularly those containing trifluoromethyl groups which enhance metabolic stability and bioavailability. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value drugs such as Maraviroc, Sitagliptin, and Deferasirox. This innovation addresses critical bottlenecks in traditional synthesis by employing a non-metallic iodine-promoted strategy that eliminates the need for expensive transition metal catalysts. By leveraging readily available aryl ethanones and trifluoroethylimine hydrazides, this technology offers a streamlined route that is not only chemically efficient but also operationally simple, requiring neither stringent anhydrous nor oxygen-free environments. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for key pharmaceutical intermediates while maintaining rigorous purity standards.
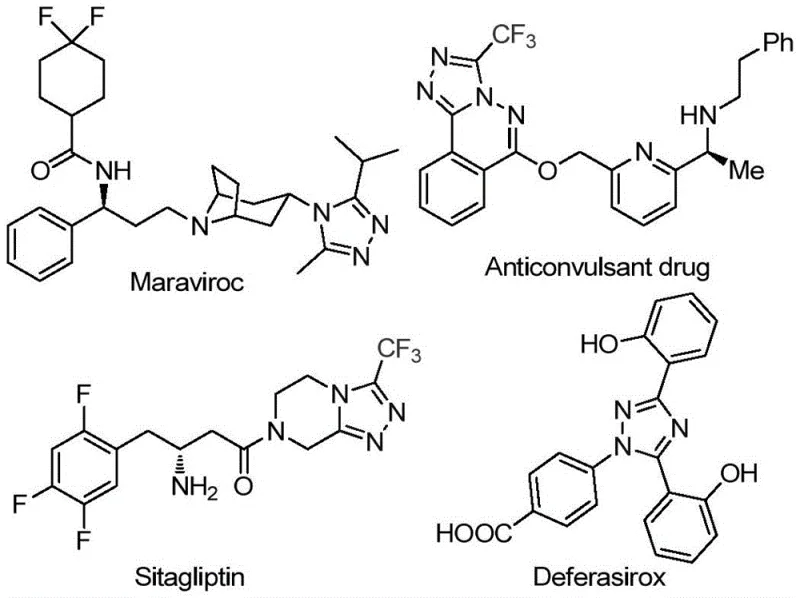
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, especially those bearing both trifluoromethyl and acyl groups, has been fraught with synthetic challenges. Conventional methodologies often rely heavily on transition metal catalysis, which introduces significant complications regarding residual metal removal—a critical quality attribute for active pharmaceutical ingredients (APIs). Furthermore, many existing protocols demand harsh reaction conditions, including strict exclusion of moisture and oxygen, which necessitates specialized equipment and increases operational expenditures. The reliance on scarce or toxic reagents also poses environmental and supply chain risks, potentially leading to volatility in raw material pricing and availability. These factors collectively hinder the seamless commercial scale-up of complex pharmaceutical intermediates, forcing manufacturers to accept lower yields or incur higher purification costs to meet regulatory specifications.
The Novel Approach
The methodology outlined in patent CN113105402B presents a paradigm shift by utilizing elemental iodine and dimethyl sulfoxide (DMSO) to drive the reaction through a tandem iodination and Kornblum oxidation sequence. This metal-free approach circumvents the issues associated with heavy metal contamination, thereby simplifying the downstream purification process and ensuring a cleaner impurity profile. The reaction proceeds efficiently under relatively mild thermal conditions without the need for inert atmosphere protection, drastically reducing the technical barrier for implementation. By using cheap and abundant starting materials like aryl ethanones, the process inherently lowers the cost of goods sold (COGS). This novel pathway not only enhances the functional group tolerance, allowing for diverse substitution patterns at the 3 and 4 positions, but also provides a versatile platform for generating libraries of trifluoromethylated triazoles essential for modern drug discovery and development.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this synthetic breakthrough lies in the elegant cascade reaction initiated by the interaction between aryl ethanones and iodine in DMSO. Initially, the aryl ethanone undergoes an iodination followed by a Kornblum oxidation to generate an aryl diketone intermediate in situ. This reactive species then condenses with trifluoroethylimine hydrazide to form a hydrazone intermediate. Subsequently, under the synergistic promotion of iodine and a base system comprising sodium dihydrogen phosphate and pyridine, an intramolecular cyclization occurs to forge the 1,2,4-triazole ring. This mechanism is particularly advantageous because it avoids the formation of stable metal-complex byproducts that are difficult to separate. The use of DMSO is critical not just as a solvent but as an oxidant participant, ensuring high conversion rates of the starting materials into the desired heterocyclic product.
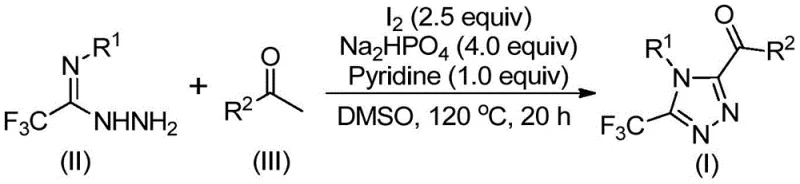
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining product integrity. Traditional strong acid or base mediated cyclizations often lead to decomposition of sensitive functional groups or polymerization side reactions. In contrast, the buffered environment provided by the phosphate/pyridine system moderates the reaction kinetics, minimizing the generation of unknown impurities. The specific stoichiometry, utilizing a molar ratio of sodium dihydrogen phosphate to pyridine to iodine of roughly 4:1:2.5, is optimized to balance the oxidation potential with the cyclization efficiency. This precise control ensures that the final high-purity pharmaceutical intermediates meet the stringent requirements of global regulatory bodies, reducing the risk of batch rejection and ensuring consistent quality across production runs.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical implementation of this synthesis involves a straightforward two-stage heating protocol that is amenable to standard reactor configurations found in most fine chemical facilities. The process begins with the activation of the ketone substrate, followed by the addition of the nitrogen source and cyclization promoters. This operational simplicity is a key driver for its adoption in contract development and manufacturing organizations (CDMOs) looking to reduce lead times. The detailed standardized synthesis steps, including specific temperature ramps and workup procedures, are outlined below to facilitate immediate technology transfer and process validation.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical implications of adopting this patented method are profound. The elimination of precious metal catalysts removes a major cost center and mitigates the supply risk associated with fluctuating prices of metals like palladium or copper. Furthermore, the use of commodity chemicals such as acetophenones and elemental iodine ensures a stable and diversified supply base, reducing dependency on single-source vendors. The robustness of the reaction against atmospheric moisture means that production schedules are less likely to be disrupted by environmental control failures, enhancing overall plant throughput and reliability.
- Cost Reduction in Manufacturing: The substitution of expensive catalytic systems with inexpensive elemental iodine and common bases results in a drastic reduction in raw material costs. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, lowers the consumption of auxiliary materials and reduces waste disposal costs. This cumulative effect leads to substantial cost savings in the manufacturing of trifluoromethylated heterocycles, improving the margin profile for downstream API production.
- Enhanced Supply Chain Reliability: Since the starting materials are widely available commercially and do not require specialized storage conditions like strict inert atmospheres, the supply chain becomes more resilient. The ability to source reagents from multiple global suppliers prevents bottlenecks and ensures continuous production flow. This reliability is crucial for meeting the just-in-time delivery demands of large pharmaceutical clients who prioritize security of supply above all else.
- Scalability and Environmental Compliance: The process is designed for scalability, having been demonstrated to expand easily from gram to multi-kilogram scales without loss of efficiency. From an environmental standpoint, the absence of heavy metals simplifies effluent treatment and aligns with increasingly strict green chemistry regulations. This compliance reduces the regulatory burden and potential fines associated with hazardous waste management, making the facility more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-promoted triazole synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations for potential partners.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly reducing raw material costs and simplifying downstream purification.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: Unlike many traditional heterocyclic syntheses, this method does not require strict anhydrous or oxygen-free conditions, making it highly robust and easier to operate on a large industrial scale.
Q: Can this method be scaled for commercial API production?
A: Yes, the patent explicitly demonstrates that the reaction can be easily expanded from gram-level laboratory synthesis to larger scales, offering excellent potential for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient heterocyclic synthesis in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113105402B are successfully translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. Our capability to handle complex fluorinated intermediates positions us as a preferred partner for companies seeking to optimize their API supply chains.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your development timelines and reduce overall production costs.
