Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Global Pharmaceutical Supply Chains
Introduction to Advanced Triazole Manufacturing Technologies
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles, particularly those incorporating trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN113105402B introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value active pharmaceutical ingredients (APIs) such as Maraviroc, Sitagliptin, and Deferasirox. 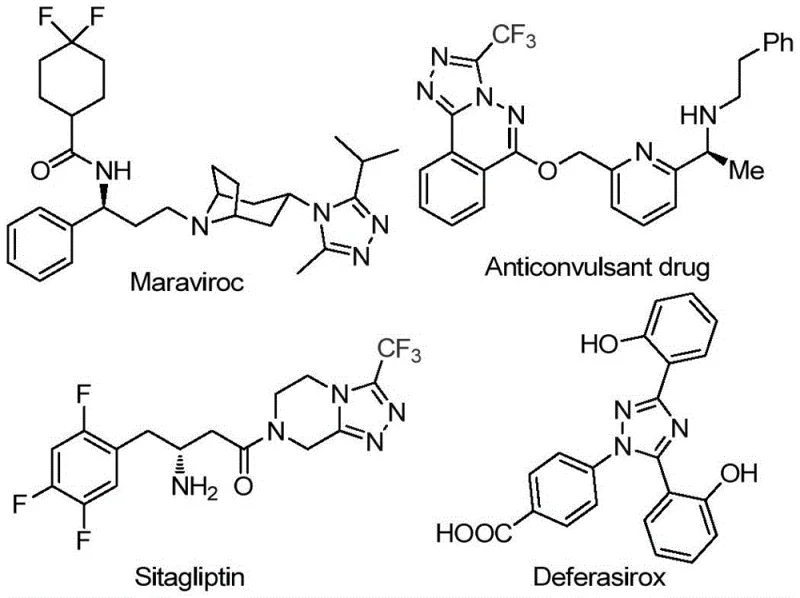 This innovation addresses critical bottlenecks in traditional heterocycle synthesis by eliminating the dependency on toxic heavy metal catalysts and严苛 anhydrous conditions. For R&D directors and procurement managers, this represents a pivotal shift towards greener, more cost-effective manufacturing processes that maintain high purity standards while simplifying the supply chain for complex intermediates.
This innovation addresses critical bottlenecks in traditional heterocycle synthesis by eliminating the dependency on toxic heavy metal catalysts and严苛 anhydrous conditions. For R&D directors and procurement managers, this represents a pivotal shift towards greener, more cost-effective manufacturing processes that maintain high purity standards while simplifying the supply chain for complex intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole scaffolds has relied heavily on transition metal catalysis or multi-step sequences requiring stringent exclusion of moisture and oxygen. Traditional pathways often involve the use of expensive palladium or copper catalysts, which not only inflate raw material costs but also introduce significant downstream processing challenges. The removal of trace heavy metals to meet pharmacopeial limits (often <10 ppm) necessitates specialized scavenging resins or extensive recrystallization protocols, drastically increasing production time and waste generation. Furthermore, many existing methods suffer from limited substrate scope, failing to tolerate diverse functional groups on the aryl rings, which restricts the chemical space available for medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast, the methodology disclosed in CN113105402B leverages a metal-free, iodine-promoted cascade reaction that operates efficiently in dimethyl sulfoxide (DMSO). This novel approach utilizes readily available aryl ethyl ketones and trifluoroethylimide hydrazides as starting materials, bypassing the need for pre-functionalized precursors. The reaction mechanism ingeniously combines iodination and Kornblum oxidation in situ, followed by tandem cyclization, all within a single pot. By operating under aerobic conditions without the need for inert gas protection, the process significantly lowers operational complexity. This simplicity translates directly to reduced capital expenditure for reactor setups and lower energy consumption, making it an ideal candidate for cost reduction in API manufacturing where margin compression is a constant pressure.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this synthetic breakthrough lies in the dual role of elemental iodine and DMSO. Initially, the aryl ethyl ketone undergoes an iodination followed by a Kornblum oxidation to generate an aryl diketone intermediate. This oxidative transformation is critical as it activates the carbonyl carbon for subsequent nucleophilic attack. 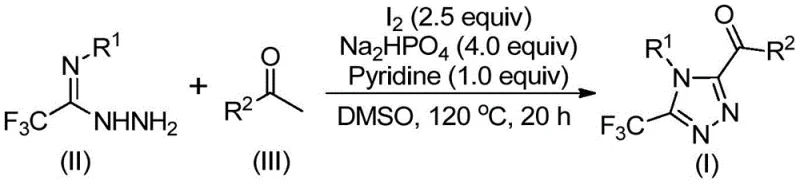 Following this activation, the trifluoroethylimide hydrazide condenses with the oxidized intermediate to form a hydrazone species. In the presence of the base (sodium dihydrogen phosphate and pyridine) and excess iodine, this hydrazone undergoes an intramolecular cyclization. The iodine acts as a mild Lewis acid and oxidant to facilitate the ring closure,最终 yielding the stable 1,2,4-triazole core with the trifluoromethyl group intact at the 3-position. This mechanistic pathway ensures high regioselectivity and minimizes the formation of isomeric byproducts, a common issue in triazole synthesis.
Following this activation, the trifluoroethylimide hydrazide condenses with the oxidized intermediate to form a hydrazone species. In the presence of the base (sodium dihydrogen phosphate and pyridine) and excess iodine, this hydrazone undergoes an intramolecular cyclization. The iodine acts as a mild Lewis acid and oxidant to facilitate the ring closure,最终 yielding the stable 1,2,4-triazole core with the trifluoromethyl group intact at the 3-position. This mechanistic pathway ensures high regioselectivity and minimizes the formation of isomeric byproducts, a common issue in triazole synthesis.
From an impurity control perspective, the absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling or over-oxidation, which often plague conventional methods. The use of DMSO as both solvent and reactant in the oxidation step ensures high conversion rates, while the specific stoichiometry of iodine (2.5 equivalents) and base (4.0 equivalents of Na2HPO4) creates a buffered environment that suppresses decomposition of the sensitive hydrazide moiety. For quality control teams, this means a cleaner crude reaction profile, simplifying the purification workflow and ensuring that the final high-purity pharmaceutical intermediates meet rigorous specifications with minimal effort.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The operational protocol for this synthesis is designed for maximum efficiency and safety in a standard chemical plant environment. The process begins with the dissolution of aryl ethyl ketone and iodine in DMSO, followed by a controlled heating phase to initiate oxidation. Subsequently, the hydrazide component and buffering agents are introduced, and the temperature is ramped to drive the cyclization to completion. The detailed standardized synthesis steps, including precise molar ratios and temperature ramps validated across multiple substrates, are outlined below to ensure reproducibility at scale.
- Mix aryl ethyl ketone and elemental iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add sodium dihydrogen phosphate, pyridine, additional iodine, and trifluoroethylimide hydrazide to the mixture.
- Heat the reaction to 110-130°C for 12-20 hours, then filter and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted technology offers transformative advantages in terms of cost structure and logistical reliability. The shift away from precious metal catalysts removes a volatile cost component from the bill of materials, as iodine is abundant and significantly cheaper than palladium or rhodium complexes. Moreover, the simplified workup procedure—often requiring only filtration and standard chromatography—reduces the consumption of specialized purification media and solvents. This streamlining of the manufacturing process directly contributes to substantial cost savings in pharmaceutical intermediate production, allowing for more competitive pricing in long-term supply contracts without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts is the primary driver for cost optimization in this process. Traditional methods often require catalyst loadings of 1-5 mol% of precious metals, which not only increases direct material costs but also necessitates expensive recovery or disposal protocols to meet environmental regulations. By utilizing elemental iodine, a commodity chemical, the process drastically reduces the raw material cost per kilogram of product. Additionally, the ability to run the reaction under ambient atmospheric conditions removes the need for nitrogen or argon blanketing systems, further lowering utility costs and infrastructure requirements for the manufacturing facility.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically aryl ethyl ketones and trifluoroethylimide hydrazides, are commercially available in bulk quantities from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions that often occur with proprietary or single-source catalysts. The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens, alkoxy, and alkyl substituents, means that the same process platform can be used to manufacture a diverse library of analogues. This flexibility allows supply chain planners to consolidate production campaigns and reduce lead times for high-purity intermediates, ensuring continuity of supply for downstream API synthesis.
- Scalability and Environmental Compliance: The patent data demonstrates that the reaction scales effectively from gram to multi-kilogram levels without loss of efficiency, a critical factor for commercial scale-up of complex intermediates. The use of DMSO, a high-boiling polar aprotic solvent, facilitates heat management in large reactors, while the absence of heavy metals simplifies wastewater treatment and waste disposal compliance. This aligns perfectly with modern green chemistry initiatives and regulatory pressures to reduce the environmental footprint of chemical manufacturing. Companies adopting this technology can expect smoother regulatory audits and lower costs associated with environmental health and safety (EHS) compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and limitations for potential partners.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the process described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, completely avoiding the need for costly palladium or copper catalysts and the subsequent heavy metal removal steps.
Q: What are the typical reaction conditions and yields for this triazole synthesis?
A: The reaction proceeds in DMSO solvent under aerobic conditions without strict anhydrous requirements. Experimental data indicates yields ranging from 37% to 86% depending on the substrate, with optimal conditions at 110-130°C.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is easily scalable from gram level to industrial production due to the use of cheap, commercially available raw materials and simple post-treatment procedures like filtration and chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient, metal-free synthetic routes in the modern pharmaceutical landscape. Our technical team has extensively evaluated the methodology described in CN113105402B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including residual solvent analysis and heavy metal testing, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole meets the highest industry standards.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced technology for your next project. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your trusted partner in delivering high-quality intermediates with speed and precision.
