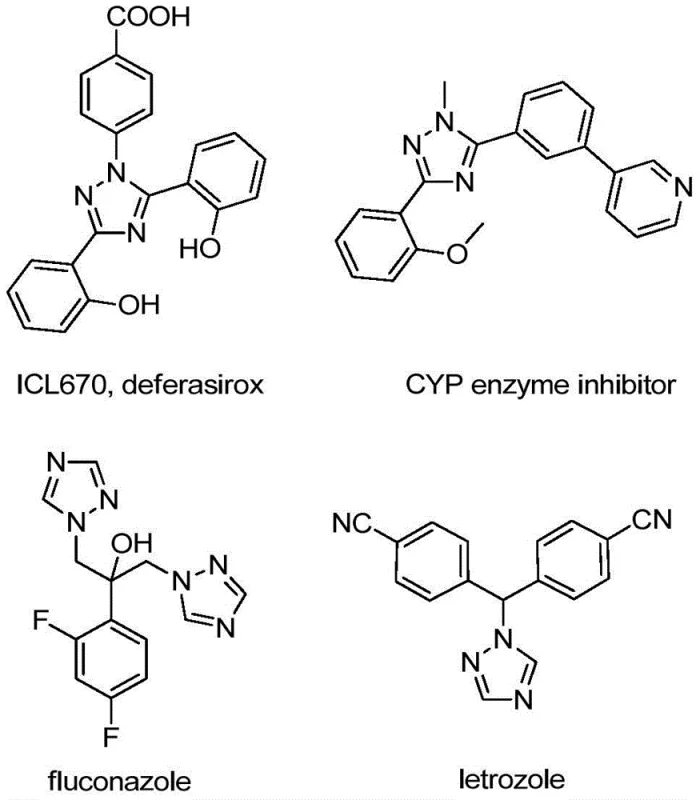
Revolutionizing Triazole Synthesis: Scalable and Cost-Efficient Production of 5-Trifluoromethyl 1,2,4-Triazole Intermediates for Pharmaceutical Manufacturing
The patent CN110467579B introduces a groundbreaking synthetic methodology for producing 5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry with profound implications for pharmaceutical manufacturing. This innovative approach addresses longstanding challenges in synthesizing trifluoromethyl-containing heterocycles by eliminating the need for expensive transition metal catalysts and stringent reaction conditions that have traditionally complicated large-scale production. The methodology demonstrates exceptional versatility through substrate design flexibility, enabling the preparation of diverse triazole derivatives with varying substitution patterns at the 4 and 5 positions while maintaining high reaction efficiency. Crucially, the process operates effectively under ambient conditions without requiring anhydrous or anaerobic environments, substantially reducing operational complexity and associated costs compared to conventional synthetic routes. This patent represents a strategic solution for manufacturers seeking reliable access to high-value triazole intermediates essential for developing next-generation pharmaceuticals and advanced materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted heterocycles typically rely on either pre-formed heterocycle trifluoromethylation using expensive and often unstable reagents or coupling reactions with specialized trifluoromethyl synthons like trifluorodiazoethane. These methods frequently require transition metal catalysts such as palladium or copper complexes that necessitate extensive purification steps to remove toxic metal residues, significantly increasing production costs and complicating regulatory compliance for pharmaceutical applications. The stringent anhydrous and anaerobic conditions mandated by many conventional protocols create substantial operational challenges during scale-up, requiring specialized equipment and rigorous process controls that extend manufacturing timelines and increase capital investment. Furthermore, the limited substrate scope and functional group tolerance of existing methodologies restrict their applicability across diverse molecular architectures needed for modern drug discovery programs. The reliance on complex multi-step sequences also contributes to lower overall yields and higher waste generation compared to ideal synthetic pathways.
The Novel Approach
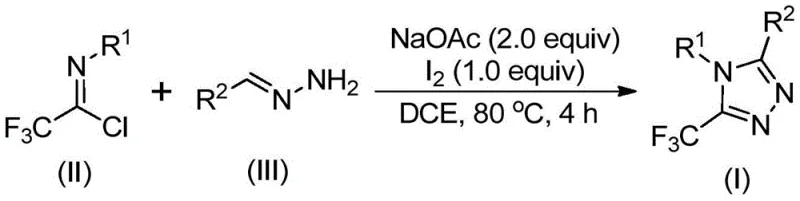
The patented methodology overcomes these limitations through an elegant iodine-catalyzed cyclization process that utilizes readily available starting materials including sodium acetate, trifluoroethylimidoyl chloride, and hydrazones under mild reaction conditions. This innovative approach eliminates the need for expensive transition metal catalysts while maintaining excellent reaction efficiency across a broad substrate scope with diverse functional group tolerance. The process operates effectively at 80°C in standard organic solvents like dichloroethane without requiring anhydrous or anaerobic environments, dramatically simplifying manufacturing requirements and reducing operational complexity. The reaction mechanism proceeds through base-promoted intermolecular carbon-nitrogen bond formation followed by isomerization and iodine-mediated oxidative cyclization to deliver high-purity triazole products with minimal purification requirements. This streamlined approach enables direct scale-up from laboratory to commercial production while maintaining consistent product quality and yield across different batch sizes.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with base-promoted nucleophilic attack by the hydrazone nitrogen on the electrophilic carbon of trifluoroethylimidoyl chloride (II), forming an intermediate amidine species through carbon-nitrogen bond formation. This initial step is facilitated by sodium acetate acting as a mild base under the reaction conditions of 80°C in dichloroethane solvent. The resulting intermediate undergoes spontaneous isomerization to generate a more stable tautomeric form that positions the molecule for subsequent cyclization events. Elemental iodine then promotes oxidative iodination at a strategic position on the intermediate structure, creating an electrophilic center that triggers intramolecular cyclization through nucleophilic attack by the adjacent nitrogen atom. This key cyclization step is followed by aromatization through dehydrogenation, ultimately yielding the fully conjugated 5-trifluoromethyl substituted 1,2,4-triazole product with excellent regioselectivity.
The mechanism inherently controls impurity formation through its stepwise progression that avoids highly reactive intermediates prone to side reactions. The mild reaction conditions prevent decomposition pathways commonly observed in high-temperature or strongly acidic/basic processes used in alternative synthetic routes. The absence of transition metals eliminates potential sources of metal-catalyzed side products that would require additional purification steps in pharmaceutical manufacturing. The well-defined reaction pathway ensures consistent product quality across different substrate combinations while maintaining high functional group tolerance as demonstrated by successful reactions with various substituted aryl groups at both R1 and R2 positions. This controlled mechanism directly contributes to the high purity profiles observed in the final products without requiring extensive post-reaction purification beyond standard column chromatography.
How to Synthesize 5-Trifluoromethyl Substituted Triazoles Efficiently
This patented methodology provides a robust framework for manufacturing high-purity triazole intermediates through a carefully optimized sequence that balances reaction efficiency with operational simplicity. The process begins with precise stoichiometric control of reactants where trifluoroethylimidoyl chloride serves as the key building block for introducing the trifluoromethyl group at the critical position in the triazole ring structure. The reaction proceeds through a two-stage process where initial condensation occurs under mild heating followed by iodine-mediated cyclization that completes the heterocyclic ring formation with excellent regioselectivity. The methodology has been validated across multiple substrate combinations demonstrating consistent performance regardless of substitution patterns on both reactant components. Detailed standardized synthesis steps for commercial implementation are provided below to ensure reliable scale-up from laboratory development to full production capacity.
- Combine sodium acetate, trifluoroethylimidoyl chloride (II), and hydrazone (III) in dichloroethane solvent under ambient conditions without requiring anhydrous or anaerobic environments.
- Heat the reaction mixture to 80°C for four hours to facilitate base-promoted carbon-nitrogen bond formation and subsequent isomerization processes.
- Introduce elemental iodine as the catalyst to enable oxidative iodination followed by intramolecular electrophilic substitution and aromatization to form the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits by addressing critical pain points in pharmaceutical intermediate procurement and supply chain management. The elimination of transition metal catalysts removes significant cost drivers associated with catalyst procurement, handling, and post-reaction removal processes that typically add multiple purification steps to traditional manufacturing workflows. By operating under ambient conditions without requiring specialized environmental controls, the process reduces capital expenditure on specialized equipment while simultaneously lowering operational costs through simplified process validation requirements across different production scales.
- Cost Reduction in Manufacturing: The complete avoidance of expensive transition metal catalysts eliminates both the direct material costs of these reagents and the substantial downstream processing expenses required to remove metal residues from final products. This streamlined approach reduces overall manufacturing costs through simplified process design that requires fewer unit operations while maintaining high product quality standards essential for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The utilization of readily available starting materials including commercially accessible aldehydes for hydrazone preparation creates a robust supply chain foundation with multiple sourcing options for critical raw materials. This flexibility significantly reduces vulnerability to single-source dependencies while enabling rapid response to changing demand patterns through simplified material procurement processes that don't require specialized handling or storage conditions.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production without requiring fundamental modifications to reaction parameters or equipment configuration. The elimination of hazardous reagents and reduction in purification steps substantially lowers environmental impact while simplifying regulatory compliance through reduced waste generation and safer operating conditions that align with green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation in pharmaceutical intermediate manufacturing. These insights have been developed through comprehensive evaluation of the reaction mechanism, scalability parameters, and commercial production requirements specific to triazole-based pharmaceutical intermediates.
Q: How does this method eliminate heavy metal contamination concerns in pharmaceutical manufacturing?
A: The iodine-catalyzed process completely avoids transition metal catalysts that typically require extensive purification steps to remove toxic residues, thereby ensuring cleaner final products that meet stringent pharmaceutical purity requirements without additional processing.
Q: What advantages does this synthesis offer for supply chain reliability compared to conventional methods?
A: By utilizing readily available starting materials like sodium acetate and commercially accessible aldehydes for hydrazone preparation, this method eliminates dependency on specialized reagents and complex handling procedures, significantly improving supply chain robustness and reducing lead times.
Q: How does the absence of anhydrous/anaerobic requirements impact manufacturing scalability?
A: The elimination of strict moisture and oxygen-free conditions simplifies reactor design and operation, enabling seamless scale-up from laboratory to commercial production without requiring specialized equipment or additional process validation steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates while maintaining stringent purity specifications through our state-of-the-art QC labs. Our technical team has successfully implemented this patented iodine-catalyzed methodology across multiple client projects, demonstrating consistent ability to deliver high-purity triazole intermediates meeting exacting pharmaceutical standards through rigorous process validation and quality control protocols. We understand the critical importance of reliable supply for time-sensitive drug development programs and have established robust manufacturing capabilities specifically designed for complex heterocyclic compound production.
Leverage our expertise with a Customized Cost-Saving Analysis tailored to your specific triazole intermediate requirements by contacting our technical procurement team today to request detailed COA data and route feasibility assessments for your next-generation pharmaceutical development program.