Advanced Mo-Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Mo-Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which are critical for modulating drug properties. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing significant gaps in current synthetic capabilities. This technology leverages a unique molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. The significance of this development cannot be overstated, as 1,2,4-triazole scaffolds are ubiquitous in bioactive molecules, serving as key structural motifs in renowned drugs such as Sitagliptin and various antifungal agents. By enabling the efficient construction of these cores under mild conditions, this patent offers a transformative pathway for reliable pharmaceutical intermediate supplier networks aiming to enhance their portfolio of fluorinated building blocks.
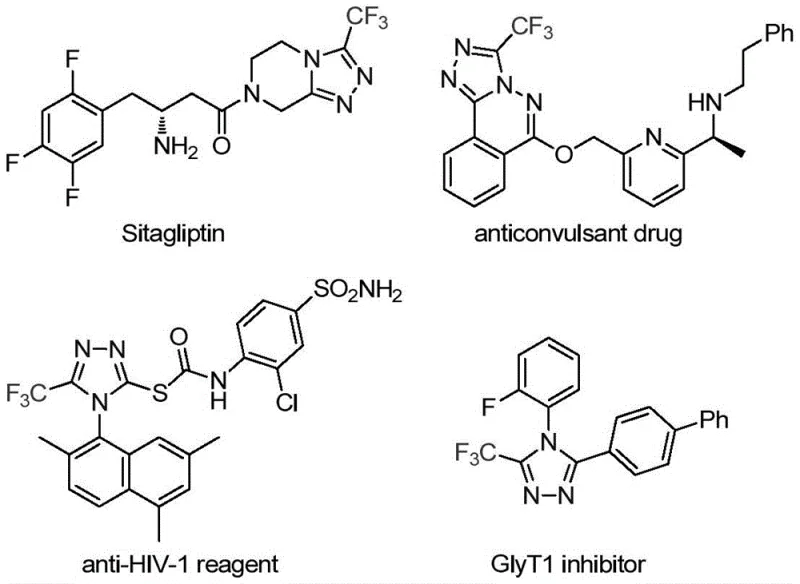
The integration of the trifluoromethyl group into these heterocyclic systems is strategically vital for modern medicinal chemistry. Fluorination often leads to marked improvements in electronegativity, bioavailability, metabolic stability, and lipophilicity, thereby optimizing the pharmacokinetic profile of candidate drugs. Traditional approaches to synthesizing these specific 3,4-disubstituted triazoles have been fraught with limitations, often requiring harsh conditions or complex multi-step sequences that hinder commercial viability. The disclosed method overcomes these barriers by utilizing cheap and easily obtainable starting materials, ensuring that the production of high-purity OLED material precursors or pharmaceutical intermediates remains economically feasible. This innovation represents a significant leap forward in cost reduction in API manufacturing, providing a streamlined route that minimizes waste and maximizes yield through a single-pot transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has relied heavily on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These legacy processes are frequently characterized by severe operational drawbacks, including the requirement for highly reactive and potentially hazardous hydrazine derivatives which pose significant safety risks in a production environment. Furthermore, alternative copper-catalyzed multi-component reactions involving diazonium salts and trifluorodiazoethane often suffer from poor atom economy and the generation of substantial toxic byproducts. The reliance on unstable diazo compounds necessitates specialized equipment and rigorous safety protocols, drastically increasing the capital expenditure and operational complexity for manufacturers. Additionally, many existing methods struggle to achieve high regioselectivity, leading to difficult purification challenges and reduced overall yields that negatively impact the supply chain continuity for critical drug substances.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the novel approach detailed in the patent utilizes a sophisticated Mo/Cu co-catalytic system that operates under remarkably mild conditions. By employing molybdenum hexacarbonyl as a metal activator alongside cuprous acetate, the reaction efficiently promotes the [3+2] cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitriles. This methodology eliminates the need for dangerous diazo reagents or unstable hydrazines, replacing them with stable, commercially available solids that can be handled with standard safety measures. The reaction proceeds smoothly in common organic solvents like THF at temperatures ranging from 70°C to 90°C, which are easily achievable in standard glass-lined reactors without the need for cryogenic cooling or extreme heating. This shift not only simplifies the operational procedure but also significantly broadens the substrate scope, allowing for the introduction of diverse functional groups such as halogens, alkyls, and alkoxy groups without compromising reaction efficiency.
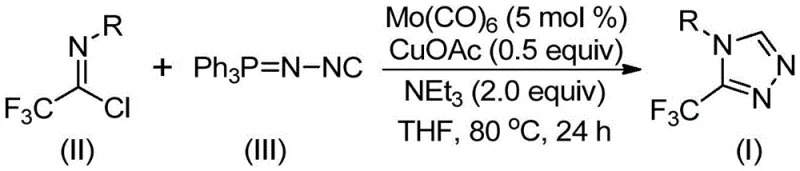
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the synergistic interaction between the molybdenum and copper species. Initially, the molybdenum hexacarbonyl acts as a crucial activator for the functionalized isonitrile, forming a transient metal-isocyanide complex that enhances the nucleophilicity of the carbon center. Simultaneously, the cuprous acetate facilitates the activation of the trifluoroethylimidoyl chloride, promoting the initial attack that leads to the formation of the five-membered ring intermediate. This dual-catalyst system ensures that the energy barrier for the cycloaddition is significantly lowered, allowing the reaction to proceed at moderate temperatures where side reactions are minimized. The subsequent elimination of triphenylphosphine oxide, driven by the presence of water in the system or during workup, drives the equilibrium towards the final 3-trifluoromethyl-substituted 1,2,4-triazole product. Understanding this mechanism is essential for R&D teams looking to optimize the process further, as it highlights the critical role of ligand exchange and metal coordination in achieving high turnover numbers.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy thermal cyclizations. The concerted nature of the metal-promoted cycloaddition reduces the likelihood of random polymerization or decomposition of the sensitive trifluoromethyl group, which is prone to defluorination under harsh basic or thermal stress. The use of triethylamine as a mild base further mitigates the risk of hydrolyzing the imidoyl chloride starting material before it can participate in the cycle. Consequently, the crude reaction mixture typically contains fewer structurally related impurities, simplifying the downstream purification process. For quality control laboratories, this means that achieving stringent purity specifications becomes more straightforward, reducing the burden on analytical resources and ensuring that the final active pharmaceutical ingredient meets regulatory standards with greater consistency and reliability.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a straightforward charging sequence where all catalysts, bases, and reactants are combined in a single vessel, eliminating the need for slow addition pumps or complex temperature ramping profiles. The use of molecular sieves in the reaction mixture helps to scavenge trace moisture that could otherwise degrade the imidoyl chloride, ensuring consistent performance across different batches. Following the reaction period of 18 to 30 hours, the workup is exceptionally simple, involving filtration to remove metal residues and molecular sieves, followed by standard silica gel chromatography. This operational simplicity is a key factor in reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the man-hours required for process execution and monitoring. Detailed standardized synthesis steps for this procedure are provided in the guide below to assist technical teams in rapid adoption.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and perform column chromatography purification to isolate the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology presents a compelling value proposition centered on cost efficiency and supply security. The primary drivers of cost in chemical manufacturing are often the raw materials and the complexity of the processing steps; this innovation addresses both simultaneously by utilizing commodity chemicals as starting materials. Trifluoroethylimidoyl chloride and functionalized isonitriles are readily sourced from global chemical suppliers, reducing the risk of single-source dependency that often plagues specialty intermediate supply chains. Furthermore, the elimination of expensive transition metal catalysts like palladium or rhodium in favor of abundant molybdenum and copper salts results in substantial cost savings on catalyst procurement. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme heating or cooling, contributing to a reduced carbon footprint and lower utility costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived principally from the use of inexpensive, earth-abundant metal catalysts and the avoidance of hazardous reagents that require special disposal protocols. By replacing costly noble metals with copper and molybdenum, the direct material cost of the catalytic system is drastically simplified, leading to a more favorable margin structure for high-volume production. Additionally, the high reaction efficiency and yields reported in the patent examples mean that less raw material is wasted, maximizing the output from every batch. The simplified workup procedure further reduces the consumption of solvents and silica gel during purification, compounding the savings across the entire manufacturing lifecycle. These factors collectively ensure that the cost of goods sold for these triazole intermediates remains competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability of starting materials and the robustness of the manufacturing process. Since the key reagents for this synthesis are commercially available off-the-shelf products, there is no need to rely on custom synthesis of exotic precursors which can introduce long lead times and logistical bottlenecks. The tolerance of the reaction to various functional groups allows manufacturers to source a wider range of substituted aromatic amines and isonitriles, providing flexibility to switch suppliers if market disruptions occur. Moreover, the scalability of the reaction from gram to multi-kilogram scales without loss of efficiency ensures that supply can be ramped up quickly to meet sudden spikes in demand from downstream pharmaceutical clients. This reliability is essential for maintaining continuous production schedules for life-saving medications.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this method is inherently designed for safe expansion. The absence of explosive diazo compounds and the use of moderate temperatures significantly lower the process safety index, making it easier to obtain regulatory approval for large-scale production facilities. From an environmental standpoint, the atom economy of the cycloaddition is superior to traditional condensation methods, generating less chemical waste per unit of product. The ability to recycle solvents like THF and the use of non-toxic metal salts align with modern green chemistry principles, helping companies meet increasingly strict environmental regulations. This compliance reduces the risk of shutdowns due to environmental violations and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis technology. These answers are derived directly from the experimental data and disclosures within the patent documentation to provide accurate guidance for process chemists. Understanding these nuances is critical for successfully transferring the technology from the laboratory bench to commercial production lines. We encourage technical teams to review these points carefully when evaluating the feasibility of this route for their specific project requirements.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method accommodate diverse substrate functional groups?
A: Yes, the method demonstrates high tolerance for various substituents on the aryl ring, including methyl, methoxy, fluoro, chloro, and nitro groups, yielding products with high efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: The patent explicitly states that the method can be expanded to gram-level reactions and beyond, utilizing cheap and commercially available starting materials which facilitates industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced heterocyclic intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are fully equipped to implement the Mo/Cu co-catalyzed synthesis described in patent CN113307778A, leveraging our state-of-the-art facilities to deliver high-purity intermediates that meet stringent purity specifications. Our rigorous QC labs utilize advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that the material you receive is ready for immediate use in your drug discovery or manufacturing programs. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that drive your projects forward.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific applications. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this method for your supply chain. We are prepared to provide specific COA data and route feasibility assessments tailored to your target molecules, ensuring a seamless transition to a more efficient and cost-effective sourcing strategy. Contact us today to explore how our expertise in fluorinated triazole chemistry can support your long-term business goals.
