Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
Introduction to Next-Generation Triazole Synthesis
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-rich heterocycles, particularly those bearing fluorinated motifs which are pivotal in modern drug design. Patent CN114920707B discloses a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic routes. This technology leverages the dual functionality of N,N-dimethylformamide (DMF), utilizing it not merely as a solvent but as an active carbon source to drive the cyclization process. The significance of this approach is underscored by the prevalence of the 1,2,4-triazole scaffold in high-value therapeutics, ranging from antidiabetic agents like Sitagliptin to potent Factor IXa inhibitors and GlyT1 inhibitors.
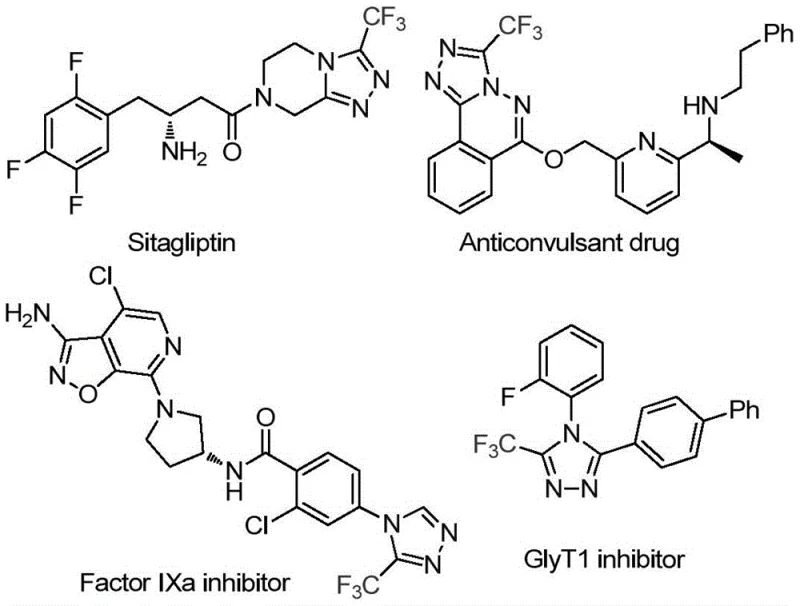
By integrating molecular iodine as a promoter under aerobic conditions, this invention offers a streamlined pathway that bypasses the need for complex catalysts or harsh environments. For R&D directors and procurement specialists, this represents a shift towards more sustainable and economically viable manufacturing processes. The ability to synthesize these core structures with high efficiency and minimal operational complexity positions this method as a superior alternative for producing high-purity pharmaceutical intermediates on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing trifluoromethyl-substituted 1,2,4-triazoles often suffer from significant logistical and economic drawbacks that hinder large-scale production. Conventional methodologies frequently rely on pre-functionalized starting materials that are expensive to procure and difficult to store, necessitating strict inventory control and specialized handling procedures. Furthermore, many existing protocols demand rigorous anhydrous and anaerobic conditions, requiring specialized equipment such as gloveboxes or Schlenk lines which drastically increase capital expenditure and operational overhead. The use of transition metal catalysts in older methods also introduces the risk of heavy metal contamination, necessitating costly purification steps to meet stringent regulatory limits for active pharmaceutical ingredients. Additionally, multi-step sequences often result in cumulative yield losses and generate substantial chemical waste, conflicting with modern green chemistry principles and increasing the overall environmental footprint of the manufacturing process.
The Novel Approach
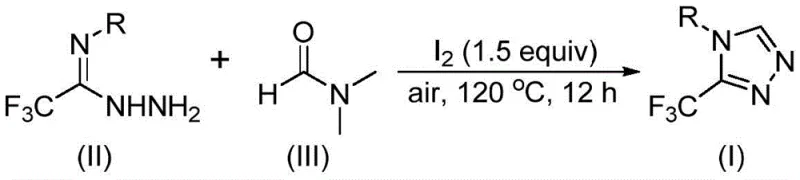
In stark contrast, the method described in CN114920707B revolutionizes the synthesis landscape by employing a tandem cyclization strategy that is both operationally simple and chemically elegant. This novel approach utilizes readily available trifluoroethyliminohydrazide and common organic solvent DMF as the sole carbon source, effectively merging the roles of solvent and reactant to streamline the reaction matrix. The reaction proceeds smoothly under an air atmosphere at moderate temperatures between 110°C and 130°C, eliminating the need for inert gas protection or moisture-sensitive setups. Molecular iodine serves as an efficient and inexpensive promoter to activate the DMF, facilitating the formation of the triazole ring through a cascade mechanism that avoids the use of precious metal catalysts. This simplification of reaction conditions not only reduces the technical barrier for execution but also enhances the safety profile of the process, making it an ideal candidate for cost reduction in API manufacturing where scalability and safety are paramount concerns.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a fascinating example of solvent-participatory synthesis, where the DMF molecule plays a critical role in constructing the heterocyclic core. Detailed analysis suggests that both the methyl and formyl groups of DMF can potentially serve as carbon sources, though the formyl pathway is often dominant in generating the specific 1,2,4-triazole architecture. In the proposed mechanism, the formyl group of DMF undergoes a condensation reaction with the hydrazide moiety of the starting material to generate a hydrazone intermediate. This is followed by an intramolecular cyclization event that eliminates a molecule of dimethylamine, driving the equilibrium towards the formation of the desired 3-trifluoromethyl-substituted 1,2,4-triazole product. Alternatively, when the N-methyl group acts as the carbon source, DMF reacts with iodine to form an amine salt species which subsequently engages in nucleophilic addition with the hydrazide. This pathway involves the elimination of nitrogen-methylformamide to generate an azadiene intermediate, which then undergoes oxidative aromatization to yield the final stable triazole ring system.
Understanding these mechanistic nuances is crucial for impurity control and process optimization during scale-up. The use of molecular iodine is particularly strategic as it facilitates the activation of the relatively inert DMF molecule, lowering the energy barrier for C-N bond formation without introducing toxic heavy metals. The oxidative aromatization step ensures the thermodynamic stability of the final product, minimizing the presence of partially cyclized byproducts that could complicate downstream purification. For quality assurance teams, this mechanistic clarity allows for better prediction of potential side reactions and the implementation of targeted analytical methods to monitor reaction progress. The robustness of this mechanism across various substrates, as evidenced by the successful synthesis of derivatives with diverse electronic properties, confirms the versatility of this catalytic system for generating high-purity heterocyclic compounds required in drug discovery pipelines.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis protocol is designed to be accessible for both laboratory research and industrial production environments. The procedure involves combining the trifluoroethyliminohydrazide substrate with molecular iodine in DMF, followed by heating under air to effect the cyclization. This straightforward setup minimizes the need for specialized training or equipment, allowing chemical teams to focus on substrate exploration and optimization rather than troubleshooting complex reaction conditions. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are outlined below to ensure reproducibility and safety.
- Combine molecular iodine (1.5 equivalents) and trifluoroethyliminohydrazide substrate in a reaction vessel with DMF serving as both solvent and reactant.
- Heat the reaction mixture to 110-130°C under an air atmosphere and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible benefits that directly impact the bottom line and operational resilience. By shifting away from exotic reagents and complex catalytic systems, manufacturers can significantly reduce the volatility associated with raw material sourcing and pricing. The reliance on commodity chemicals like DMF and iodine ensures a stable supply chain that is less susceptible to geopolitical disruptions or market fluctuations common with specialty fine chemicals. Furthermore, the simplified operational requirements translate into lower utility costs and reduced maintenance burdens on production facilities, enhancing the overall economic efficiency of the manufacturing campaign.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the dual use of DMF as both solvent and reactant drastically cut down on raw material expenses. Traditional methods often require stoichiometric amounts of costly coupling agents or ligands, whereas this process utilizes inexpensive molecular iodine in catalytic or near-stoichiometric quantities. The removal of heavy metal catalysts also obviates the need for expensive scavenging resins or complex purification protocols to meet residual metal specifications, resulting in substantial cost savings during the downstream processing phase. Additionally, the high atom economy of the reaction minimizes waste disposal costs, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key reagents, including DMF, iodine, and the hydrazide precursors, are commercially available in bulk quantities from multiple global suppliers. This redundancy in the supply base mitigates the risk of production stoppages due to single-source dependency, a critical factor for maintaining continuous manufacturing operations. The stability of the starting materials under ambient conditions further reduces logistics costs, as there is no need for cold chain shipping or specialized storage infrastructure. This reliability ensures that production schedules can be met consistently, supporting just-in-time manufacturing models and reducing inventory holding costs.
- Scalability and Environmental Compliance: The reaction's tolerance to air and moisture makes it inherently safer and easier to scale from kilogram to tonne levels without the engineering challenges associated with inert atmosphere reactors. This ease of scale-up accelerates the timeline from process development to commercial launch, allowing companies to respond faster to market demands. From an environmental perspective, the avoidance of toxic heavy metals and the use of a common solvent that can be potentially recovered and recycled align with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden and potential liability associated with hazardous waste management, fostering a more sustainable and socially responsible manufacturing practice.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for making informed decisions about process adoption and integration into existing manufacturing workflows.
Q: What are the key advantages of using DMF as a carbon source in this synthesis?
A: Using DMF eliminates the need for separate, expensive C1 building blocks. It acts simultaneously as the reaction solvent and the carbon donor, significantly simplifying the workup process and reducing raw material costs compared to traditional multi-component couplings.
Q: Does this reaction require strict anhydrous or anaerobic conditions?
A: No, one of the major operational benefits of this patented method is that it proceeds efficiently under ambient air atmosphere without the need for rigorous anhydrous conditions, making it highly suitable for large-scale industrial manufacturing.
Q: What is the substrate scope for the R-group in the trifluoroethyliminohydrazide?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens and trifluoromethyl, allowing for diverse derivative synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic route for producing high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this cost-effective methodology can be tailored to your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis and discuss your requirements for specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable and efficient manufacturing solutions.
