Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Novel Molybdenum-Copper Catalysis
Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Novel Molybdenum-Copper Catalysis
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which significantly enhance metabolic stability and lipophilicity. A groundbreaking development in this domain is detailed in Chinese patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses critical bottlenecks in synthesizing these privileged scaffolds, which are ubiquitous in bioactive molecules such as the antidiabetic drug Sitagliptin and various antifungal agents. By leveraging a unique molybdenum and copper co-catalytic system, this process enables the direct assembly of the triazole ring under remarkably mild conditions, offering a streamlined pathway for producing high-purity pharmaceutical intermediates.
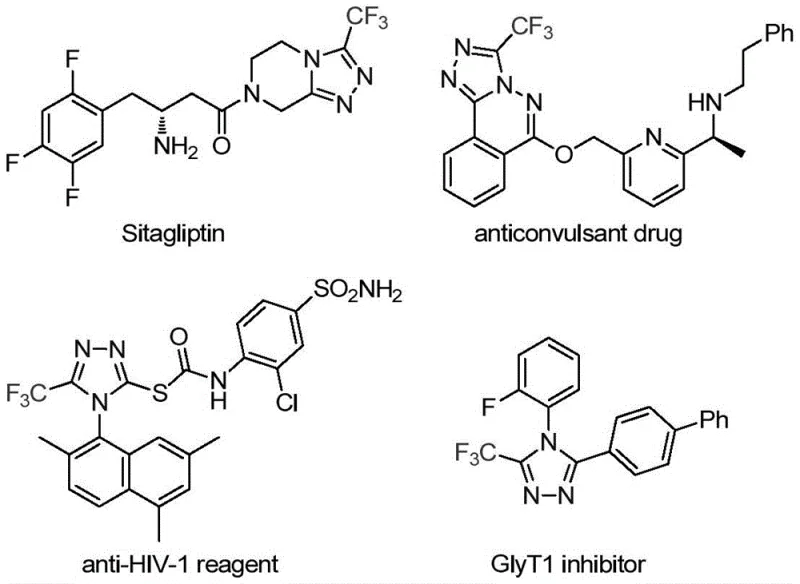
The strategic importance of the 1,2,4-triazole core cannot be overstated in modern drug discovery. As illustrated in the structural diversity of known therapeutics, the incorporation of a trifluoromethyl group at the 3-position further amplifies the pharmacological profile by modulating electronegativity and membrane permeability. For R&D directors and process chemists, the ability to access these structures reliably is paramount. The disclosed method not only simplifies the synthetic route but also expands the chemical space available for library synthesis, allowing for the rapid generation of analogs with varied electronic and steric properties essential for structure-activity relationship (SAR) studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on cumbersome multi-step sequences that often suffer from poor atom economy and harsh reaction conditions. Traditional routes frequently involve the cyclization of trifluoroacetyl hydrazine with amidine derivatives or the hydrazinolysis of oxazolinone precursors, processes that require stringent temperature controls and generate significant amounts of hazardous waste. Furthermore, copper-catalyzed multi-component reactions utilizing diazonium salts or trifluorodiazoethane pose serious safety risks due to the explosive nature of diazo compounds, making them unsuitable for large-scale commercial manufacturing. These legacy methods often result in low overall yields and complex purification challenges, driving up the cost of goods and extending lead times for critical API intermediates.
The Novel Approach
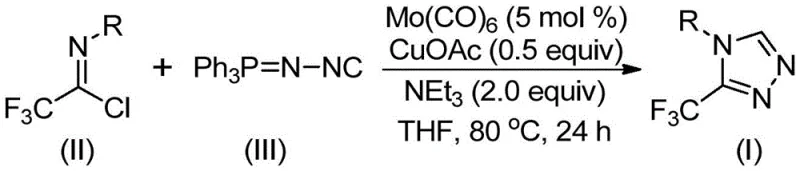
In stark contrast, the innovative protocol described in the patent utilizes a direct cycloaddition strategy between trifluoroethylimidoyl chloride and functionalized isonitriles, catalyzed by an inexpensive molybdenum-copper system. This approach eliminates the need for unstable diazo reagents and avoids the use of toxic hydrazines, significantly improving the safety profile of the operation. The reaction proceeds smoothly in common organic solvents like THF at moderate temperatures ranging from 70°C to 90°C, demonstrating exceptional functional group tolerance. As shown in the general reaction scheme below, this one-pot transformation efficiently constructs the heterocyclic core while expelling triphenylphosphine oxide, a byproduct that is easily removed during workup.
The versatility of this novel approach is evidenced by its compatibility with a wide array of substrates, including those bearing electron-donating and electron-withdrawing groups on the aromatic ring. Whether the substituent is a bulky tert-butyl group, a halogen like fluorine or chlorine, or even sensitive functionalities like nitro or thioether groups, the system maintains high conversion rates. This broad substrate scope is a game-changer for medicinal chemists who require flexible synthetic tools to explore diverse chemical spaces without redesigning the entire synthetic route for each new analog, thereby accelerating the drug discovery timeline.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cyclization
The success of this transformation lies in the synergistic interaction between the molybdenum and copper catalysts, which orchestrate the activation of distinct reaction partners. Mechanistically, molybdenum hexacarbonyl acts as a potent activator for the functionalized isonitrile, forming a reactive metal-carbene or metal-isocyanide complex that primes the molecule for nucleophilic attack. Simultaneously, the cuprous acetate facilitates the [3+2] cycloaddition step, guiding the alignment of the trifluoroethylimidoyl chloride with the activated isonitrile species. This dual-activation mode lowers the energy barrier for ring closure, allowing the formation of the five-membered triazole intermediate under much milder thermal conditions than would be required in a purely thermal process.
Following the initial cycloaddition, the reaction pathway involves a crucial elimination step where triphenylphosphine oxide is expelled from the intermediate structure. This step is driven by the thermodynamic stability of the phosphine oxide bond and is facilitated by the presence of water or moisture within the reaction system, often introduced via the molecular sieves or ambient conditions. The final aromatization yields the stable 3-trifluoromethyl-1,2,4-triazole product. Understanding this mechanism is vital for process optimization, as it highlights the importance of maintaining the correct stoichiometric balance between the catalysts and the base, triethylamine, to ensure the continuous turnover of the catalytic cycle and prevent catalyst deactivation.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol is designed to be operationally simple, utilizing commercially available starting materials that do not require specialized synthesis prior to use. The following guide outlines the standardized procedure derived from the patent examples, ensuring reproducibility and safety for technical teams looking to adopt this methodology for their specific intermediate needs.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in THF solvent.
- Heat the reaction mixture to a temperature range of 70-90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that directly impact the bottom line and operational resilience. By replacing hazardous and expensive reagents with commodity chemicals, the overall cost structure of the manufacturing process is significantly optimized. The elimination of complex multi-step sequences reduces the number of unit operations, which in turn minimizes solvent consumption, energy usage, and labor costs associated with isolation and purification. This streamlined workflow translates into a more competitive pricing model for the final API intermediate, providing a strategic advantage in cost-sensitive markets.
- Cost Reduction in Manufacturing: The utilization of inexpensive catalysts such as cuprous acetate and molybdenum hexacarbonyl, combined with readily available organic solvents like THF, drastically lowers the raw material expenditure. Furthermore, the high reaction efficiency and selectivity reduce the formation of difficult-to-separate impurities, minimizing the loss of valuable material during purification and enhancing the overall mass balance of the process.
- Enhanced Supply Chain Reliability: Since all key starting materials, including the functionalized isonitrile and trifluoroethylimidoyl chloride precursors, are commercially accessible or easily synthesized from bulk chemicals, the risk of supply disruption is markedly reduced. This reliance on stable, non-proprietary feedstocks ensures a consistent supply of the triazole intermediate, safeguarding production schedules against the volatility often associated with specialized reagents.
- Scalability and Environmental Compliance: The mild reaction conditions (70-90°C) and the absence of explosive diazo compounds make this process inherently safer and easier to scale from gram to kilogram and eventually to tonnage levels. The simplified workup procedure, involving standard filtration and chromatography, generates less hazardous waste compared to traditional hydrazine-based routes, facilitating easier compliance with increasingly stringent environmental regulations and reducing waste disposal costs.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope, optimization, and downstream processing. These insights are drawn directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what can be expected during technology transfer and scale-up activities.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies using THF as the preferred organic solvent at temperatures between 70°C and 90°C. The reaction typically requires 18 to 30 hours to reach completion, utilizing a dual catalytic system of Mo(CO)6 and CuOAc with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the methodology demonstrates excellent substrate tolerance. It successfully accommodates various substituents including alkyl groups (methyl, t-butyl), halogens (fluoro, chloro), alkoxy groups, and even electron-withdrawing groups like nitro and methylthio, yielding products with good to excellent efficiency.
Q: Is the purification process complex for large-scale manufacturing?
A: The post-processing is designed for simplicity. It involves standard filtration followed by silica gel adsorption and column chromatography. The use of common solvents and the absence of difficult-to-remove heavy metal residues simplify the downstream processing compared to traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly analyzed the capabilities of this molybdenum-copper co-catalyzed system and is fully prepared to support your projects from early-stage development through to commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your pipeline. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can accelerate your time-to-market and enhance the profitability of your final drug products.
