Revolutionizing Triazole Synthesis: A Cost-Effective Route for High-Purity Pharmaceutical Intermediates
Revolutionizing Triazole Synthesis: A Cost-Effective Route for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust, scalable, and economically viable synthetic routes for constructing privileged heterocyclic scaffolds, particularly those containing fluorine motifs which are ubiquitous in modern drug design. A groundbreaking advancement in this domain is detailed in Chinese Patent CN114920707B, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift by utilizing the common organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source for the ring construction. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: it transforms a ubiquitous, low-cost commodity chemical into a critical building block for high-value bioactive molecules, thereby addressing key pain points regarding raw material availability and process complexity in the synthesis of complex pharmaceutical intermediates.
The significance of the 1,2,4-triazole core cannot be overstated, as it serves as the structural backbone for a diverse array of therapeutic agents, including factor IXa inhibitors and GlyT1 inhibitors, as well as agrochemical fungicides. The introduction of a trifluoromethyl group at the 3-position further enhances the metabolic stability and lipophilicity of these candidates, making them highly desirable in medicinal chemistry campaigns. By leveraging the dual functionality of DMF, this patented methodology circumvents the traditional reliance on expensive, hazardous, or difficult-to-handle one-carbon synthons. This strategic utilization of solvent-as-reagent aligns perfectly with the principles of green chemistry and process intensification, offering a streamlined pathway that reduces the overall step count and waste generation associated with conventional triazole synthesis.
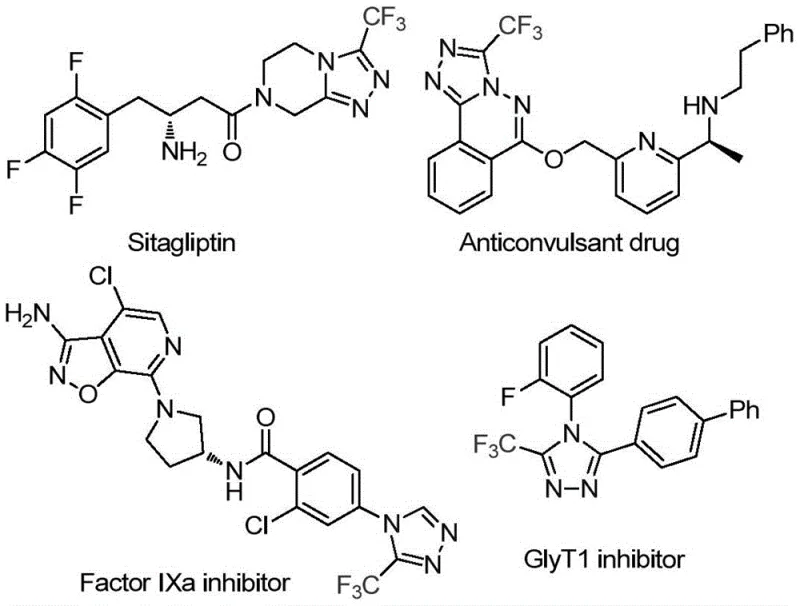
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder large-scale manufacturing and cost-effective production. Traditional routes often necessitate the use of pre-functionalized hydrazines or nitriles that require multi-step preparation from expensive starting materials, leading to a cumulative loss in yield and a significant increase in the cost of goods sold (COGS). Furthermore, many established protocols rely on harsh reaction conditions, such as strong acids, high pressures, or the use of toxic heavy metal catalysts that leave behind difficult-to-remove impurities, posing severe risks to product quality and regulatory compliance. The requirement for strictly anhydrous and oxygen-free environments in many of these legacy methods adds another layer of operational burden, demanding specialized equipment like gloveboxes or Schlenk lines, which drastically increases capital expenditure and limits the feasibility of scaling these processes to multi-ton quantities required for commercial API manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the method disclosed in CN114920707B introduces a remarkably simple yet powerful strategy that utilizes molecular iodine as a promoter to activate DMF for tandem cyclization. This approach eliminates the need for exotic reagents, relying instead on the synergy between readily available trifluoroethyliminohydrazide and the solvent DMF itself. The reaction proceeds smoothly under an air atmosphere at moderate temperatures ranging from 110 to 130°C, removing the necessity for inert gas protection and allowing for operation in standard glass-lined reactors. This simplicity translates directly into operational excellence, as the process tolerates a wide range of functional groups on the aromatic ring, including alkyl, alkoxy, alkylthio, and halogen substituents, without compromising the integrity of the final triazole scaffold. The ability to generate the heterocyclic core in a single pot with high atom economy represents a substantial leap forward in process chemistry, enabling manufacturers to achieve cost reduction in API manufacturing through simplified logistics and reduced waste disposal costs.
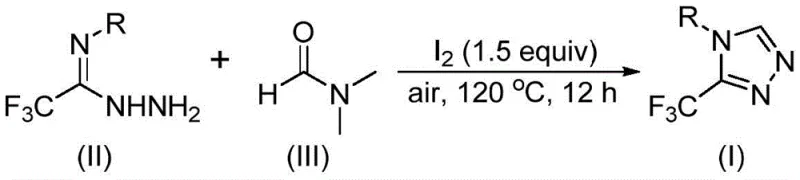
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
From a mechanistic perspective, the elegance of this transformation lies in the versatile activation of DMF by molecular iodine, which facilitates two distinct pathways for carbon incorporation depending on which fragment of the DMF molecule participates in the reaction. In the first plausible pathway, the formyl group of DMF acts as the carbon source, undergoing a condensation reaction with the hydrazide moiety of the trifluoroethyliminohydrazide to form a hydrazone intermediate. This intermediate subsequently undergoes intramolecular cyclization accompanied by the elimination of dimethylamine, ultimately yielding the desired 3-trifluoromethyl-substituted 1,2,4-triazole product. This pathway highlights the electrophilic nature of the DMF carbonyl carbon when activated, allowing for nucleophilic attack by the hydrazine nitrogen without the need for additional dehydrating agents.
Alternatively, the N-methyl group of DMF can serve as the carbon synthon through a more complex sequence involving oxidative activation. In this scenario, DMF initially reacts with iodine to generate an amine salt species, which then eliminates hydrogen iodide to become susceptible to nucleophilic addition by the trifluoroethyliminohydrazide. Following the elimination of N-methylformamide, an azadiene intermediate is generated, which then undergoes intramolecular nucleophilic addition and subsequent oxidative aromatization to furnish the target triazole. Understanding these mechanistic nuances is critical for R&D teams aiming to optimize reaction parameters, as it explains why other solvents like acetonitrile or toluene fail to support the transformation; they lack the specific structural features required to act as the C1 donor. This deep mechanistic understanding ensures that impurity profiles can be tightly controlled by monitoring the stoichiometry of iodine and the reaction temperature, ensuring the production of high-purity 1,2,4-triazole derivatives suitable for sensitive pharmaceutical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometric ratios and thermal profiles outlined in the patent data to maximize yield and minimize byproduct formation. The process is designed to be robust, utilizing a molar ratio of trifluoroethyliminohydrazide to iodine of approximately 1:1.5, which provides the optimal balance between promoting the reaction and avoiding excessive iodination side reactions. The reaction mixture is typically heated to 120°C for a duration of 12 hours, although the window of 10 to 15 hours allows for flexibility based on the specific electronic nature of the substrate. Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are provided below to assist process engineers in technology transfer.
- Charge a reaction vessel with molecular iodine, trifluoroethyliminohydrazide, and excess DMF solvent under an air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based cyclization technology offers transformative benefits that extend far beyond the laboratory bench, fundamentally altering the cost structure and reliability of the supply chain for triazole-based intermediates. The primary driver of value is the drastic simplification of the raw material basket; by replacing expensive, specialized C1 synthons with bulk commodity DMF, manufacturers can insulate themselves from the price volatility associated with niche fine chemicals. This substitution results in substantial cost savings, as DMF is produced on a massive global scale with a stable and predictable pricing history, ensuring long-term budget certainty for multi-year supply contracts. Furthermore, the elimination of sensitive reagents that require cold chain logistics or special handling permits reduces the administrative and logistical overhead associated with raw material intake and storage.
- Cost Reduction in Manufacturing: The economic impact of this process is profound because it removes the need for costly transition metal catalysts and anhydrous solvents, which are often significant line items in the bill of materials for heterocycle synthesis. By operating under air and using DMF as both solvent and reactant, the process inherently reduces the volume of waste solvent generated per kilogram of product, lowering disposal fees and environmental compliance costs. Additionally, the high reaction efficiency observed across a broad substrate scope means that less starting material is wasted on failed batches or low-yielding transformations, directly improving the overall material throughput and profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials that are not subject to the geopolitical or production bottlenecks often seen with specialized reagents. Since trifluoroethyliminohydrazide can be rapidly synthesized from common aromatic amines and hydrazine hydrate, the entire upstream supply chain is decentralized and robust, reducing the risk of single-source supplier failure. This redundancy ensures continuous production capability even during market disruptions, allowing pharmaceutical companies to maintain consistent inventory levels and meet critical delivery deadlines for their own downstream API synthesis without interruption.
- Scalability and Environmental Compliance: The scalability of this method is exceptional due to its tolerance for ambient conditions and the absence of pyrophoric or highly toxic reagents, making it ideal for commercial scale-up of complex pharmaceutical intermediates in standard multipurpose plants. The simplified work-up procedure, which involves basic filtration and chromatography, minimizes the generation of hazardous aqueous waste streams, aligning with increasingly stringent global environmental regulations. This ease of scale-up ensures that the transition from clinical trial material to commercial production is seamless, reducing the time-to-market for new drug candidates that rely on this trifluoromethyl-triazole scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-promoted cyclization technology, providing clarity for stakeholders evaluating its integration into their existing manufacturing portfolios. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, ensuring that decision-makers have access to accurate and actionable information regarding process feasibility and product quality.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the essential carbon source (C1 synthon), eliminating the need for expensive, specialized one-carbon reagents and significantly simplifying the raw material supply chain.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, the method described in patent CN114920707B operates effectively under ambient air atmosphere without the need for rigorous anhydrous conditions, greatly reducing operational complexity and equipment costs.
Q: What is the substrate scope for the R-group in this reaction?
A: The reaction demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating groups like ethyl and methylthio, as well as electron-withdrawing groups like fluoro and trifluoromethyl, yielding products with moderate to good efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
As the demand for fluorinated heterocycles continues to surge in the development of next-generation therapeutics, partnering with an experienced CDMO capable of executing complex cyclization chemistries is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from bench-scale optimization to full-scale manufacturing without compromising on quality or timeline. Our state-of-the-art facilities are equipped to handle the specific thermal and safety requirements of iodine-promoted reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required for GMP API synthesis.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this innovative synthetic route. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in pharmaceutical development.
