Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A recent technological breakthrough detailed in patent CN113105402B introduces a highly efficient preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, specifically those incorporating valuable trifluoromethyl groups. This innovation addresses long-standing challenges in heterocyclic chemistry by utilizing a non-metallic iodine-promoted system that operates under relatively mild conditions without the need for stringent anhydrous or oxygen-free environments. The significance of this development cannot be overstated for R&D directors and procurement specialists alike, as 1,2,4-triazole motifs are pervasive in high-value therapeutics, including antiviral agents like Maraviroc and antidiabetic drugs like Sitagliptin. By leveraging this novel methodology, manufacturers can achieve substantial improvements in process safety and environmental compliance while accessing a versatile library of fluorinated heterocycles essential for modern drug discovery.
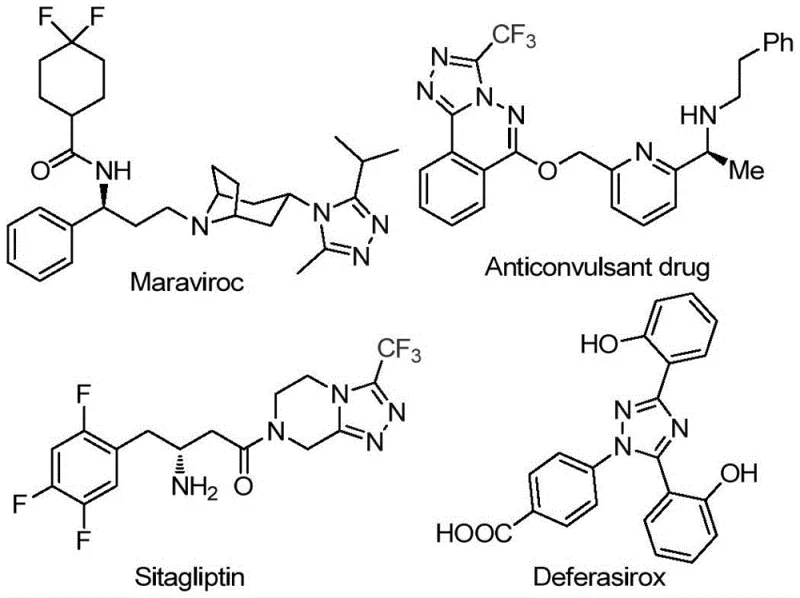
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, particularly those bearing both trifluoromethyl and acyl functionalities, has been fraught with significant technical and economic hurdles. Traditional methodologies often rely heavily on transition metal catalysts, which introduce complex downstream processing requirements to remove trace metal impurities to meet stringent regulatory limits for pharmaceutical products. Furthermore, many existing protocols necessitate the use of expensive, specialized reagents and require rigorous exclusion of moisture and oxygen, demanding sophisticated reactor setups and inert gas manifolds that drive up capital expenditure. The reliance on heavy metals also poses substantial environmental liabilities, complicating waste disposal and increasing the overall carbon footprint of the manufacturing process. Additionally, conventional routes frequently suffer from limited substrate scope, failing to tolerate diverse functional groups which restricts the chemical space available for medicinal chemists during lead optimization phases.
The Novel Approach
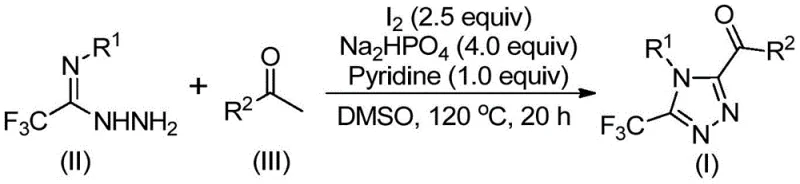
In stark contrast, the method disclosed in patent CN113105402B offers a paradigm shift by employing elemental iodine as a promoter in dimethyl sulfoxide (DMSO), effectively bypassing the need for transition metals entirely. This approach utilizes cheap and readily available starting materials, such as aryl ethyl ketones and trifluoroethylimide hydrazides, which are commoditized chemicals with stable supply chains. The reaction proceeds through a tandem sequence involving iodination and Kornblum oxidation, followed by cyclization, all within a single pot. Crucially, the process tolerates ambient atmospheric conditions, eliminating the operational burden of maintaining anhydrous or oxygen-free environments. This simplification not only reduces the complexity of the reaction setup but also enhances the safety profile of the operation, making it an ideal candidate for scale-up in standard multipurpose chemical reactors without requiring specialized metallurgy or extensive safety interlocks.
Mechanistic Insights into Iodine-Promoted Cyclization and Kornblum Oxidation
The mechanistic pathway of this transformation is a sophisticated interplay of oxidative functionalization and heterocyclization driven by the unique properties of the iodine-DMSO system. Initially, the aryl ethyl ketone undergoes an iodination event facilitated by elemental iodine, generating an alpha-iodo ketone intermediate. In the presence of DMSO, this intermediate is subjected to a Kornblum oxidation, converting the alpha-iodo moiety into a carbonyl group to form an aryl diketone species in situ. This oxidative step is critical as it generates the necessary 1,3-dicarbonyl equivalent required for subsequent ring closure. The trifluoroethylimide hydrazide then acts as a bidentate nucleophile, condensing with the newly formed diketone to establish the hydrazone linkage. Under the thermal conditions provided (110-130°C) and in the presence of the base system (sodium dihydrogen phosphate and pyridine), an intramolecular cyclization occurs. This final step closes the five-membered triazole ring, expelling water and establishing the aromatic 1,2,4-triazole core with high regioselectivity.
From an impurity control perspective, this mechanism offers distinct advantages due to the clean nature of the byproducts. The primary byproducts are inorganic salts and water, which are easily removed during the aqueous workup or filtration steps described in the patent. The absence of metal-ligand complexes means there is no risk of metal-catalyzed side reactions or difficult-to-remove organometallic residues that often plague transition metal-catalyzed couplings. The use of pyridine and sodium dihydrogen phosphate serves to buffer the reaction medium, preventing the degradation of acid-sensitive functional groups on the aromatic rings while facilitating the dehydration steps necessary for aromatization. This controlled environment ensures that the final product, the 3,4,5-trisubstituted 1,2,4-triazole, is obtained with high purity, minimizing the need for extensive recrystallization or preparative HPLC purification, which are cost-prohibitive at large scales.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for process chemistry teams looking to rapidly generate libraries of fluorinated triazoles. The protocol involves a sequential addition strategy where the aryl ethyl ketone and a portion of iodine are first heated in DMSO to effect the initial oxidation. Subsequently, the remaining reagents, including the hydrazide, base, and additional iodine, are introduced to drive the cyclization to completion. The reaction temperatures are moderate, ranging from 90°C to 130°C, which are well within the operating range of standard glass-lined or stainless steel reactors. Post-reaction processing is straightforward, involving simple filtration to remove inorganic salts followed by silica gel column chromatography for final purification. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route presents a compelling value proposition centered on cost efficiency and supply security. The elimination of precious metal catalysts such as palladium, rhodium, or copper removes a significant variable cost component from the bill of materials, while simultaneously eradicating the need for expensive scavenger resins or activated carbon treatments used for metal removal. This translates directly into a leaner cost structure for the manufacturing of these critical pharmaceutical intermediates. Furthermore, the reliance on commodity chemicals like acetophenones and elemental iodine ensures that raw material sourcing is robust and less susceptible to the geopolitical volatility often associated with specialized catalytic ligands or rare earth metals.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the downstream processing workflow. By avoiding heavy metal catalysts, manufacturers save significantly on the costs associated with wastewater treatment and hazardous waste disposal, as the effluent profile is much cleaner. The ability to run the reaction without strict anhydrous conditions reduces energy consumption related to solvent drying and nitrogen purging, further lowering the utility costs per kilogram of product. Additionally, the high atom economy of the cyclization step minimizes raw material waste, ensuring that a larger proportion of the input mass is converted into valuable product rather than discarded byproducts.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for any long-term production campaign, and this method excels by utilizing feedstocks that are produced on a multi-ton scale globally. Aryl ethyl ketones and iodine are staple chemicals in the fine chemical industry, meaning supply disruptions are rare compared to custom-synthesized catalysts. This reliability allows for better inventory planning and reduces the need for safety stock holdings, freeing up working capital. The robustness of the reaction conditions also means that production can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without requiring specialized equipment modifications, enhancing overall supply chain flexibility.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this protocol is inherently designed for expansion. The use of DMSO, a high-boiling polar aprotic solvent, facilitates heat management during the exothermic oxidation phases, reducing the risk of thermal runaway events. From an environmental standpoint, the process aligns well with green chemistry principles by avoiding toxic heavy metals and reducing the E-factor (mass of waste per mass of product). This compliance ease accelerates regulatory approvals and environmental permits, allowing companies to bring products to market faster while maintaining a strong corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of this iodine-promoted method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, simplifying post-reaction purification and reducing environmental compliance costs while maintaining high yields.
Q: Can this synthesis protocol be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is easily expandable to gram-level and beyond, utilizing cheap, commercially available starting materials like aryl ethyl ketones and elemental iodine.
Q: Does this process require strict anhydrous or oxygen-free conditions?
A: No, one of the primary operational benefits is that the reaction does not require anhydrous or oxygen-free conditions, significantly lowering equipment requirements and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocyclic synthesis in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the iodine-promoted pathway described in CN113105402B and validated its potential for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your R&D timeline and your commercial manufacturing budget.
