Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazole derivatives, which serve as critical scaffolds in numerous bioactive molecules. A significant technological advancement in this domain is detailed in patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. This innovation addresses long-standing challenges in heterocyclic chemistry by utilizing a non-metallic iodine-promoted system that operates under relatively mild conditions compared to traditional transition metal catalysis. The relevance of this chemical architecture is underscored by its presence in major pharmaceutical agents such as Maraviroc, Sitagliptin, and Deferasirox, highlighting the immense commercial value of mastering this synthesis. 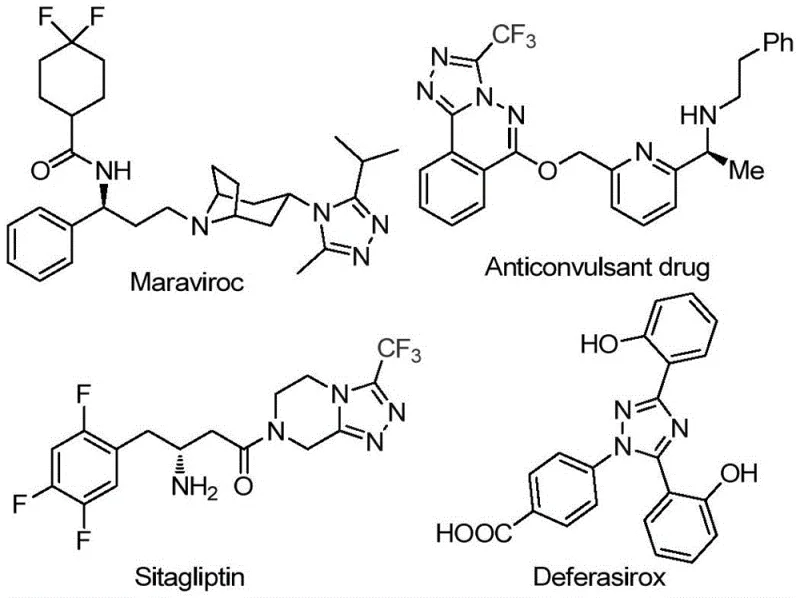 . For R&D directors and procurement specialists, understanding this patent provides a strategic advantage in sourcing high-purity intermediates while navigating the complexities of modern regulatory compliance and cost efficiency.
. For R&D directors and procurement specialists, understanding this patent provides a strategic advantage in sourcing high-purity intermediates while navigating the complexities of modern regulatory compliance and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings has relied heavily on methodologies that involve harsh reaction conditions and expensive catalytic systems. Traditional routes often necessitate the use of toxic heavy metal catalysts, which introduce significant downstream processing burdens related to residual metal removal and environmental waste disposal. Furthermore, many existing protocols require stringent anhydrous and oxygen-free environments, demanding specialized equipment such as Schlenk lines or gloveboxes that increase capital expenditure and operational complexity. These factors collectively contribute to elevated production costs and extended lead times, creating bottlenecks for supply chain managers who require consistent, large-volume deliveries. Additionally, the substrate scope in conventional methods is frequently limited, struggling to accommodate diverse functional groups without compromising yield or selectivity, thereby restricting the chemical space available for drug discovery teams.
The Novel Approach
In stark contrast, the methodology outlined in the patent data presents a paradigm shift by employing elemental iodine and dimethyl sulfoxide (DMSO) to facilitate the transformation. This approach eliminates the dependency on precious transition metals, thereby inherently reducing the cost of goods sold (COGS) and simplifying the purification workflow. The reaction proceeds through a tandem sequence involving iodination and Kornblum oxidation, followed by cyclization, all within a single pot. This telescoped process not only enhances atom economy but also significantly reduces solvent consumption and waste generation. The operational simplicity allows the reaction to be conducted without rigorous exclusion of air or moisture, making it exceptionally amenable to scale-up in standard stainless steel reactors. For procurement managers, this translates to a more reliable supply chain with reduced risk of batch failure due to environmental sensitivity.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this synthetic breakthrough lies in the dual role of iodine and DMSO, which work synergistically to activate the aryl ethanone substrate. Initially, the aryl ethanone undergoes an iodination event followed by a Kornblum oxidation to generate an aryl diketone intermediate in situ. This reactive species then engages in a condensation reaction with trifluoroethylimide hydrazide, forming a hydrazone intermediate that is primed for ring closure. 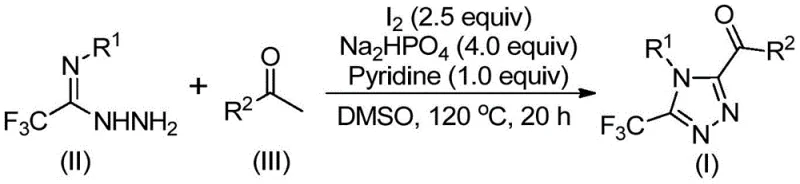 . The subsequent intramolecular cyclization is promoted by the basic environment provided by pyridine and sodium dihydrogen phosphate, driving the formation of the stable 1,2,4-triazole ring system. This mechanistic pathway avoids the high-energy barriers associated with metal-catalyzed C-H activation, offering a smoother energy profile that is easier to control on a large scale.
. The subsequent intramolecular cyclization is promoted by the basic environment provided by pyridine and sodium dihydrogen phosphate, driving the formation of the stable 1,2,4-triazole ring system. This mechanistic pathway avoids the high-energy barriers associated with metal-catalyzed C-H activation, offering a smoother energy profile that is easier to control on a large scale.
From an impurity control perspective, this mechanism offers distinct advantages for quality assurance teams. The absence of metal catalysts removes the risk of metal leaching, a critical parameter for pharmaceutical intermediates intended for final API synthesis. The byproducts of the reaction are primarily inorganic salts and iodine residues, which are straightforward to remove via aqueous workup and standard column chromatography or crystallization. The tolerance for various substituents on both the hydrazide and the ketone components allows for the generation of a diverse library of analogs, as evidenced by the successful synthesis of compounds I-1 through I-5 with varying electronic and steric properties. 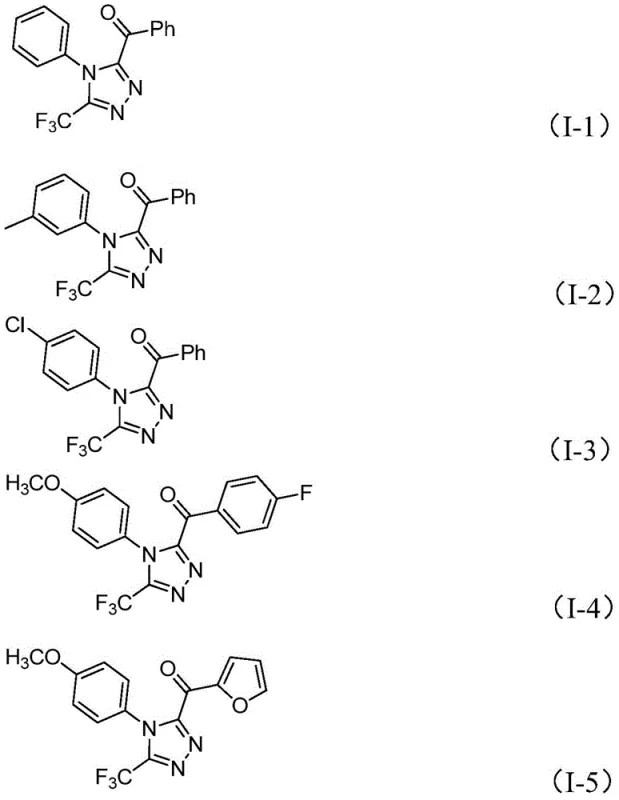 . This flexibility ensures that the process remains robust even when scaling up different variants of the core structure for structure-activity relationship (SAR) studies.
. This flexibility ensures that the process remains robust even when scaling up different variants of the core structure for structure-activity relationship (SAR) studies.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
Implementing this synthesis in a production environment requires careful attention to the stoichiometry and thermal profiles defined in the patent specifications. The process begins with the dissolution of aryl ethanone and elemental iodine in DMSO, followed by a controlled heating phase to initiate the oxidation. Subsequent addition of the hydrazine component and bases must be timed precisely to maximize the conversion to the desired triazole product. While the general concept is straightforward, the precise optimization of temperature ramps and addition rates is critical for maintaining high purity and yield. Detailed standardized synthesis steps see the guide below.
- Mix aryl ethanone and iodine in DMSO solvent and heat to 90-110°C for 4-6 hours to initiate iodination.
- Add sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization and obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this iodine-promoted methodology offers substantial strategic benefits. The primary driver of value is the drastic simplification of the raw material portfolio; elemental iodine and DMSO are commodity chemicals with stable global pricing and abundant availability, unlike specialized ligands or palladium catalysts that are subject to geopolitical supply shocks. This stability directly contributes to cost reduction in pharmaceutical intermediate manufacturing by insulating the production budget from volatile precious metal markets. Furthermore, the elimination of heavy metal catalysts removes the need for expensive scavenging resins or complex extraction protocols, significantly lowering the operational expenditure associated with downstream processing and waste management.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the removal of costly transition metals and the reduction in purification steps. By avoiding the use of expensive catalysts, the direct material costs are significantly lowered, and the simplified workup procedure reduces labor and solvent usage. The ability to run the reaction without strict inert gas protection further decreases utility costs related to nitrogen or argon consumption. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved due to the reliance on widely available bulk chemicals rather than niche reagents. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to moisture or oxygen sensitivity. This reliability ensures consistent lead times for high-purity pharmaceutical intermediates, allowing procurement managers to maintain lower safety stock levels while meeting production schedules. The scalability of the method from gram to multi-kilogram batches ensures that supply can be ramped up quickly to meet sudden increases in demand.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles by reducing toxic waste streams. The absence of heavy metals simplifies the disposal of reaction byproducts, reducing the regulatory burden and associated disposal costs. The process is inherently safer to operate at scale due to the lack of pyrophoric reagents or high-pressure hydrogenation steps. This ease of scale-up facilitates the commercial production of complex pharmaceutical intermediates, ensuring that the technology can transition smoothly from pilot plant to full-scale commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps bridge the gap between laboratory innovation and industrial application, ensuring that all stakeholders have a clear view of the process capabilities.
Q: Does this synthesis method require expensive heavy metal catalysts?
A: No, the patented process utilizes elemental iodine as a promoter, completely eliminating the need for toxic and costly transition metal catalysts, which simplifies purification and reduces environmental impact.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: The method is robust and does not require strict anhydrous or oxygen-free conditions, making it highly suitable for standard industrial reactor setups without specialized inert atmosphere equipment.
Q: Is this process scalable for commercial production volumes?
A: Yes, the patent explicitly states that the reaction can be easily expanded from gram-level laboratory synthesis to industrial scale production due to the simplicity of operation and availability of raw materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial supply chains. Our team of process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this iodine-promoted synthesis are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the exacting standards required by global pharmaceutical clients. Our commitment to quality ensures that the impurity profiles are tightly controlled, facilitating smoother regulatory filings for your downstream applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for high-purity pharmaceutical intermediates.
