Optimizing Remdesivir Commercial Production Through Novel One-Pot Synthetic Strategies
The global pharmaceutical landscape has witnessed an unprecedented demand for effective antiviral therapeutics, with Remdesivir standing out as a critical intervention for SARS-CoV-2 infections. As the industry strives to meet this demand, the efficiency of the supply chain relies heavily on the robustness of the underlying chemical manufacturing processes. Patent CN115947759A introduces a transformative preparation method that addresses longstanding bottlenecks in Remdesivir synthesis. This innovation shifts the paradigm from multi-step, protection-heavy sequences to a streamlined one-pot reaction strategy. By leveraging transient boronic acid protection, the method circumvents the need for harsh acidic deprotection conditions that traditionally plague nucleoside analog synthesis. For R&D Directors and Supply Chain Heads, this represents a significant opportunity to enhance production throughput while maintaining the rigorous quality standards required for active pharmaceutical ingredients. The structural integrity of the final molecule is preserved more effectively, ensuring that the therapeutic efficacy remains uncompromised during scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
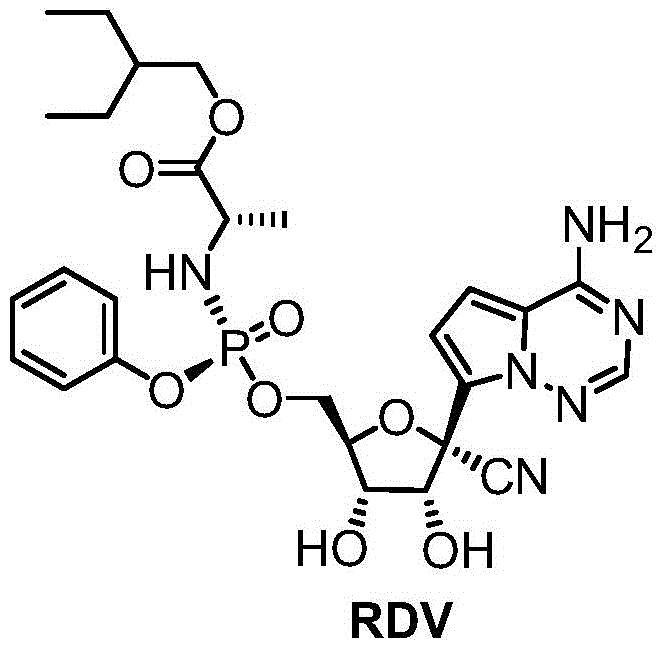
The Limitations of Conventional Methods
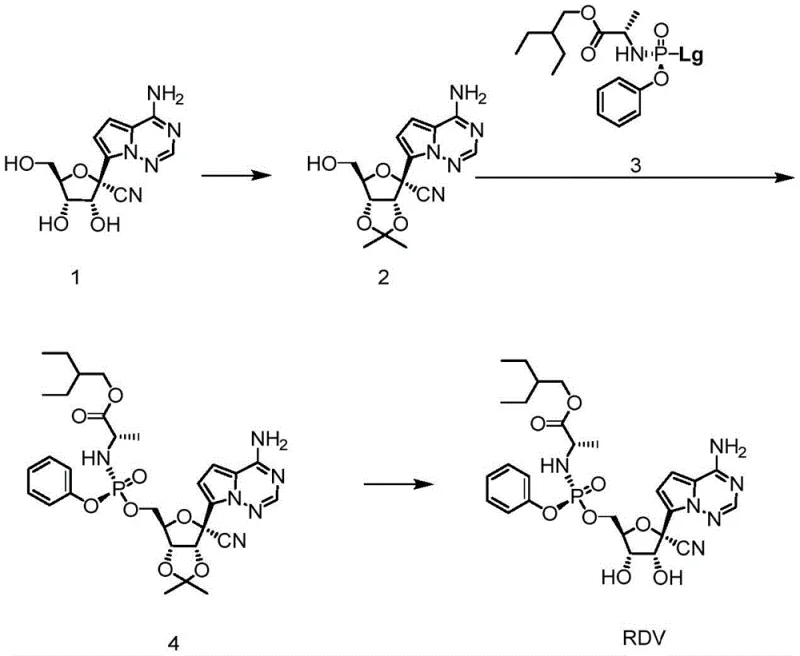
Historically, the industrial synthesis of Remdesivir has been hindered by the reliance on acetonide protection strategies for the ribose hydroxyl groups. In these conventional pathways, the 2′ and 3′ hydroxyl positions must be protected to prevent side reactions during the installation of the phosphoramidate prodrug moiety at the 5-position. This typically involves reacting the starting nucleoside with 2,2-dimethoxypropane under acidic conditions, followed by a separate deprotection step using hydrochloric acid after phosphorylation. These additional unit operations introduce significant inefficiencies, including increased solvent consumption, extended reaction times, and potential yield losses during isolation. Furthermore, the use of strong acids for deprotection poses a risk to the stability of the cyanopyrrolo[2,1-f][1,2,4]triazine base, potentially generating difficult-to-remove impurities that complicate downstream purification. For procurement managers, these complexities translate directly into higher raw material costs and longer lead times, creating vulnerabilities in the supply chain during periods of high demand.
The Novel Approach
In stark contrast, the methodology disclosed in CN115947759A eliminates the discrete protection and deprotection stages by utilizing a boronic acid-mediated transient protection strategy. This approach allows the reaction to proceed in a quasi-one-pot fashion, where the boronic acid reversibly binds to the diol system of the ribose sugar, shielding it during the phosphorylation event without requiring a separate chemical cleavage step. The process begins with the reaction of Compound 1 with a boric acid compound in toluene, forming a reactive intermediate that is immediately subjected to phosphorylation conditions. By removing the need for isolation of the protected intermediate and the subsequent acidic hydrolysis, the overall process mass intensity is drastically reduced. This simplification not only accelerates the manufacturing timeline but also enhances the safety profile of the operation by minimizing the handling of corrosive reagents. For a reliable API intermediate supplier, adopting such a route signifies a commitment to process intensification and operational excellence, directly addressing the need for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Boronic Acid Mediated Phosphorylation
The core chemical innovation lies in the ability of the boronic acid to act as a reversible protecting group under the specific reaction conditions employed. When Compound 1 is treated with phenylboronic acid or diphenylboronic anhydride in toluene at elevated temperatures ranging from 100-120°C, a cyclic boronate ester is formed in situ. This species effectively masks the 2′,3′-diol functionality, preventing it from participating in unwanted nucleophilic attacks during the subsequent addition of the phosphoramidate electrophile. The presence of magnesium chloride serves as a Lewis acid catalyst, activating the phosphoramidate reactant 9 and facilitating the nucleophilic attack by the 5-hydroxyl group. Following the phosphorylation, the reaction mixture is treated with DIPEA (N,N-Diisopropylethylamine) to scavenge generated acids and drive the equilibrium towards product formation. Crucially, the boronate ester is labile enough to be hydrolyzed during the aqueous workup phase, specifically when the pH is adjusted to the 4-8 range using citric acid. This seamless transition from protection to deprotection within the same reaction vessel is what defines the efficiency of this route, offering a sophisticated solution to a classic synthetic challenge.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-catalyzed deprotection. The avoidance of strong mineral acids prevents the depurination or degradation of the sensitive glycosidic bond, which is a common failure mode in nucleoside chemistry. Additionally, the use of mild organic bases like DIPEA and weak acids like citric acid for pH adjustment ensures that the chiral centers on the ribose ring remain intact, preserving the stereochemical purity essential for biological activity. The reaction conditions are optimized to balance the rate of boronate formation with the rate of phosphorylation, ensuring that the concentration of the free diol is kept low enough to prevent bis-phosphorylation side products. For R&D teams evaluating technology transfer, understanding this mechanistic nuance is vital for defining critical process parameters (CPPs) such as temperature control and reagent stoichiometry. The result is a cleaner reaction profile that simplifies the burden on analytical quality control labs, allowing for faster release of high-purity OLED material or pharmaceutical intermediates depending on the application context.
How to Synthesize Remdesivir Efficiently
The implementation of this synthesis route requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent examples to ensure reproducibility and optimal yield. The process begins with the activation of the nucleoside starting material in a non-polar solvent, followed by the sequential addition of phosphorylating agents and activators without intermediate isolation. This continuous flow of chemistry minimizes exposure to atmospheric moisture and oxygen, which can degrade sensitive reagents like the phosphoramidate. Detailed standardized synthesis steps see the guide below for specific operational parameters regarding mixing speeds, addition rates, and quenching protocols.
- React Compound 1 with a boric acid compound in toluene at 100-120°C to form a transient protected intermediate.
- Add acetonitrile, magnesium chloride, and phosphoramidate reactant 9, followed by DIPEA, and stir at room temperature.
- Adjust pH with citric acid, extract with ethyl acetate, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond simple chemical yield. The consolidation of multiple reaction steps into a single pot significantly reduces the equipment footprint required for production, allowing manufacturers to increase batch frequency without capital expenditure on new reactors. This intensification of the process leads to substantial cost savings by lowering utility consumption, reducing solvent waste disposal costs, and minimizing labor hours associated with charging and discharging multiple vessels. Furthermore, the elimination of harsh acidic deprotection steps reduces the corrosion burden on manufacturing equipment, extending asset life and decreasing maintenance downtime. These factors collectively contribute to a more resilient supply chain capable of responding rapidly to market fluctuations. By partnering with a manufacturer utilizing this technology, clients can secure a more stable supply of high-purity pharmaceutical intermediates with reduced risk of production delays caused by complex purification bottlenecks.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the acetonide protection and deprotection sequence. In traditional manufacturing, these steps account for a significant portion of raw material costs, particularly the specialized protecting group reagents and the large volumes of solvents required for their removal. By bypassing these stages, the new method drastically simplifies the material bill. Additionally, the higher overall yield achieved through the one-pot strategy means that less starting material is required to produce the same amount of final API, directly lowering the cost of goods sold. The reduction in waste generation also aligns with green chemistry principles, potentially lowering environmental compliance costs and waste treatment fees. This economic efficiency allows for more competitive pricing structures without compromising on the quality or purity of the final product delivered to the market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of multi-step syntheses, where a failure in any single unit operation can halt the entire production line. The simplified nature of this one-pot process reduces the number of potential failure points, thereby enhancing operational reliability. The reagents used, such as phenylboronic acid and magnesium chloride, are commodity chemicals with robust global supply networks, reducing the risk of raw material shortages compared to specialized protecting group reagents. Moreover, the shorter cycle time per batch means that inventory turnover is accelerated, allowing suppliers to maintain lower safety stock levels while still meeting delivery commitments. This agility is crucial for maintaining the flow of critical antiviral medications during public health emergencies, ensuring that patients receive timely access to life-saving treatments without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden challenges related to heat transfer and mixing efficiency. The exothermic nature of phosphorylation reactions can be difficult to manage in large vessels, but the controlled addition protocol described in this patent mitigates these risks. The absence of a separate acidic hydrolysis step eliminates the generation of large volumes of acidic wastewater, simplifying effluent treatment and reducing the environmental footprint of the facility. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking sustainable partners. The process is designed to be robust across different scales, from pilot plant trials to multi-ton commercial campaigns, ensuring that the quality attributes established during development are maintained throughout the product lifecycle. This scalability ensures that the supply of complex polymer additives or pharmaceutical intermediates can grow in tandem with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Remdesivir synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding technology adoption and procurement strategies.
Q: How does the boronic acid method improve upon traditional acetonide protection?
A: Traditional methods require harsh acidic conditions for deprotection which can degrade the sensitive nucleoside structure. The boronic acid method allows for transient protection under milder conditions, eliminating the need for a separate deprotection step and improving overall yield.
Q: What are the scalability benefits of this one-pot reaction design?
A: By consolidating multiple reaction steps into a single vessel, the process reduces equipment occupancy time, minimizes solvent usage, and lowers the risk of material loss during intermediate isolation, making it highly suitable for large-scale commercial manufacturing.
Q: Does this synthesis route impact the purity profile of the final Remdesivir API?
A: Yes, the reduction in synthetic steps and the avoidance of harsh acidic hydrolysis significantly reduce the formation of degradation impurities, resulting in a cleaner crude product that simplifies downstream purification and ensures stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality management. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative synthetic routes like the one described in CN115947759A are implemented with precision. Our facilities are equipped with state-of-the-art reaction vessels capable of handling the specific thermal and mixing requirements of boronic acid-mediated chemistries. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch meets the identity and potency standards required by global regulatory bodies. Our commitment to quality ensures that the Remdesivir supplied is not only chemically pure but also free from genotoxic impurities and residual solvents, safeguarding the integrity of your downstream formulation processes.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your margin structure. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. By choosing NINGBO INNO PHARMCHEM, you are securing a partnership dedicated to scientific innovation, operational reliability, and the consistent delivery of high-quality pharmaceutical intermediates.
