Optimized Industrial Synthesis of Remdesivir Intermediates for Scalable API Manufacturing
Optimized Industrial Synthesis of Remdesivir Intermediates for Scalable API Manufacturing
The global demand for effective antiviral therapeutics has necessitated the development of robust, scalable, and cost-efficient synthetic routes for critical active pharmaceutical ingredients (APIs). Patent CN112321589B discloses a novel and highly efficient synthesis method for Remdesivir (GS-5734) and its key intermediates, addressing significant bottlenecks found in earlier generations of manufacturing processes. This technology leverages a streamlined sequence starting directly from 4-chloropyrrolo[2,1-f][1,2,4]triazine, bypassing complex protection-deprotection sequences that traditionally plague nucleoside analogue synthesis. By optimizing the cyanation and ammonolysis steps, the disclosed method achieves superior reaction yields and product purity while drastically minimizing the generation of hazardous three wastes. For R&D directors and supply chain leaders, this represents a pivotal advancement in securing a reliable supply chain for antiviral drug substances.
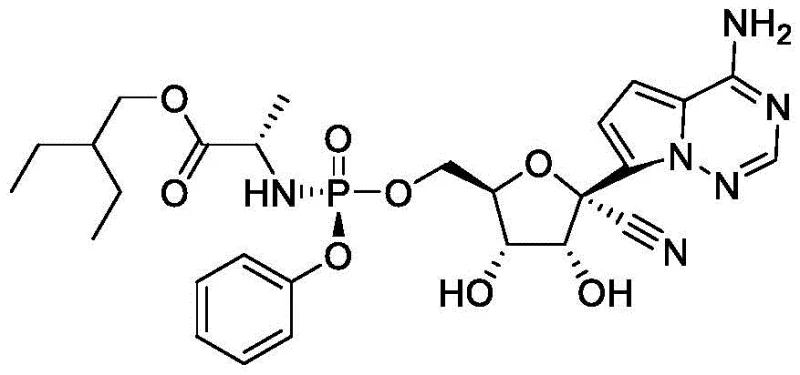
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Remdesivir has been fraught with inefficiencies that hinder cost-effective scale-up. Early routes, such as those described in the Journal of Medicinal Chemistry (2017), relied on a bromination-lithiation strategy to couple the heterocyclic base with the ribose sugar. This approach suffered from low yields during the addition and cyanation stages and required rigorous chiral separation techniques that were impractical for large-scale operations. Furthermore, subsequent improvements reported in 2016 utilized Grignard reagents for coupling, which consumed large equivalents of reagents and necessitated the use of trimethylsilyl (TMS) protection groups to prevent amino group interference. Perhaps most critically, these conventional methods relied heavily on boron trichloride for debenzylation, a reagent known for its extreme corrosivity and the generation of substantial acidic waste streams, complicating environmental compliance and increasing disposal costs.
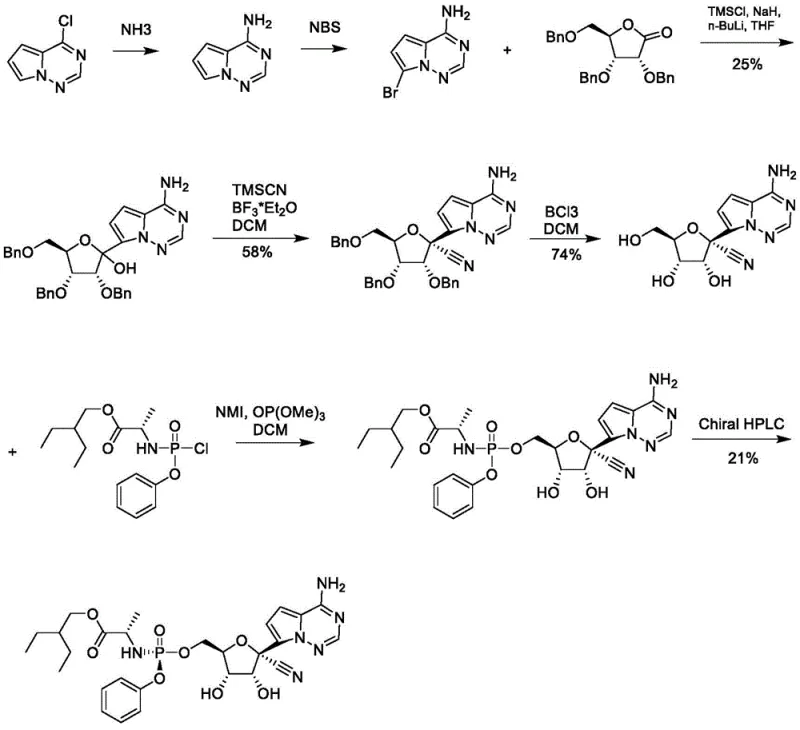
The Novel Approach
In stark contrast, the methodology outlined in CN112321589B introduces a paradigm shift by utilizing 4-chloropyrrolo[2,1-f][1,2,4]triazine directly as the starting material without additional active hydrogen protection. This strategic modification allows for a direct addition reaction with 2,3,5-tribenzyloxy-D-ribono-1,4-lactone, significantly enhancing the reaction yield by eliminating side reactions associated with unprotected amines. The subsequent cyanation is performed under Lewis acid catalysis, which proceeds with high efficiency and selectivity. Moreover, the debenzylation step is transformed from a hazardous chemical cleavage into a clean, catalytic hydrogenation using palladium on carbon, which not only improves safety but also facilitates the recovery and recycling of the precious metal catalyst. This holistic optimization results in a shorter, more linear synthetic route that is inherently more suitable for commercial amplification.
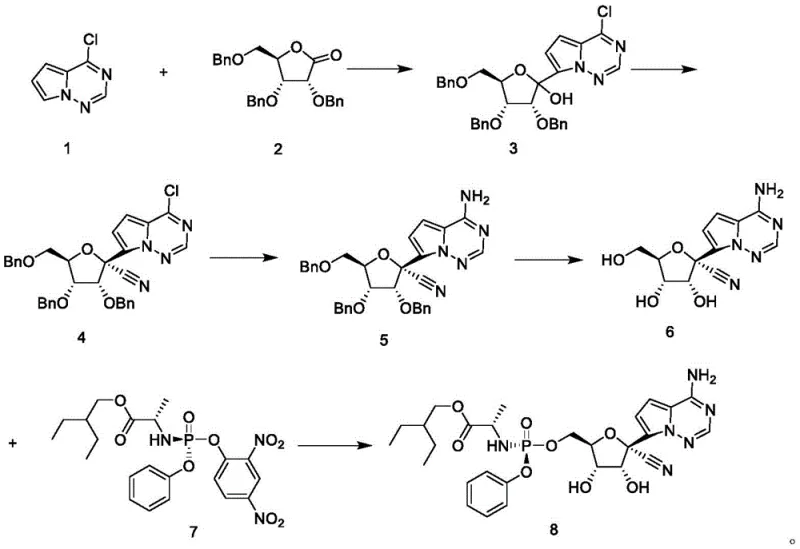
Mechanistic Insights into Lewis Acid Catalyzed Cyanation and Copper-Mediated Ammonolysis
The core chemical innovation of this process lies in the precise control of reactivity during the functionalization of the nucleoside scaffold. The cyanation step, converting the hemiaminal intermediate to the critical nitrile species, is driven by strong Lewis acids such as boron trifluoride acetic acid or tin tetrachloride. These catalysts activate the hydroxyl group for displacement by trimethylsilyl cyanide, ensuring the formation of the desired stereochemistry at the anomeric center while suppressing epimerization. This mechanistic pathway avoids the harsh conditions often required for nitrile introduction, thereby preserving the integrity of the sensitive furanose ring and the heterocyclic base. The absence of competing nucleophiles, thanks to the initial choice of the chloro-triazine precursor, ensures that the cyanide ion attacks exclusively at the intended position, leading to a cleaner impurity profile.
Furthermore, the substitution of the chloro group with an amino group via copper-catalyzed ammonolysis represents a significant improvement over traditional nucleophilic aromatic substitution methods. By employing copper salts such as cuprous iodide or cuprous bromide in polar solvents like dimethyl sulfoxide or alcohols, the reaction activates the C-Cl bond for displacement by ammonia under relatively mild thermal conditions. This catalytic cycle prevents the degradation of the nitrile group, which is susceptible to hydrolysis under strongly basic or acidic conditions found in non-catalyzed variants. The synergy between the Lewis acid cyanation and the copper-mediated amination creates a robust chemical environment where high conversion rates are achieved without the need for excessive reagent equivalents, directly translating to reduced raw material consumption and lower production costs.
How to Synthesize Remdesivir Efficiently
The synthesis of Remdesivir via this patented route involves a logical sequence of five distinct chemical transformations, each optimized for high yield and operational simplicity. The process begins with the cryogenic addition of the heterocyclic base to the protected lactone, followed by Lewis acid-mediated cyanation to establish the quaternary center. Subsequent steps involve the installation of the primary amine via ammonolysis, removal of benzyl protecting groups through hydrogenation, and final coupling with the phosphoramidate moiety. Detailed standard operating procedures for each stage, including specific temperature ramps, quenching protocols, and crystallization parameters, are essential for reproducing the high purity (>99.5%) reported in the patent examples.
- Perform addition condensation between 4-chloropyrrolo[2,1-f][1,2,4]triazine and 2,3,5-tribenzyloxy-D-ribono-1,4-lactone using LDA at -75°C.
- Execute Lewis acid-catalyzed cyanation using trimethylsilyl cyanide and boron trifluoride acetic acid to form the nitrile intermediate.
- Conduct copper-catalyzed ammonolysis to substitute the chloro group with an amino group, followed by palladium-catalyzed hydrogenation for debenzylation.
- Complete the synthesis by coupling the nucleoside with the phosphoramidate chloride using tert-butyl magnesium chloride and titanium tetrachloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical elegance. The elimination of hazardous reagents like boron trichloride and the reduction of protection-deprotection cycles fundamentally alter the cost structure of manufacturing. By simplifying the workflow, manufacturers can reduce the number of unit operations, which decreases labor costs, energy consumption, and equipment occupancy time. Additionally, the reliance on crystallization for purification rather than column chromatography ensures that the process is readily transferable to multi-ton reactors without the bottlenecks associated with preparative HPLC or flash chromatography.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route directly correlates to significant cost savings by removing entire classes of expensive reagents and protecting groups. The avoidance of TMS protection and the subsequent deprotection steps eliminates the purchase of silylating agents and the solvents required for their removal. Furthermore, the ability to recover and reuse the palladium catalyst from the hydrogenation step provides a recurring economic benefit, lowering the effective cost per kilogram of the final API. The high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, reducing the loss of valuable intermediates during purification and maximizing overall material throughput.
- Enhanced Supply Chain Reliability: Utilizing widely available and stable starting materials such as 4-chloropyrrolo[2,1-f][1,2,4]triazine enhances supply chain resilience. Unlike specialized organometallic reagents that may have long lead times or single-source dependencies, the reagents specified in this protocol, including common Lewis acids and copper salts, are commodity chemicals available from multiple global suppliers. The robustness of the reaction conditions, which tolerate a range of solvents and temperatures, ensures that production schedules are less likely to be disrupted by minor variations in raw material quality or environmental conditions, guaranteeing consistent delivery timelines for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior to prior art. Replacing corrosive boron trichloride with catalytic hydrogenation significantly reduces the generation of acidic waste and the risk of equipment corrosion, lowering maintenance costs and extending the lifespan of reactor vessels. The process generates fewer organic waste streams due to higher atom economy and fewer synthetic steps, simplifying wastewater treatment and reducing the carbon footprint of the manufacturing facility. These factors make the process highly scalable, allowing for seamless transition from pilot plant batches to full commercial production volumes while maintaining strict adherence to increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Remdesivir synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on yield expectations, safety profiles, and scalability potential for prospective manufacturing partners.
Q: How does this synthesis route improve upon conventional Remdesivir manufacturing methods?
A: This route eliminates the need for TMS protection of the amino group and avoids the use of hazardous boron trichloride for debenzylation, significantly reducing waste and simplifying purification.
Q: What catalysts are utilized in the key transformation steps?
A: The process employs Lewis acids such as boron trifluoride acetic acid for cyanation, copper salts like cuprous iodide for ammonolysis, and palladium on carbon for efficient hydrogenation.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method utilizes robust crystallization steps and avoids chromatographic purification, making it highly amenable to multi-kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent laboratory data to commercial reality requires deep technical expertise and state-of-the-art infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical efficiencies of this novel route are fully realized in practice. Our facilities are equipped with advanced cryogenic reactors for the initial addition step and high-pressure hydrogenation units for the debenzylation phase, all supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international pharmacopoeia standards.
We invite pharmaceutical innovators and generic manufacturers to collaborate with us to leverage this cutting-edge synthesis technology for their antiviral portfolios. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can accelerate your time-to-market and enhance your competitive position in the global antiviral landscape.
