Revolutionizing Tetrahydro-Beta-Carbolinone Synthesis: Scalable Cobalt-Catalyzed Process for Pharmaceutical Intermediates
The patent CN115260188A introduces a groundbreaking cobalt-catalyzed methodology for synthesizing tetrahydro-beta-carbolinone (THBC) compounds, addressing critical limitations in traditional pharmaceutical intermediate production. This innovation represents a significant advancement over existing palladium-dependent processes by utilizing cost-effective cobalt catalysts and commercially accessible reagents under optimized reaction conditions. The methodology enables efficient construction of the THBC scaffold—a core structural element found in numerous bioactive molecules—including antiviral natural products like bauerine C and anxiolytic drug candidates such as SL651498. By eliminating reliance on precious metal catalysts while maintaining high substrate compatibility, this approach delivers a more sustainable and economically viable pathway for manufacturing complex heterocyclic intermediates essential to modern drug development pipelines. The patent demonstrates robust reaction efficiency across diverse functional groups, establishing a foundation for scalable production of high-purity pharmaceutical building blocks that meet stringent regulatory requirements for therapeutic applications.
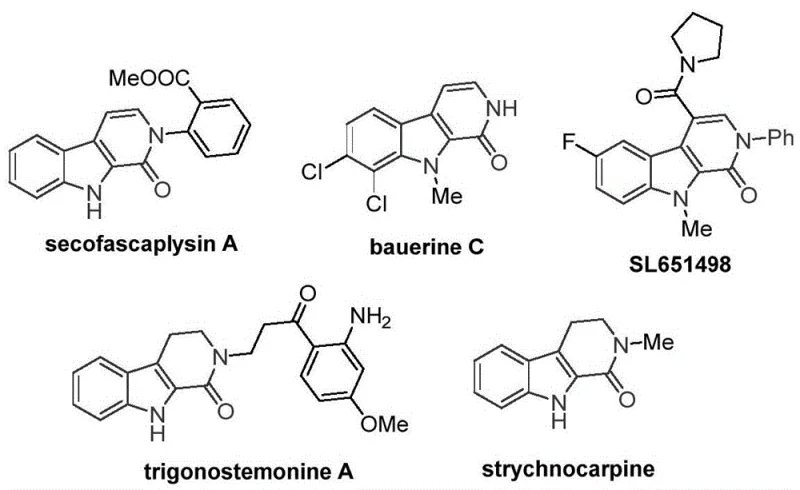
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for tetrahydro-beta-carbolinone compounds predominantly rely on palladium-catalyzed systems that present multiple operational and economic challenges. These methods require expensive palladium precursors with strict handling protocols due to their sensitivity to air and moisture, significantly increasing raw material costs and complicating manufacturing logistics. The necessity for rigorous metal removal steps to achieve pharmaceutical-grade purity generates substantial waste streams and extends processing timelines, creating bottlenecks in supply chain continuity. Furthermore, palladium-based processes often exhibit narrow substrate scope with poor tolerance for halogenated or electron-deficient functional groups, limiting their applicability for diverse intermediate production. The high temperatures and prolonged reaction times typically required in conventional approaches also contribute to increased energy consumption and reduced overall process efficiency, making large-scale implementation economically unattractive despite the structural importance of these compounds in drug discovery.
The Novel Approach
The patented cobalt-catalyzed methodology overcomes these limitations through a strategically designed carbonylation process that operates under significantly milder conditions. By utilizing cobalt acetate tetrahydrate as the catalyst precursor with silver carbonate as the oxidant, the system achieves efficient C-H activation at 130°C without requiring inert atmosphere maintenance typically associated with palladium chemistry. The substitution of toxic carbon monoxide with stable 1,3,5-tricarboxylic acid phenol ester enhances operational safety while maintaining reaction efficiency across a broad range of tryptamine derivatives. Crucially, the process demonstrates exceptional functional group tolerance—accepting halogenated, alkylated, and alkoxy-substituted substrates—enabling production of diverse THBC intermediates from a single platform. The simplified workup procedure involving standard filtration and column chromatography eliminates complex metal removal steps, directly translating to reduced processing time and improved environmental metrics through minimized solvent usage and waste generation.
Mechanistic Insights into Cobalt-Catalyzed Carbonylation
The reaction proceeds through a well-defined catalytic cycle initiated by oxidation of cobalt(II) to cobalt(III) by silver carbonate, followed by coordination with the tryptamine derivative to form a key intermediate complex. Subsequent C-H bond activation at the indole 2-position generates a cyclometalated species that undergoes migratory insertion with carbon monoxide released from the phenol ester substitute. This step forms an acylcobalt(III) intermediate that subsequently experiences reductive elimination to yield the cyclized product after hydrolysis. The mechanism benefits from the synergistic effect of pivalic acid as an additive that facilitates proton transfer while triethylamine maintains optimal basicity for the transformation. This carefully balanced catalytic system operates with precise stoichiometric control—using a molar ratio of tryptamine derivative:triethylamine:pivalic acid:cobalt catalyst:CO substitute:oxidant at 1:3.5:2:0.3:3:1.5—to maximize conversion efficiency while minimizing side reactions.
Impurity control is achieved through multiple mechanistic features inherent to this cobalt system. The selective C-H activation pathway prevents common side products associated with alternative cyclization methods, while the controlled release of carbon monoxide from the phenol ester substitute avoids over-carbonylation issues. The reaction's insensitivity to moisture compared to palladium systems reduces hydrolysis byproducts, and the mild temperature profile (130°C) prevents thermal degradation of sensitive functional groups. Post-reaction purification leverages standard column chromatography without requiring specialized metal scavenging techniques, ensuring consistent production of high-purity intermediates meeting pharmaceutical specifications. This integrated approach to impurity management directly supports regulatory compliance by minimizing genotoxic impurities and residual metal content in the final product.

How to Synthesize Tetrahydro-Beta-Carbolinone Efficiently
This cobalt-catalyzed carbonylation process represents a significant advancement in the synthesis of tetrahydro-beta-carbolinone intermediates by replacing traditional palladium systems with a more economical and operationally simpler methodology. The patent demonstrates exceptional versatility across diverse substrate classes while maintaining high efficiency under standardized conditions that facilitate seamless technology transfer to manufacturing environments. The following section provides a detailed step-by-step guide for implementing this innovative synthesis route in laboratory or pilot-scale settings, ensuring consistent production of high-purity intermediates suitable for pharmaceutical applications.
- Combine tryptamine derivative with cobalt acetate tetrahydrate, pivalic acid, and triethylamine in dioxane under inert atmosphere
- Add silver carbonate and TFBen as oxidant and carbon monoxide substitute, then heat to 130°C for 20 hours
- Perform post-treatment via filtration, silica gel mixing, and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by delivering a more resilient and cost-effective manufacturing solution. The elimination of precious metal catalysts fundamentally transforms the economic profile of THBC production while simultaneously enhancing supply chain robustness through reliance on globally available raw materials. These advantages position the process as an ideal solution for organizations seeking reliable access to high-purity intermediates without exposure to volatile catalyst markets or complex purification requirements that typically disrupt production timelines.
- Cost Reduction in Manufacturing: The substitution of palladium with cobalt catalysts eliminates expensive metal removal protocols required for regulatory compliance, substantially reducing processing costs through simplified purification workflows. The use of commercially abundant reagents like triethylamine and pivalic acid further optimizes raw material expenses while maintaining high reaction efficiency across diverse substrates. This strategic reagent selection creates significant cost savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through reliance on widely available starting materials including standard tryptamine derivatives and common solvents like dioxane. The elimination of specialized catalyst handling requirements removes potential supply chain bottlenecks associated with precious metal logistics, ensuring consistent production capacity even during market fluctuations. This robust material sourcing profile supports reliable delivery schedules critical for pharmaceutical manufacturing operations.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production with minimal equipment modification requirements due to its moderate operating conditions. The simplified waste stream profile resulting from eliminated metal removal steps significantly reduces environmental impact while lowering disposal costs. These features collectively support seamless transition from development to full-scale manufacturing while meeting increasingly stringent sustainability requirements in pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this patented synthesis methodology. Each response is derived directly from the experimental data and technical disclosures within patent CN115260188A, providing evidence-based insights for procurement and R&D decision-makers evaluating this innovative approach.
Q: How does the cobalt-catalyzed method improve upon traditional palladium-based synthesis?
A: The cobalt system eliminates expensive palladium catalysts while maintaining high functional group tolerance. This avoids costly metal removal steps required in palladium processes, significantly reducing purification complexity and associated waste streams without compromising yield or purity.
Q: What are the scalability advantages of this process for commercial production?
A: The reaction operates under mild conditions (130°C) with readily available reagents and standard equipment. The documented scalability from milligram to gram scale in patent examples demonstrates straightforward transition to industrial reactors, with no specialized infrastructure required for commercial implementation.
Q: How does the elimination of transition metal purification steps reduce costs?
A: By replacing palladium with cobalt, the process removes the need for extensive heavy metal clearance protocols. This eliminates multiple purification stages and associated solvent consumption, leading to substantial cost savings through reduced processing time and simplified quality control procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-Beta-Carbolinone Supplier
Our patented cobalt-catalyzed methodology represents a transformative approach to manufacturing tetrahydro-beta-carbolinone intermediates with significant implications for pharmaceutical development pipelines. As a specialized CDMO partner, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our integrated manufacturing platform ensures seamless technology transfer from laboratory validation to full-scale production, delivering consistent high-quality intermediates that meet global regulatory standards for pharmaceutical applications.
Leverage our technical expertise through a Customized Cost-Saving Analysis tailored to your specific production requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments that demonstrate how this innovative process can optimize your supply chain while ensuring reliable access to high-purity tetrahydro-beta-carbolinone intermediates.
