Advanced Cobalt-Catalyzed Synthesis of Tetrahydro-Beta-Carbolinone Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for privileged scaffolds that underpin modern drug discovery. Patent CN115260188A introduces a transformative methodology for constructing the tetrahydro-beta-carbolinone core, a structural motif prevalent in numerous bioactive natural products and therapeutic candidates. This nitrogen-containing heterocyclic framework is critical for molecules exhibiting antiviral properties, such as bauerine C, and anxiolytic potential, exemplified by the candidate drug SL651498. The significance of this scaffold cannot be overstated, as it serves as a foundational building block for complex alkaloids like secofascaplysin A and strychnocarpine. By leveraging a novel transition metal catalysis strategy, this invention addresses long-standing challenges in efficiency and scalability, offering a viable pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines.
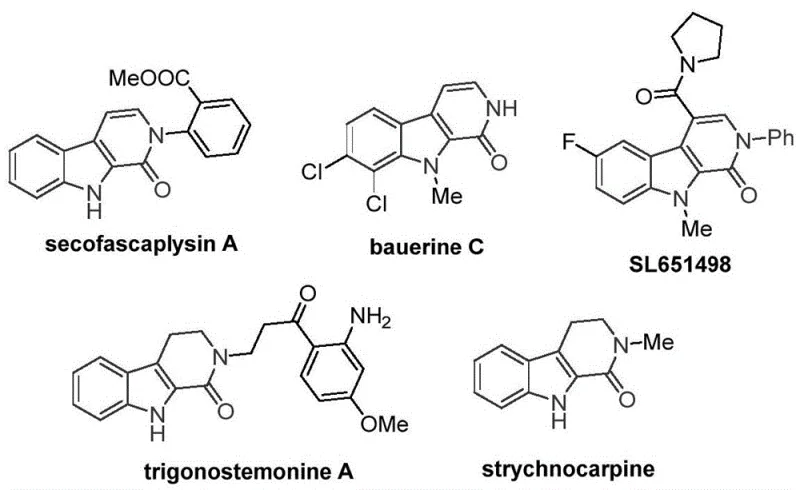
Historically, the construction of the tetrahydro-beta-carbolinone skeleton via carbonylation reactions has been heavily reliant on palladium catalysis. While effective, palladium-based systems present inherent limitations that hinder widespread industrial adoption, primarily due to the high cost of the precious metal and the stringent regulatory requirements for residual metal levels in final active pharmaceutical ingredients. Furthermore, traditional methods often suffer from narrow substrate scope and require harsh conditions that can compromise sensitive functional groups. In contrast, the novel approach detailed in the patent utilizes a cobalt-catalyzed C-H activation strategy. This shift from precious metals to base metals represents a paradigm shift in cost reduction in API manufacturing. The new method employs cobalt acetate tetrahydrate as a catalyst, which is not only inexpensive but also exhibits remarkable catalytic activity when paired with silver carbonate as an oxidant and 1,3,5-tricarboxylic acid phenol ester as a safe carbon monoxide source.

Mechanistic Insights into Cobalt-Catalyzed C-H Activation Carbonylation
The mechanistic pathway of this transformation offers deep insights into its efficiency and selectivity, which are paramount for R&D directors focused on impurity control. The cycle initiates with the oxidation of the cobalt(II) precursor by silver carbonate, generating a high-valent cobalt(III) species that coordinates with the tryptamine derivative substrate. This coordination is crucial for directing the subsequent C-H bond activation at the 2-position of the indole ring, forming a stable cobalt(III) metallacycle. The precision of this activation step ensures that the reaction proceeds with high regioselectivity, minimizing the formation of unwanted isomers that would complicate downstream purification. Following C-H activation, the carbon monoxide moiety, released in situ from the thermal decomposition of the 1,3,5-tricarboxylic acid phenol ester, inserts into the cobalt-carbon bond. This insertion generates an acylcobalt(III) intermediate, which is the key precursor to the final lactam ring structure.
The final stage of the catalytic cycle involves reductive elimination and hydrolysis, which releases the desired tetrahydro-beta-carbolinone product and regenerates the active cobalt species. This mechanism highlights the dual role of the additives; pivalic acid acts as a proton shuttle to facilitate C-H cleavage, while triethylamine serves as a base to neutralize acidic byproducts. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and purity. For instance, maintaining the reaction temperature between 120°C and 140°C is critical to ensure sufficient energy for CO release and insertion without degrading the sensitive indole nucleus. The robustness of this catalytic system against various electronic environments on the aromatic ring further underscores its utility for synthesizing diverse libraries of high-purity tetrahydro-beta-carbolinone derivatives.
How to Synthesize Tetrahydro-Beta-Carbolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized conditions outlined in the patent to achieve the reported high yields. The procedure is designed to be operationally simple, avoiding the need for specialized high-pressure equipment typically associated with carbonylation reactions. By utilizing a solid CO surrogate, the process eliminates the safety hazards of handling toxic carbon monoxide gas, making it accessible to standard organic synthesis facilities. The following guide summarizes the critical steps for executing this transformation effectively, ensuring reproducibility and safety.
- Combine cobalt acetate tetrahydrate, silver carbonate, 1,3,5-tricarboxylic acid phenol ester, pivalic acid, triethylamine, and the tryptamine derivative substrate in dioxane solvent.
- Heat the reaction mixture to a temperature range of 120-140°C and maintain stirring for 16 to 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target tetrahydro-beta-carbolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this cobalt-catalyzed protocol offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs associated with the catalyst system. Replacing palladium with cobalt acetate tetrahydrate removes the volatility linked to precious metal markets, stabilizing the cost of goods sold over long-term production contracts. Additionally, the use of commercially available reagents such as silver carbonate and triethylamine ensures a secure supply chain, as these chemicals are produced at scale by multiple global vendors, reducing the risk of single-source bottlenecks. The simplicity of the workup procedure, involving filtration and standard column chromatography, further contributes to cost reduction in pharmaceutical intermediate manufacturing by minimizing labor hours and solvent consumption during purification.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with earth-abundant cobalt significantly lowers the direct material costs per kilogram of product. This economic advantage is compounded by the high atom economy of the reaction, where the CO surrogate efficiently transfers the carbonyl group without generating excessive waste. Furthermore, the mild reaction conditions reduce energy consumption compared to high-pressure alternatives, leading to lower utility costs in large-scale reactors. The elimination of complex ligand synthesis, often required for palladium systems, streamlines the procurement process and reduces inventory complexity.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than specialized organometallic complexes enhances supply chain resilience. Tryptamine derivatives, the key starting materials, are readily accessible from established chemical suppliers, ensuring consistent feedstock availability. The robustness of the reaction across a wide range of substrates means that a single standardized protocol can be applied to produce various analogues, simplifying production scheduling and inventory management. This flexibility allows manufacturers to respond rapidly to changing market demands for different tetrahydro-beta-carbolinone variants without requalifying entirely new processes.
- Scalability and Environmental Compliance: The protocol has been demonstrated to be scalable, with the patent noting successful expansion to gram-scale synthesis without loss of efficiency. This scalability is essential for transitioning from clinical trial material to commercial production volumes. From an environmental perspective, the use of a solid CO source mitigates the risks associated with greenhouse gas emissions and toxic gas leaks. The waste stream is primarily composed of benign organic byproducts and silver salts, which can be managed through standard waste treatment protocols, aligning with increasingly stringent environmental regulations in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their manufacturing portfolios. Understanding these details is crucial for assessing the feasibility of adopting this route for specific project requirements.
Q: What are the advantages of using cobalt over palladium for this synthesis?
A: Cobalt catalysts are significantly more earth-abundant and cost-effective compared to precious metal palladium catalysts. This switch reduces raw material costs and simplifies heavy metal removal processes in pharmaceutical manufacturing.
Q: What is the source of the carbonyl group in this reaction?
A: The reaction utilizes 1,3,5-tricarboxylic acid phenol ester as a solid carbon monoxide substitute. This avoids the need for handling hazardous gaseous CO directly, enhancing operational safety.
Q: Is this method compatible with diverse functional groups?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating various substituents such as halogens (Cl, Br), alkyl groups (Me), and alkoxy groups (OMe) on the indole ring without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-Beta-Carbolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries translate seamlessly into industrial reality. We are committed to delivering high-purity tetrahydro-beta-carbolinone intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the cobalt-catalyzed C-H activation technology described in CN115260188A positions us as a strategic partner for clients seeking cost-effective and sustainable solutions for complex heterocyclic synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this cobalt-based protocol. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to optimize your supply chain and bring life-saving medications to market faster and more efficiently.
