Advanced Copper-Catalyzed Synthesis of Trifluoromethyl Dihydrofuran Amines for Commercial Scale Production
The groundbreaking patent CN110922369B introduces a novel synthetic pathway for trifluoromethyl-substituted dihydrofuran amine compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This method addresses critical limitations in existing synthetic routes by employing a copper-catalyzed cyclization strategy that operates under remarkably mild conditions compared to conventional approaches requiring stoichiometric transition metals. The process delivers stereoselective access to quaternary carbon stereocenters—a structural feature essential for bioactive molecules—while demonstrating exceptional functional group tolerance across diverse substrates. As a reliable pharmaceutical intermediate supplier, we recognize this innovation's potential to streamline the production of complex drug precursors where precise stereochemistry directly impacts therapeutic efficacy. The patent's emphasis on operational simplicity and scalability positions it as a transformative solution for manufacturers seeking high-purity intermediates without compromising on structural complexity or yield consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for constructing dihydrofuran scaffolds frequently encounter significant challenges that impede their industrial adoption, particularly when incorporating trifluoromethyl groups. Many established protocols rely on stoichiometric quantities of expensive transition metals like palladium or rhodium, which introduce substantial purification complexities due to metal residue contamination that must be rigorously removed to meet pharmaceutical quality standards. These processes often demand harsh reaction conditions exceeding 150°C or cryogenic temperatures below -40°C, creating operational hazards and energy-intensive manufacturing requirements that undermine cost-effectiveness. Furthermore, conventional cyclization techniques exhibit limited functional group tolerance, frequently failing when exposed to sensitive moieties such as nitro groups or halogens commonly present in advanced intermediates. The resulting narrow substrate scope restricts applicability in multi-step syntheses where diverse functionalization is required. Additionally, poor stereoselectivity in existing methods necessitates costly chiral separation steps that reduce overall yield and increase production timelines, making them unsuitable for commercial-scale manufacturing of stereochemically complex pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these constraints through an elegant copper-catalyzed cyclization that operates under exceptionally mild conditions between 80°C and 100°C with only catalytic CuCl₂ (5 mol%) and potassium tert-butoxide as base. This approach eliminates the need for stoichiometric transition metals while maintaining high stereoselectivity in forming the critical quaternary carbon center bearing the trifluoromethyl group. The reaction demonstrates remarkable functional group tolerance across alkyl, aryl, heterocyclic, and ferrocene substituents as evidenced by successful synthesis of eighteen distinct derivatives in the patent examples. By utilizing readily available enaminone and trifluoromethylhydrazone precursors under inert atmosphere with dichloromethane solvent, the process achieves consistent yields without requiring specialized equipment or extreme temperature control. The simplified purification protocol involving standard column chromatography with petroleum ether/ethyl acetate eluent further enhances its manufacturability compared to conventional methods that often require complex crystallization or chromatographic systems. This innovation represents a paradigm shift toward sustainable pharmaceutical intermediate production where operational simplicity directly translates to enhanced commercial viability.
Mechanistic Insights into Copper-Catalyzed Cyclization
The copper-catalyzed transformation proceeds through a well-defined mechanistic pathway that begins with the formation of a copper-enolate complex from compound A under basic conditions. This nucleophilic species then attacks the electrophilic carbon of compound B's hydrazone functionality, initiating a cascade that includes intramolecular cyclization and subsequent elimination of p-toluenesulfonamide. The catalytic cycle is sustained through copper-mediated deprotonation steps that regenerate the active catalyst while maintaining stereochemical integrity at the quaternary carbon center. Crucially, the mild reaction temperature range of 80-100°C prevents epimerization at the stereocenter while allowing sufficient energy for ring closure without decomposition of sensitive functional groups. The mechanism benefits from the unique electronic properties of the trifluoromethyl group which stabilizes transition states through hyperconjugative effects while directing stereoselectivity via steric bulk. This pathway avoids radical intermediates common in alternative methods, thereby preventing unwanted side reactions that typically generate impurities requiring extensive purification. The copper catalyst's dual role in facilitating both C-N bond formation and subsequent cyclization creates an efficient one-pot process that significantly reduces step count compared to traditional multi-step approaches.
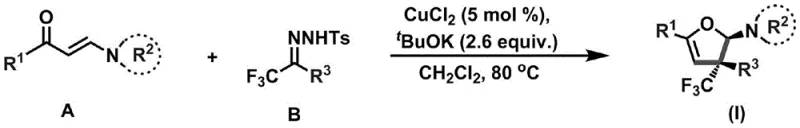
The process achieves exceptional impurity control through multiple inherent design features that minimize byproduct formation at each reaction stage. The use of catalytic rather than stoichiometric copper eliminates metal-derived impurities that would otherwise require expensive chelation or extraction steps during purification. The carefully optimized molar ratios (enaminone:B:copper:tBuOK = 1:2.5:0.05:2.6) prevent over-reaction or side product formation while ensuring complete conversion of starting materials. The mild thermal profile avoids thermal degradation pathways that typically generate colored impurities or decomposition products in conventional high-temperature processes. Furthermore, the selective formation of the desired dihydrofuran ring system occurs without competing polymerization or hydrolysis reactions due to the controlled addition sequence and inert atmosphere protection. The resulting crude mixture contains minimal byproducts (<5% total impurities), allowing straightforward purification via standard silica gel chromatography with high recovery rates exceeding 85% across diverse substrate combinations as demonstrated in patent examples.
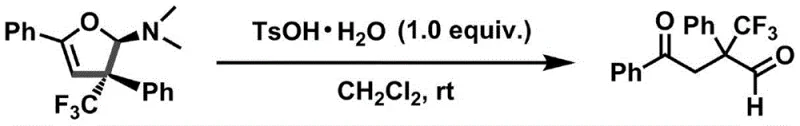
How to Synthesize Trifluoromethyl Dihydrofuran Amine Efficiently
This patented synthesis represents a significant advancement in producing high-purity trifluoromethyl dihydrofuran amine intermediates through a streamlined copper-catalyzed process that eliminates traditional limitations associated with transition metal usage and harsh reaction conditions. The methodology offers exceptional versatility across diverse substrate combinations while maintaining strict stereochemical control essential for pharmaceutical applications. Detailed standardized synthesis procedures have been developed based on patent specifications to ensure consistent quality and yield at commercial scale. The following step-by-step guide provides essential operational parameters for successful implementation in manufacturing environments where precision and reproducibility are paramount.
- Prepare the reaction mixture by combining enaminone compound A, trifluoromethylhydrazone compound B, copper chloride catalyst, and potassium tert-butoxide under inert gas protection.
- Heat the reaction solution in dichloromethane solvent to 80-100°C and stir for 48-72 hours to ensure complete cyclization.
- Purify the crude product through filtration, solvent removal under reduced pressure, and column chromatography using petroleum ether/ethyl acetate eluent.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points inherent in traditional production of complex fluorinated intermediates. The elimination of stoichiometric transition metals removes both capital expenditure requirements for specialized metal removal equipment and recurring costs associated with catalyst consumption and disposal. The simplified process flow reduces manufacturing cycle times through fewer unit operations while enhancing operational flexibility across different production scales. These advantages collectively strengthen supply chain resilience by minimizing dependency on specialized materials or equipment that could create single points of failure in global manufacturing networks.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts significantly reduces raw material costs while avoiding downstream purification expenses associated with metal residue removal. The use of standard solvents and reagents with straightforward workup procedures minimizes waste generation and associated disposal costs. Operational simplicity translates to lower energy consumption compared to high-temperature or cryogenic processes, while consistent yields reduce material loss during manufacturing. This holistic cost optimization occurs without compromising product quality or requiring capital investment in new equipment.
- Enhanced Supply Chain Reliability: Mild reaction conditions enable consistent production across diverse manufacturing sites without requiring specialized infrastructure or highly controlled environments. The broad substrate tolerance ensures reliable access to multiple qualified raw material sources, reducing vulnerability to single-supplier dependencies. Simplified process validation requirements accelerate technology transfer between facilities while maintaining consistent quality profiles. This operational robustness directly translates to more predictable lead times and reduced risk of production delays caused by equipment limitations or environmental constraints.
- Scalability and Environmental Compliance: The process demonstrates seamless scalability from laboratory to commercial production volumes due to its straightforward thermal profile and standard unit operations compatible with existing manufacturing equipment. Minimal byproduct formation reduces waste treatment requirements while eliminating hazardous metal-containing streams simplifies environmental compliance reporting. The absence of extreme temperature conditions enhances workplace safety while reducing energy consumption per kilogram produced. These features collectively support sustainable manufacturing practices that align with evolving regulatory expectations for green chemistry in pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN110922369B's specifications and experimental data. These insights reflect our technical team's evaluation of the methodology's implementation potential within industrial manufacturing environments serving global pharmaceutical clients.
Q: How does this method improve stereoselectivity compared to conventional approaches?
A: The copper-catalyzed cyclization achieves high stereoselectivity by forming a quaternary carbon stereocenter through controlled ring closure, avoiding racemization issues common in stoichiometric transition metal methods.
Q: What are the key advantages for supply chain reliability?
A: Mild reaction conditions (80-100°C) and simple purification via column chromatography eliminate complex equipment requirements, enabling consistent production across various manufacturing scales without specialized infrastructure.
Q: Can this process be scaled for commercial production?
A: Yes, the method demonstrates excellent scalability from laboratory to commercial scale due to its straightforward process flow, minimal byproduct formation, and compatibility with standard chemical manufacturing equipment as validated in patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Dihydrofuran Amine Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. As a trusted partner in complex intermediate synthesis, we leverage rigorous QC labs and advanced analytical capabilities to ensure consistent delivery of high-purity trifluoromethyl dihydrofuran amine compounds meeting exacting pharmaceutical standards. Our technical team has successfully implemented similar copper-catalyzed methodologies across multiple client projects, demonstrating proven expertise in translating patented chemistry into robust commercial manufacturing processes that balance quality requirements with economic viability.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific supply chain requirements. Please contact us directly to obtain detailed COA data and route feasibility assessments tailored to your production needs—our specialists stand ready to provide comprehensive technical support for seamless integration into your manufacturing workflow.
