Advanced Catalytic Synthesis of Trifluoromethyl Dihydrofuran Amines for Commercial Pharmaceutical Applications
Advanced Catalytic Synthesis of Trifluoromethyl Dihydrofuran Amines for Commercial Pharmaceutical Applications
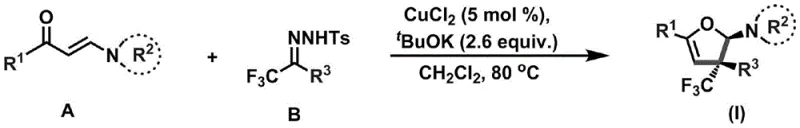
The integration of trifluoromethyl groups into heterocyclic scaffolds represents a critical frontier in modern medicinal chemistry, driven by the unique metabolic stability and lipophilicity these motifs impart to drug candidates. Patent CN110922369B discloses a groundbreaking methodology for the stereoselective construction of trifluoromethyl-substituted dihydrofuran amines, addressing long-standing challenges in accessing these complex molecular architectures. This technology leverages a copper-catalyzed cyclization strategy that operates under remarkably mild conditions, utilizing simple acyclic precursors to generate high-value intermediates with quaternary carbon stereocenters. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent offers a robust pathway to diversify chemical libraries while optimizing process efficiency. The ability to construct these privileged structures from readily available enaminones and trifluoromethylhydrazones marks a significant departure from traditional, resource-intensive synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydrofuran derivatives has relied heavily on classical cycloaddition strategies such as [4+1] or [3+2] annulations, which often suffer from severe limitations in industrial applicability. Many established protocols necessitate the use of stoichiometric amounts of expensive transition metals, leading to substantial cost burdens and complex downstream purification requirements to meet stringent residual metal specifications. Furthermore, conventional methods frequently demand harsh reaction conditions, including extreme temperatures or strong acidic/basic environments, which severely limit functional group tolerance and result in poor chemoselectivity. The introduction of trifluoromethyl groups into these heterocycles has been particularly challenging, as the unique electronic properties of fluorine often destabilize reactive intermediates or inhibit cyclization kinetics. Consequently, existing literature methods often exhibit narrow substrate scope, failing to accommodate diverse aryl or alkyl substituents required for modern drug discovery programs.
The Novel Approach
The methodology outlined in CN110922369B revolutionizes this landscape by employing a catalytic system that effectively couples enaminones with trifluoromethylhydrazones to forge the dihydrofuran ring in a single pot. This novel approach eliminates the need for stoichiometric metal reagents, utilizing merely 5 mol% of copper chloride alongside potassium tert-butoxide as a base to drive the transformation. The reaction proceeds smoothly in dichloromethane at moderate temperatures ranging from 80°C to 100°C, demonstrating exceptional compatibility with a wide array of functional groups including nitro, cyano, and halogen substituents. By shifting from stoichiometric to catalytic regimes, this process not only reduces raw material costs but also simplifies the impurity profile, facilitating easier purification via standard column chromatography. This strategic shift enables the efficient production of stereoselective derivatives that were previously difficult or impossible to access through traditional means.
Mechanistic Insights into CuCl2-Catalyzed Cyclization
The core of this technological breakthrough lies in the intricate interplay between the copper catalyst and the nitrogen-containing precursors, which facilitates a cascade of bond-forming events leading to the dihydrofuran core. Mechanistically, the reaction likely initiates with the activation of the trifluoromethylhydrazone by the base, generating a diazo-like species or a nucleophilic nitrogen center that attacks the electron-deficient beta-carbon of the enaminone. The copper species plays a pivotal role in stabilizing transient intermediates and promoting the subsequent intramolecular cyclization that closes the five-membered oxygen-containing ring. This catalytic cycle ensures high turnover numbers and maintains stereocontrol, resulting in the formation of the quaternary carbon stereocenter with high fidelity. The tolerance of the catalytic system towards various R1 and R3 substituents, ranging from simple phenyl groups to complex heterocycles and ferrocene moieties, underscores the versatility of this mechanistic pathway.
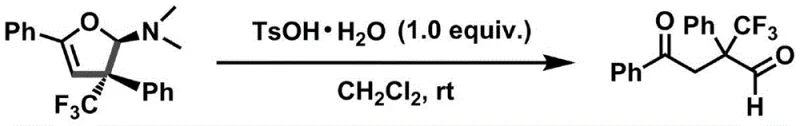
From an impurity control perspective, the mildness of the reaction conditions is paramount in preventing the decomposition of the sensitive trifluoromethyl group and avoiding side reactions such as polymerization or over-oxidation. The use of potassium tert-butoxide provides a sufficiently strong basic environment to drive the reaction forward without inducing elimination pathways that could degrade the enaminone substrate. Furthermore, the choice of dichloromethane as a solvent ensures good solubility for both organic substrates and the inorganic base, creating a homogeneous reaction medium that maximizes collision frequency and reaction rate. The final product, characterized by its distinct dihydrofuran amine structure, can be further derivatized; for instance, acid-mediated hydrolysis using p-toluenesulfonic acid cleanly converts the amine into a 1,4-dicarbonyl compound, expanding the utility of this scaffold in downstream synthesis.
How to Synthesize Trifluoromethyl Dihydrofuran Amine Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity, leveraging the robust protocol defined in the patent documentation. The process begins with the careful preparation of the reaction mixture under an inert atmosphere to prevent oxidation of sensitive intermediates, followed by a controlled heating phase to activate the catalytic cycle. Operators must adhere to specific molar ratios, typically maintaining a 1:2.5 ratio between the enaminone and the trifluoromethylhydrazone to ensure complete conversion of the limiting reagent. The detailed standardized synthesis steps below outline the exact procedural requirements for achieving reproducible results on both laboratory and pilot scales.
- Mix enaminone (Compound A), trifluoromethylhydrazone (Compound B), CuCl2 catalyst, and potassium tert-butoxide base under inert gas protection.
- Add dichloromethane solvent and heat the reaction mixture to 80-100°C, stirring for 48-72 hours to complete the cyclization.
- Filter insoluble solids, concentrate the solution under reduced pressure, and purify the crude product via column chromatography to obtain the target dihydrofuran amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic methodology translates directly into tangible operational efficiencies and risk mitigation strategies. By replacing stoichiometric metal reagents with a catalytic system, manufacturers can drastically reduce the consumption of expensive raw materials and minimize the generation of hazardous heavy metal waste, thereby lowering disposal costs and environmental compliance burdens. The mild reaction conditions allow for the use of standard stainless steel reactors without the need for specialized corrosion-resistant linings, facilitating easier technology transfer from R&D to commercial production facilities. Additionally, the broad substrate scope means that a single production line can be adapted to manufacture a diverse portfolio of analogues simply by swapping starting materials, enhancing supply chain flexibility and responsiveness to market demands.
- Cost Reduction in Manufacturing: The transition to a copper-catalyzed process significantly lowers the cost of goods sold by eliminating the need for precious metal catalysts often used in alternative cyclization methods. Since the catalyst loading is low (5 mol%) and the base is inexpensive potassium tert-butoxide, the overall reagent cost is optimized. Furthermore, the simplified workup procedure involving filtration and concentration reduces solvent usage and processing time, contributing to substantial cost savings in utility and labor expenses compared to multi-step traditional syntheses.
- Enhanced Supply Chain Reliability: The starting materials, specifically enaminones and trifluoromethylhydrazones, are commercially available or easily synthesized from commodity chemicals, ensuring a stable and continuous supply chain. This reliance on accessible feedstocks mitigates the risk of bottlenecks associated with exotic or proprietary reagents. The robustness of the reaction against variations in substrate electronics means that supply chain disruptions for specific substituted precursors can often be managed by switching to alternative analogues without re-optimizing the entire process.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of benign inorganic salts that are easily removed during the aqueous workup or filtration steps. The absence of stoichiometric heavy metals simplifies the regulatory approval process for pharmaceutical intermediates, as residual metal limits are easier to meet. This environmental friendliness aligns with green chemistry principles, making the process attractive for companies aiming to reduce their carbon footprint and adhere to increasingly strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl dihydrofuran synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent specification, providing clarity on process capabilities and application potential. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What are the key advantages of this Cu-catalyzed method over traditional dihydrofuran synthesis?
A: Unlike conventional methods that often require stoichiometric transition metals and harsh conditions, this patent-disclosed process utilizes a catalytic amount of Copper Chloride (5 mol%) under mild temperatures (80-100°C), significantly improving functional group tolerance and reducing heavy metal waste.
Q: Can this synthetic route be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process demonstrates excellent substrate applicability and uses readily available acyclic substrates (enaminones and hydrazones). The mild reaction conditions and simple workup procedure (filtration and chromatography) make it highly suitable for commercial scale-up.
Q: What is the downstream application potential of these trifluoromethyl dihydrofuran amines?
A: These compounds serve as valuable synthetic intermediates carrying a quaternary carbon stereocenter. They can be quantitatively converted into trifluoromethyl-containing 1,4-dicarbonyl compounds using p-toluenesulfonic acid, which are crucial precursors for constructing diverse heterocyclic drug molecules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Dihydrofuran Amine Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this patented laboratory methodology into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of this copper-catalyzed cyclization are perfectly managed at scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of trifluoromethyl dihydrofuran amine meets the highest quality standards required by global pharmaceutical clients.
We invite you to collaborate with us to leverage this innovative synthesis for your next drug development program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and optimize your supply chain economics.
