Advanced Copper-Catalyzed Synthesis of Beta-Carbonyl Sulfones for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex molecular architectures, particularly those containing sulfur functionalities which are prevalent in bioactive molecules. Patent CN111978216A introduces a significant advancement in this domain by disclosing a novel preparation method for beta-carbonyl sulfone compounds. These structural units are critical scaffolds found in numerous drug candidates exhibiting anticancer, antitumor, antibacterial, and anti-inflammatory activities. Furthermore, they serve as versatile intermediates for synthesizing optically active beta-hydroxy sulfones, chiral lactones, and various N-heterocyclic compounds. The disclosed technology addresses long-standing challenges in organic synthesis by utilizing 1,1-disubstituted olefins and sulfonyl hydrazides as primary raw materials, mediated by a copper salt catalyst in an organic solvent system. This approach not only simplifies the operational workflow but also significantly expands the substrate range compared to prior art, offering a compelling solution for the cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-carbonyl sulfones has been plagued by significant operational and chemical inefficiencies that hinder large-scale adoption. Traditional synthetic routes predominantly rely on the reaction of sulfinic acids or their salts with corresponding alpha-halogenated ketones. However, the availability of diverse sulfinic acid salts is relatively limited, creating a bottleneck in supply chain reliability for specialized derivatives. Alternative methods involve the direct oxidation of beta-carbonyl sulfides, beta-carbonyl sulfoxides, or beta-hydroxy sulfones, which often necessitate harsh reaction conditions and stoichiometric amounts of strong oxidants. More recent radical-mediated oxysulfonation reactions have shown promise but frequently suffer from narrow substrate scopes and poor functional group tolerance. For instance, prior art such as CN108383763B describes a method requiring peroxide oxidants and cumbersome multi-step catalyst addition protocols, alongside mandatory cation exchange membrane treatment before purification. These complexities increase production costs, extend lead times, and generate substantial chemical waste, making them less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN111978216A represents a paradigm shift towards green and efficient synthesis. By employing 1,1-disubstituted olefins and sulfonyl hydrazides, the process leverages readily available and stable starting materials that are easily sourced from global chemical suppliers. The reaction proceeds under remarkably mild conditions, typically at temperatures between 40°C and 80°C, with a preferred optimum around 65°C. Crucially, molecular oxygen serves a dual role as both the terminal oxidant and the oxygen source for the carbonyl group, eliminating the need for expensive or hazardous chemical oxidants. This innovation drastically simplifies the post-reaction workup, as there is no need for specialized membrane treatments or complex quenching procedures. The result is a streamlined process that delivers high-purity beta-carbonyl sulfones with yields reaching up to 93%, demonstrating superior efficiency and economic viability for industrial applications.
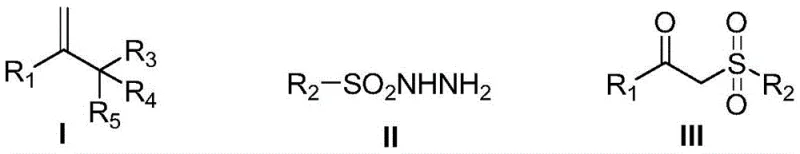
Mechanistic Insights into Copper-Catalyzed Oxysulfonation
The core of this technological breakthrough lies in the copper-catalyzed activation of the sulfonyl hydrazide and the subsequent radical addition to the olefinic double bond. The mechanism likely involves the generation of a sulfonyl radical species from the hydrazide precursor through single-electron transfer mediated by the copper catalyst in the presence of oxygen. This radical species then adds across the carbon-carbon double bond of the 1,1-disubstituted olefin, forming a new carbon-sulfur bond and a carbon-centered radical intermediate. Subsequent trapping of this intermediate by molecular oxygen, followed by further oxidation and rearrangement, leads to the formation of the carbonyl functionality adjacent to the sulfone group. The use of copper trifluoromethanesulfonate as the preferred catalyst ensures high turnover numbers and stability throughout the reaction cycle. This mechanistic pathway is exceptionally tolerant of diverse functional groups, allowing for the incorporation of halogens, esters, ethers, and heterocycles without side reactions or decomposition, which is a critical requirement for the synthesis of high-purity OLED material precursors and advanced pharmaceutical building blocks.
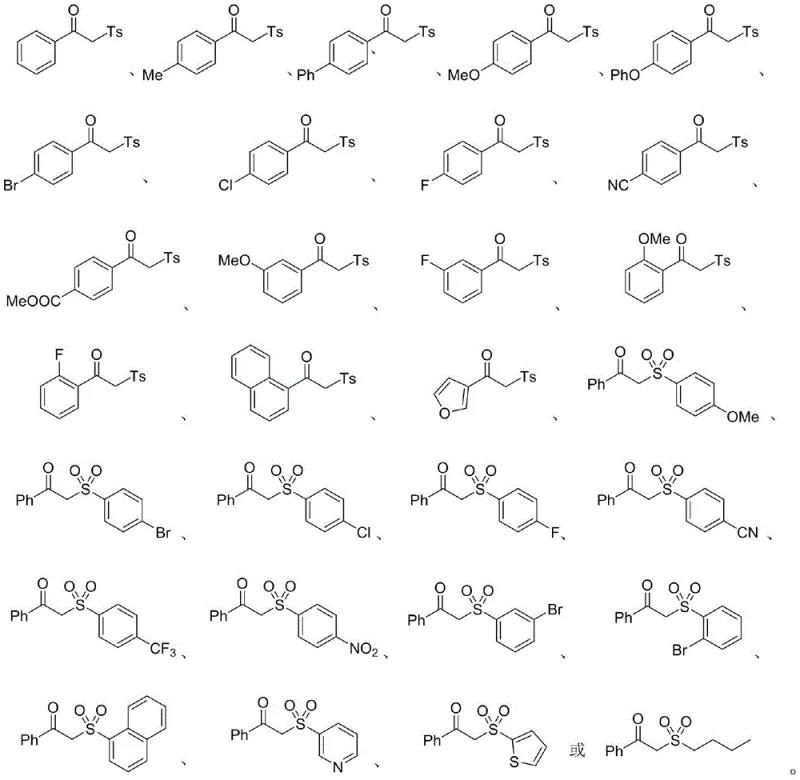
Impurity control is inherently managed by the selectivity of the radical process and the mild reaction environment. Unlike harsh acidic or basic conditions used in traditional methods that might promote hydrolysis or elimination side reactions, this neutral to slightly acidic catalytic system preserves sensitive moieties. The reaction produces minimal byproducts, primarily nitrogen gas and water, which are easily removed, thereby simplifying the purification process. The broad substrate scope is evidenced by the successful synthesis of derivatives containing electron-withdrawing groups like cyano and nitro, as well as electron-donating groups like methoxy and methyl, on both the olefin and the sulfonyl hydrazide components. This versatility ensures that the process can be adapted for the commercial scale-up of complex polymer additives and specialty chemicals with varying electronic properties, providing a reliable platform for diverse chemical manufacturing needs.
How to Synthesize Beta-Carbonyl Sulfone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and atmospheric conditions to maximize yield and purity. The standard protocol involves mixing the 1,1-disubstituted olefin and the sulfonyl hydrazide in a solvent system, preferably a mixture of acetonitrile and water in a 9:1 volume ratio, although other solvents like THF or DMF are also viable. The copper catalyst is added in catalytic amounts, typically ranging from 1 mol% to 20 mol%, with 5 mol% being optimal for most substrates. The reaction vessel must be charged with oxygen or simply exposed to air, depending on the specific reactivity of the substrates, and heated to the designated temperature for a period of 4 to 24 hours. Detailed standardized synthesis steps see the guide below.
- Combine 1,1-disubstituted olefin (Formula I) and sulfonyl hydrazide (Formula II) in an organic solvent such as acetonitrile/water mixture.
- Add a copper salt catalyst, preferably copper trifluoromethanesulfonate, to the reaction mixture under an oxygen or air atmosphere.
- Heat the reaction to 40-80°C for 4-24 hours, then perform standard post-treatment and purification to isolate the beta-carbonyl sulfone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical yield. The shift from scarce sulfinic acid salts to abundant sulfonyl hydrazides and olefins fundamentally alters the cost structure of raw material acquisition. Sulfonyl hydrazides are commodity chemicals produced on a massive scale for various industries, ensuring a stable and competitive pricing landscape. This transition effectively mitigates the risk of supply disruptions associated with niche reagents, thereby enhancing supply chain reliability for long-term production contracts. Furthermore, the elimination of stoichiometric oxidants and complex purification steps like cation exchange membrane filtration significantly reduces the consumption of auxiliary materials and energy. This streamlining of the process flow translates directly into substantial cost savings in manufacturing overheads, making the final beta-carbonyl sulfone products more price-competitive in the global market.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven by the replacement of expensive and hard-to-source sulfinic acid salts with cost-effective sulfonyl hydrazides and olefins. Additionally, the use of molecular oxygen from air as the oxidant removes the recurring cost of purchasing chemical oxidants like peroxides or hypervalent iodine reagents. The simplified workup procedure, which avoids specialized membrane treatments and extensive chromatography, reduces labor hours and solvent consumption. These factors collectively contribute to a significantly reduced cost of goods sold (COGS), allowing for better margin management in high-volume production scenarios without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. 1,1-disubstituted olefins and sulfonyl hydrazides are stocked by multiple major chemical distributors worldwide, reducing dependency on single-source suppliers. The robustness of the reaction conditions, which tolerate moisture and air to a certain extent, lowers the barrier for entry for contract manufacturing organizations (CMOs), expanding the pool of potential production partners. This flexibility ensures that production schedules can be maintained even during regional logistical disruptions, securing the continuity of supply for critical downstream pharmaceutical and agrochemical applications.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The reaction generates benign byproducts such as nitrogen gas and water, minimizing the burden on waste treatment facilities. The absence of heavy metal waste (since copper is used in catalytic amounts and can be recovered) and hazardous oxidants simplifies regulatory compliance and reduces disposal costs. The mild temperature range (40-80°C) allows for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, facilitating seamless scale-up from kilogram to multi-ton scales while maintaining strict environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-carbonyl sulfone synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the advantages of this copper-catalyzed method over traditional sulfinic acid routes?
A: Traditional methods often rely on scarce sulfinic acid salts or harsh oxidation of sulfides. This novel approach uses readily available sulfonyl hydrazides and olefins, operates under mild conditions (65°C), and eliminates the need for complex post-reaction treatments like cation exchange membranes.
Q: What is the substrate scope for this beta-carbonyl sulfone synthesis?
A: The method demonstrates excellent functional group tolerance. It accommodates various substituents on the aryl rings including halogens (F, Cl, Br), electron-donating groups (methoxy, methyl), and electron-withdrawing groups (cyano, ester, nitro), as well as heteroaryl systems like furan and thiophene.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. It utilizes inexpensive copper catalysts, common solvents like acetonitrile and water, and molecular oxygen as the terminal oxidant. The high yields (up to 93%) and simple workup procedures make it ideal for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carbonyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the next generation of therapeutic agents and functional materials. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including oxygen sparging systems and rigorous QC labs capable of verifying stringent purity specifications for every batch. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to explore how this innovative technology can optimize your supply chain and reduce your overall development costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can accelerate your time-to-market and enhance your competitive edge in the marketplace.
