Advanced Silver-Catalyzed Synthesis of Beta-Carbonyl Sulfones for Pharmaceutical Applications
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient, environmentally benign, and cost-effective methodologies for constructing critical molecular scaffolds. A significant breakthrough in this domain is detailed in Chinese Patent CN110818600B, which discloses a novel method for preparing beta-carbonyl sulfone compounds. These sulfone-based framework compounds are indispensable intermediates in modern organic chemistry, serving as pivotal precursors for Julia olefination, Ramberg-Bäcklund reactions, and Smiles rearrangements. The ability to access these structures efficiently is paramount for the development of active pharmaceutical ingredients (APIs) and advanced fine chemicals. The patented technology leverages a silver-catalyzed coupling strategy that fundamentally shifts the paradigm from traditional, harsh synthetic routes to a greener, more operationally simple process. By utilizing alpha-carbonyl diazo compounds and sodium aryl sulfinates as readily available starting materials, this method achieves high yields under mild conditions, addressing long-standing challenges in sulfone chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-carbonyl sulfones has been plagued by significant operational and economic drawbacks that hinder their widespread adoption in industrial settings. Early methodologies, such as those reported by Jorgensen, necessitated a multi-step sequence involving the reaction of thiol compounds with alpha-bromocarbonyl compounds under alkaline conditions to form thioethers, followed by oxidation using excessive amounts of hazardous reagents like periodic acid (H5IO6) and chromium trioxide (CrO3). This approach not only suffers from poor atom economy due to the requirement for large excesses of oxidizing agents but also generates toxic heavy metal waste, posing severe environmental compliance issues. Similarly, strategies employing Claisen condensation, as described by Knospe, rely on strong bases like sodium ethoxide, which are corrosive and require stringent safety measures, while offering a narrow substrate scope that limits their utility for diverse molecular architectures. Furthermore, methods utilizing hypervalent iodine reagents, such as PhI(OH)OTs, involve expensive starting materials and often result in poor atom economy, making them economically unviable for large-scale commercial production.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN110818600B introduces a streamlined, one-pot catalytic cycle that eliminates the need for pre-functionalized thioethers or expensive hypervalent iodine oxidants. The core innovation lies in the use of a silver nitrate catalyst system paired with 1,10-phenanthroline as a ligand and potassium persulfate as a terminal oxidant. This combination facilitates a direct coupling between sodium aryl sulfinates and alpha-carbonyl diazo compounds in a mixed solvent system of acetonitrile and water. The reaction proceeds smoothly under air atmosphere at a moderate temperature of 70°C, typically completing within 4 hours. This transition to a radical-mediated mechanism not only simplifies the operational workflow by reducing the number of synthetic steps but also drastically improves the environmental profile of the process by avoiding toxic chromium reagents and strong bases. The robustness of this new approach is evidenced by its ability to tolerate a wide range of functional groups, thereby expanding the accessible chemical space for medicinal chemists.
Mechanistic Insights into Ag-Catalyzed Radical Coupling
The mechanistic pathway of this transformation is believed to involve a silver-mediated radical generation process that ensures high efficiency and selectivity. Initially, the silver catalyst, likely in coordination with the 1,10-phenanthroline ligand, interacts with the potassium persulfate oxidant to generate reactive sulfate radical anions. These radicals subsequently oxidize the sodium aryl sulfinate to produce aryl sulfonyl radicals. Concurrently, the silver species activates the alpha-carbonyl diazo compound, potentially forming a silver-carbene intermediate or facilitating the release of nitrogen to generate an alpha-carbonyl radical species. The cross-coupling of the aryl sulfonyl radical with the alpha-carbonyl radical leads to the formation of the desired beta-carbonyl sulfone skeleton. This radical manifold is highly advantageous as it bypasses the high energy barriers associated with ionic pathways, allowing the reaction to proceed under neutral and mild conditions. The use of water as a co-solvent further stabilizes the ionic intermediates and enhances the solubility of the inorganic oxidant, contributing to the overall reaction kinetics.
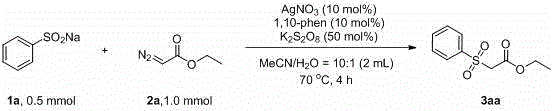
From an impurity control perspective, this catalytic system offers distinct advantages over traditional stoichiometric oxidations. The mild reaction temperature of 70°C minimizes thermal degradation of sensitive functional groups, which is a common issue in high-temperature reflux operations. Furthermore, the specificity of the radical coupling reduces the formation of over-oxidized byproducts or homocoupling impurities that often plague sulfone synthesis. The post-reaction workup is notably straightforward; quenching with saturated sodium sulfite effectively removes residual oxidants, and standard extraction followed by silica gel chromatography yields products of high purity. The patent data indicates that even on a 10 mmol scale, the reaction maintains high efficiency, suggesting that the mechanism is robust against concentration effects, which is a critical factor for process scalability.
How to Synthesize Beta-Carbonyl Sulfone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and reproducibility. The protocol is designed to be user-friendly, utilizing commercially available reagents that do not require specialized handling equipment beyond standard glassware and heating mantles. The following guide outlines the standardized procedure derived from the patent examples, ensuring that technical teams can replicate the high yields reported in the literature. For detailed operational specifics regarding stoichiometry and purification, please refer to the structured guide below.
- Combine silver nitrate catalyst, 1,10-phenanthroline ligand, potassium persulfate oxidant, sodium aryl sulfinate, and alpha-carbonyl diazo compound in a MeCN/H2O solvent system.
- Heat the reaction mixture to 70°C and stir under air atmosphere for approximately 4 hours to facilitate the radical coupling reaction.
- Quench the reaction with saturated sodium sulfite, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this silver-catalyzed methodology represents a strategic opportunity to optimize manufacturing costs and enhance supply reliability. The shift away from proprietary or exotic reagents towards commodity chemicals significantly de-risks the supply chain. By replacing expensive hypervalent iodine reagents and toxic chromium oxidants with inexpensive potassium persulfate and silver nitrate, the raw material cost profile is substantially improved. Additionally, the elimination of strong bases reduces the need for specialized corrosion-resistant equipment and lowers the costs associated with hazardous waste disposal. The simplicity of the workup procedure, which avoids complex distillation or crystallization steps, further contributes to operational efficiency, allowing for faster batch turnover times and reduced labor costs.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of low-loading catalysts and cheap oxidants. Silver nitrate, while a precious metal salt, is used in catalytic amounts (10 mol%) and is significantly more affordable than palladium or rhodium alternatives often used in cross-coupling reactions. The replacement of costly PhI(OH)OTs with potassium persulfate results in drastic savings on reagent costs per kilogram of product. Furthermore, the high atom economy of the direct coupling reaction minimizes waste generation, leading to lower downstream processing costs and reduced environmental fees associated with waste treatment.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including sodium aryl sulfinates and various alpha-carbonyl diazo compounds, are widely available from global chemical suppliers, ensuring a stable and continuous supply chain. Unlike methods relying on custom-synthesized thioethers or unstable intermediates, the substrates here are robust and can be stocked in bulk without significant degradation. This availability allows manufacturers to maintain healthy inventory levels and respond quickly to market demand fluctuations. The reaction's tolerance to air and moisture also simplifies logistics, as it does not require inert gas blanketing or anhydrous conditions, reducing the complexity of storage and handling.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the successful scale-up of this reaction to at least the 10 mmol level with maintained yields, indicating strong potential for kilogram-to-ton scale production. The use of a water-acetonitrile solvent system aligns with green chemistry principles, reducing the reliance on chlorinated solvents and lowering the overall E-factor of the process. This environmental compatibility is crucial for meeting increasingly stringent regulatory standards in the pharmaceutical and fine chemical industries. The mild conditions also enhance process safety, minimizing the risk of thermal runaways and making the technology suitable for implementation in existing multipurpose reactors without major retrofitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-carbonyl sulfone synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of the method.
Q: What are the primary advantages of this silver-catalyzed method over traditional sulfone synthesis?
A: Unlike conventional methods requiring harsh oxidants like periodic acid or expensive hypervalent iodine reagents, this protocol uses cheap potassium persulfate and operates under mild conditions with excellent atom economy.
Q: Is the catalyst system cost-effective for large-scale manufacturing?
A: Yes, the process utilizes silver nitrate, a relatively inexpensive silver salt compared to palladium or rhodium complexes, along with commercially available ligands and oxidants, significantly reducing raw material costs.
Q: Does this methodology support complex substrate scopes?
A: The patent demonstrates broad substrate tolerance, successfully synthesizing derivatives from various aryl sulfinates and complex alpha-carbonyl diazo compounds, including steroid-based substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carbonyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality intermediates. Our technical team is adept at optimizing the silver-catalyzed sulfone synthesis described in CN110818600B, leveraging our rigorous QC labs and stringent purity specifications to deliver products that meet the highest industry standards. We understand that every project has unique requirements, and our state-of-the-art facilities are equipped to handle the specific nuances of radical coupling chemistries.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you require custom synthesis of complex sulfone derivatives or scale-up services for clinical trial materials, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you streamline your supply chain with reliable, cost-effective solutions.
