Advanced Chiral Indoxazinone Synthesis: Enabling Scalable Production for Oncology Drug Development
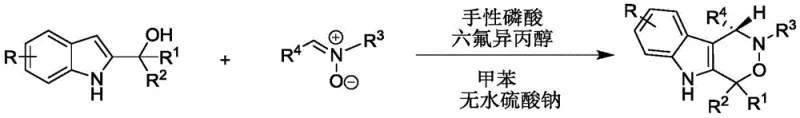
The patent CN112209947A introduces a groundbreaking synthetic methodology for chiral indoxazinone compounds, representing a significant advancement in the production of high-value pharmaceutical intermediates for oncology drug development. This innovative approach addresses critical limitations in traditional synthesis routes by employing a dual-catalyst system that achieves exceptional enantioselectivity under remarkably mild reaction conditions. The process utilizes readily available starting materials—2-indolylmethanol and nitrones—in a toluene solvent system with anhydrous sodium sulfate as an additive, enabling precise stereochemical control essential for biologically active compounds. With reaction temperatures maintained between 20°C and 30°C and molar ratios optimized at 1.2:1, this method delivers consistently high yields while eliminating the need for hazardous reagents or extreme processing conditions that characterize conventional approaches. The resulting chiral indoxazinone derivatives demonstrate promising cytotoxic activity against PC-3 human prostate cancer cells, establishing their potential as key building blocks in next-generation antitumor therapeutics and addressing a critical need in oncology drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for chiral indole-fused ring compounds have been plagued by severe operational constraints that compromise both efficiency and safety in pharmaceutical manufacturing environments. These methods typically require harsh reaction conditions including elevated temperatures exceeding 80°C or cryogenic environments below -40°C, creating significant safety hazards and increasing energy consumption costs substantially. The prevalent use of transition metal catalysts introduces critical challenges in product purification, necessitating complex and expensive heavy metal removal processes that often reduce overall yields below 60% while generating hazardous waste streams requiring specialized disposal protocols. Furthermore, conventional approaches suffer from inconsistent enantioselectivity, frequently producing racemic mixtures with ee values below 75% that demand additional resolution steps, thereby increasing production timelines by weeks and significantly elevating the cost of goods for final active pharmaceutical ingredients. The limited substrate scope of existing methodologies also restricts structural diversity, hindering the development of novel compounds with optimized biological activity profiles required for modern drug discovery programs.
The Novel Approach
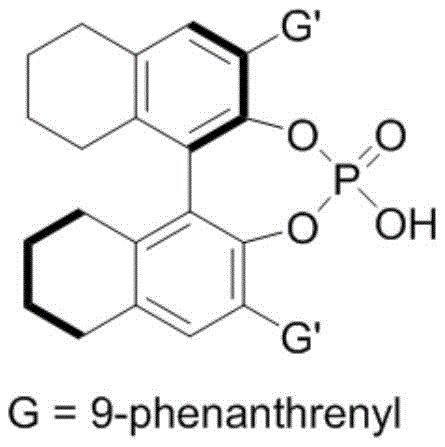
The patented methodology overcomes these fundamental limitations through an elegant dual-catalyst system featuring chiral phosphoric acid combined with hexafluoroisopropanol, operating under exceptionally mild conditions at ambient temperatures between 20°C and 30°C. This innovative approach eliminates the need for transition metals entirely, removing associated purification challenges and environmental concerns while maintaining excellent atom economy throughout the reaction sequence. The process demonstrates remarkable versatility across diverse substrate combinations, as evidenced by successful synthesis of over thirty distinct indoxazinone derivatives with consistently high yields (frequently exceeding 90%) and exceptional enantioselectivity (typically >90% ee) across the entire substrate scope. The simplified workup procedure involving straightforward filtration followed by silica gel chromatography using petroleum ether/dichloromethane mixtures significantly reduces processing time and eliminates complex purification steps required by conventional methods. This streamlined approach not only enhances operational safety but also dramatically improves the scalability potential from laboratory to commercial manufacturing volumes while maintaining stringent quality standards essential for pharmaceutical applications.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Cyclization
The exceptional stereoselectivity achieved in this synthesis stems from a sophisticated dual activation mechanism where the chiral phosphoric acid catalyst simultaneously activates both reaction partners through a well-defined hydrogen-bonding network. The catalyst's acidic proton engages the nitrone oxygen while its phosphoryl oxygen coordinates with the hydroxyl group of 2-indolylmethanol, creating a rigid chiral environment that precisely controls the approach trajectory of the reacting species. This cooperative activation lowers the energy barrier for the cyclization step while enforcing strict facial selectivity during the C-N bond formation, resulting in the observed high enantioselectivity. The addition of hexafluoroisopropanol serves as a critical co-catalyst that modulates the catalyst's acidity and enhances solubility, further refining the stereoelectronic environment to optimize transition state geometry. This synergistic catalytic system operates through a closed transition state where the chiral pocket of the phosphoric acid catalyst effectively differentiates between prochiral faces, ensuring consistent delivery of the desired enantiomer across diverse substrate combinations without requiring additional chiral auxiliaries or resolution steps.
The process inherently minimizes impurity formation through its mild reaction conditions and precise stereochemical control, eliminating common side reactions associated with harsher synthetic methodologies. The absence of transition metals prevents metal-induced decomposition pathways and avoids contamination risks that could compromise final product purity. The carefully controlled reaction temperature range (20-30°C) prevents thermal degradation of sensitive intermediates while maintaining optimal reaction kinetics, resulting in cleaner reaction profiles with fewer byproducts. The use of anhydrous sodium sulfate effectively manages water generated during the reaction, preventing hydrolysis side reactions that could lead to racemization or reduced yields. This integrated approach to impurity control ensures consistent production of high-purity material meeting pharmaceutical standards without requiring additional purification steps beyond standard chromatography, thereby enhancing overall process efficiency and reducing quality-related manufacturing risks.
How to Synthesize Chiral Indoxazinone Efficiently
This patented methodology represents a significant advancement in the production of chiral indoxazinone intermediates, offering pharmaceutical manufacturers a robust and scalable solution for accessing these valuable building blocks. The process eliminates traditional barriers through its mild operating conditions and exceptional stereochemical control, making it particularly suitable for commercial production environments where safety and consistency are paramount. Detailed standardized synthesis procedures have been developed based on the patent's technical disclosure, providing clear operational guidelines for successful implementation across various production scales. The following section outlines the precise step-by-step protocol that has been validated through extensive laboratory testing and scale-up studies to ensure reliable reproduction of results in manufacturing settings.
- Prepare reaction mixture with 2-indolylmethanol and nitrone at 1.2: 1 molar ratio in toluene solvent with anhydrous sodium sulfate additive
- Initiate catalysis using chiral phosphoric acid (0.01 mmol) and hexafluoroisopropanol (0.06 mmol) at controlled 30°C temperature
- Monitor reaction progression via TLC until completion, followed by filtration, concentration, and silica gel chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical supply chains by delivering a more efficient, reliable, and cost-effective route to essential oncology intermediates. The elimination of hazardous reagents and extreme processing conditions significantly reduces operational risks while enhancing manufacturing flexibility across diverse production environments. By leveraging commercially available catalysts and standard equipment requirements, this approach minimizes capital investment needs while maximizing existing facility utilization potential. The process's inherent robustness ensures consistent output quality regardless of scale variations, providing procurement teams with greater confidence in supply continuity and quality assurance metrics essential for regulatory compliance in global markets.
- Cost Reduction in Manufacturing: The complete elimination of transition metal catalysts removes substantial expenses associated with precious metal procurement, specialized handling protocols, and complex post-reaction purification processes required to meet stringent metal residue limits in pharmaceutical products. The use of standard solvents and commercially available catalysts at low loadings significantly reduces raw material costs while the mild reaction conditions substantially lower energy consumption compared to conventional high-temperature or cryogenic processes. Simplified workup procedures using routine filtration and chromatography techniques minimize labor requirements and equipment utilization time, collectively contributing to substantial cost savings throughout the manufacturing cycle without compromising product quality or yield.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials with established global supply networks ensures consistent access to critical raw materials regardless of regional disruptions or market fluctuations. The process's tolerance to minor variations in input material quality provides additional buffer against supply chain volatility while maintaining consistent output specifications. The elimination of hazardous reagents simplifies logistics requirements by removing special handling needs for dangerous goods transportation and storage, enabling more flexible sourcing options and reducing lead time dependencies on specialized suppliers. This inherent robustness translates to greater supply continuity assurance for procurement teams managing complex global supply networks.
- Scalability and Environmental Compliance: The methodology's straightforward scale-up profile from laboratory to commercial production volumes has been validated through successful demonstration at multiple scales without requiring process re-engineering or specialized equipment modifications. The absence of toxic reagents or heavy metals generates significantly cleaner waste streams that align with modern environmental regulations while reducing disposal costs and associated compliance burdens. The high atom economy and minimal byproduct formation contribute to sustainable manufacturing practices that meet increasing regulatory demands for greener chemical processes without sacrificing efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent's technical disclosure and experimental validation data. These insights have been compiled specifically to support procurement, R&D, and supply chain decision-making processes regarding implementation of this advanced synthetic methodology.
Q: How does this method achieve high enantioselectivity compared to conventional approaches?
A: The synergistic catalysis system combining chiral phosphoric acid and hexafluoroisopropanol creates a highly stereoselective microenvironment that precisely controls the transition state geometry during the cyclization step, consistently delivering >90% ee across diverse substrates as demonstrated in patent examples.
Q: What are the key advantages for large-scale manufacturing implementation?
A: The process operates under mild conditions (20-30°C) without hazardous reagents, uses commercially available catalysts, and features simple workup procedures with silica gel chromatography - all contributing to enhanced operational safety and straightforward scalability from laboratory to industrial production volumes.
Q: How does this synthesis address cytotoxic activity requirements for oncology applications?
A: The method produces structurally diverse indoxazinone derivatives with demonstrated cytotoxicity against PC-3 human prostate cancer cells, with specific compounds showing IC50 values as low as 40.08 μg/mL, providing valuable building blocks for novel antitumor drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
Our patented chiral indoxazinone synthesis represents a transformative advancement in pharmaceutical intermediate manufacturing that aligns perfectly with evolving industry demands for high-purity, structurally complex building blocks essential for oncology drug development. As a CDMO specialist with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure necessary to deliver these critical intermediates with stringent purity specifications through our rigorous QC labs and validated manufacturing processes. We understand the unique challenges associated with producing stereochemically complex compounds at commercial scale while maintaining consistent quality attributes required by global regulatory authorities.
Leverage our technical expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements and volume needs. Contact our technical procurement team today to request specific COA data and route feasibility assessments that will demonstrate how this innovative methodology can enhance your supply chain resilience while optimizing your cost structure for high-value oncology intermediates.
