Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharmaceutical Production
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Scalable Pharmaceutical Production
The pharmaceutical industry constantly seeks robust methodologies for constructing complex chiral scaffolds, particularly those exhibiting potent biological activities. Patent CN112209947A introduces a groundbreaking approach to synthesizing chiral indoxazinone compounds, a class of molecules characterized by their fused indole-oxazine ring systems. These structures are not merely academic curiosities; they serve as critical intermediates in the development of novel antitumor agents, specifically demonstrating cytotoxicity against PC-3 human prostate cancer cells. The disclosed technology leverages a dual catalytic system involving chiral phosphoric acids and hexafluoroisopropanol to achieve exceptional stereocontrol under remarkably mild conditions. This represents a significant leap forward from traditional synthetic routes that often suffer from harsh reaction parameters and poor enantiomeric excess. For R&D directors and process chemists, this patent offers a viable pathway to access high-value heterocyclic libraries with improved efficiency and safety profiles.
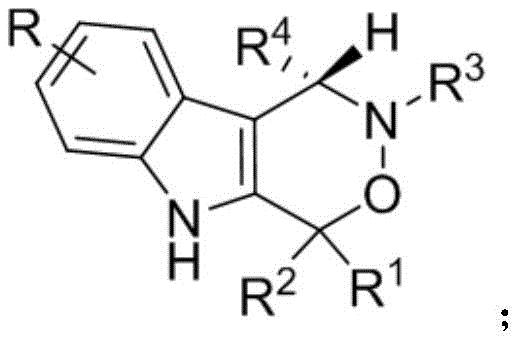
At the core of this innovation lies the structural versatility of the target molecule, as depicted in the general formula. The ability to modulate substituents at multiple positions (R, R1, R2, R3, R4) allows for extensive structure-activity relationship (SAR) studies, which is paramount for optimizing drug candidates. The patent explicitly details that R groups can range from simple alkyl chains to complex heteroaryl systems, providing a broad chemical space for exploration. This flexibility is crucial for medicinal chemists aiming to fine-tune pharmacokinetic properties or enhance binding affinity to biological targets. Furthermore, the synthesis method described ensures that this structural diversity does not come at the cost of stereochemical integrity, maintaining high optical purity across a wide substrate scope. Such consistency is a key metric for reliable pharmaceutical intermediate suppliers who must guarantee batch-to-batch reproducibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral indole-fused ring systems has been fraught with significant technical challenges that hinder industrial adoption. Prior art methods frequently rely on transition metal catalysts or extreme reaction conditions, such as high temperatures and strong acidic or basic environments, which can lead to decomposition of sensitive functional groups. These violent conditions not only pose safety risks in a manufacturing setting but also often result in racemic mixtures that require costly and wasteful resolution steps to isolate the active enantiomer. Additionally, the use of heavy metals introduces stringent regulatory hurdles regarding residual metal limits in final drug substances, necessitating complex purification protocols that drive up production costs. The background section of the patent highlights that existing chiral indole compounds often exhibit weak cytotoxic activity or insensitivity to key cancer cell lines like PC-3, limiting their therapeutic potential. Consequently, there is an urgent demand for a synthetic strategy that circumvents these pitfalls while delivering biologically active, enantiopure products.
The Novel Approach
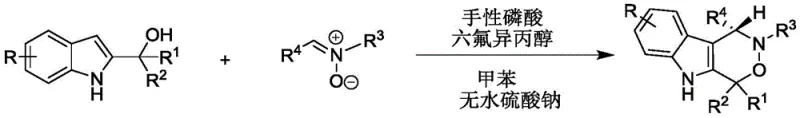
The methodology presented in CN112209947A fundamentally shifts the paradigm by employing an organocatalytic cascade reaction that operates under ambient-like conditions. By utilizing 2-indolemethanol and nitrones as starting materials, the process achieves a direct cyclization to form the indoxazinone core with high atom economy. The reaction is conducted in toluene at a mild temperature range of 20-30°C, drastically reducing energy consumption compared to thermal processes. A key feature of this novel approach is the synergistic effect between the chiral phosphoric acid and hexafluoroisopropanol (HFIP), which activates the substrates through hydrogen bonding networks rather than aggressive chemical reagents. This gentle activation mode preserves the integrity of delicate functional groups and minimizes side reactions, leading to cleaner reaction profiles and higher isolated yields. For procurement managers, this translates to cost reduction in API manufacturing by simplifying the downstream processing and eliminating the need for expensive metal scavengers.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
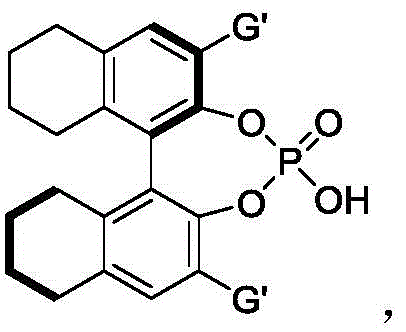
The success of this synthesis hinges on the precise molecular recognition and activation facilitated by the chiral phosphoric acid catalyst. These catalysts, typically derived from BINOL (1,1'-bi-2-naphthol) skeletons, act as Brønsted acids that protonate the nitrone or activate the hydroxyl group of the indolemethanol via hydrogen bonding. The patent specifies several preferred catalyst structures, including binaphthyl, octahydrobinaphthyl, and spiro skeleton derivatives, each tailored to create a specific chiral environment around the reactive center. The presence of bulky substituents, such as the 9-phenanthryl group shown in the preferred embodiment, creates a steric pocket that effectively discriminates between the pro-chiral faces of the incoming nucleophile. This steric shielding is what drives the high enantioselectivity observed, often exceeding 90% ee across various substrates. Understanding this mechanistic nuance is vital for scaling up the process, as slight deviations in catalyst loading or purity can impact the optical outcome.
Beyond the primary catalyst, the role of hexafluoroisopropanol (HFIP) as a co-catalyst or additive cannot be overstated in this mechanism. HFIP is known for its unique ability to stabilize charged intermediates and enhance hydrogen bond donor strength through its highly fluorinated structure. In this reaction system, it likely assists in organizing the transition state, ensuring that the 2-indolemethanol and nitrone align perfectly for the cyclization event. This dual activation strategy suppresses background non-catalyzed reactions that would otherwise lead to racemic byproducts. From an impurity control perspective, this mechanism is highly favorable because it minimizes the formation of oligomers or polymeric side products that are common in uncontrolled radical or ionic polymerizations. The result is a reaction mixture that is easier to purify, requiring less solvent and fewer chromatography steps, which directly correlates to reduced waste generation and lower environmental impact. For supply chain heads, this efficiency means faster turnaround times and more predictable production schedules.
How to Synthesize Chiral Indoxazinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters outlined in the patent to ensure maximum yield and stereoselectivity. The process begins with the preparation of the reaction vessel, where 2-indolemethanol and the nitrone derivative are dissolved in anhydrous toluene. Anhydrous sodium sulfate is added to the mixture, serving as a drying agent to sequester water produced during the condensation, thereby driving the equilibrium towards product formation. The choice of solvent is critical; toluene provides the ideal polarity balance to solubilize the organic substrates while allowing the catalyst to function effectively. Once the reagents are mixed, the chiral phosphoric acid and HFIP are introduced, initiating the catalytic cycle. The reaction is then allowed to proceed at a controlled temperature of 30°C, monitored closely by thin-layer chromatography (TLC) to determine the endpoint. Detailed standardized synthesis steps follow below.
- Prepare the reaction mixture by combining 2-indolemethanol and nitrone in toluene with anhydrous sodium sulfate.
- Add chiral phosphoric acid and hexafluoroisopropanol as co-catalysts and stir the mixture at 20-30°C.
- Monitor reaction progress via TLC, then filter, concentrate, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the economic implications of this patented technology are substantial. The shift from metal-catalyzed to organocatalytic processes eliminates the dependency on precious metals like palladium, rhodium, or iridium, which are subject to volatile market prices and supply chain disruptions. This transition inherently lowers the raw material cost base and removes the regulatory burden associated with heavy metal clearance in final pharmaceutical products. Furthermore, the mild reaction conditions (20-30°C) imply that standard glass-lined reactors can be used without the need for specialized high-pressure or cryogenic equipment, significantly reducing capital expenditure (CAPEX) for manufacturing facilities. The simplicity of the work-up procedure, involving basic filtration and concentration, streamlines the production workflow, allowing for higher throughput in existing infrastructure. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging resins and complex purification trains, leading to significant operational savings. Additionally, the high atom economy of the reaction ensures that a greater proportion of raw materials are converted into the desired product, minimizing waste disposal costs. The use of readily available solvents like toluene and common drying agents further keeps consumable expenses low. By avoiding extreme temperatures, the process also reduces energy consumption for heating or cooling, contributing to a lower carbon footprint and reduced utility bills. These cumulative efficiencies allow for a more competitive pricing structure for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as 2-indolemethanol and nitrones ensures a consistent supply of starting materials, mitigating the risk of production delays due to raw material shortages. The robustness of the organocatalytic system means that the process is less sensitive to minor fluctuations in reaction parameters, resulting in reproducible batches that meet strict quality specifications. This reliability is crucial for maintaining long-term contracts with downstream drug manufacturers who require guaranteed delivery schedules. Moreover, the scalability of the method from gram to kilogram scales has been demonstrated, proving its viability for commercial production without the need for extensive re-optimization. This stability fosters trust between suppliers and buyers, securing the supply chain against unforeseen disruptions.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, aligning with increasingly stringent global environmental regulations and green chemistry principles. The absence of toxic heavy metals simplifies wastewater treatment and reduces the environmental liability associated with chemical manufacturing. The high yields and selectivity reduce the volume of solvent required for purification, lowering the overall solvent intensity of the process. This eco-friendly profile not only aids in regulatory compliance but also enhances the brand reputation of companies adopting sustainable manufacturing practices. The ability to scale this reaction safely in large vessels without exothermic runaway risks makes it an ideal candidate for multi-ton production campaigns, ensuring a steady flow of material to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral indoxazinone compounds. These insights are derived directly from the experimental data and theoretical framework provided in the patent documentation. Understanding these nuances helps potential partners evaluate the feasibility of integrating this technology into their own development pipelines. The answers reflect the current state of the art as described in CN112209947A, offering a transparent view of the capabilities and limitations of the method.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method utilizes mild reaction conditions (20-30°C) and avoids violent conditions common in prior art, significantly improving safety and reducing energy consumption while achieving high enantioselectivity.
Q: What is the biological activity profile of the synthesized compounds?
A: The synthesized chiral indoxazinone compounds exhibit specific cytotoxic activity against PC-3 human prostate cancer cells, with certain derivatives showing potent inhibitory effects suitable for antitumor drug development.
Q: Which catalysts are preferred for achieving high enantioselectivity?
A: Binaphthyl skeleton derivatives, particularly those with 9-phenanthryl groups, combined with hexafluoroisopropanol, are preferred for maximizing yield and optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced technologies like the one described in CN112209947A to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We understand that the integrity of chiral intermediates is non-negotiable in drug development; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch meets the highest standards of optical and chemical purity. Whether you require milligram quantities for initial screening or metric tons for clinical trials, our flexible manufacturing capabilities are designed to adapt to your specific timeline and volume requirements. We are committed to being a strategic partner that adds value through technical expertise and operational excellence.
We invite you to explore how our capabilities can accelerate your drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis of chiral indoxazinones can enhance your supply chain efficiency. Let us collaborate to bring your next-generation therapeutics to market faster and more economically.
