Advanced Synthesis of Chiral Indoxazinone Compounds for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking robust synthetic routes for complex heterocyclic scaffolds that possess significant biological activity. Patent CN112209947A introduces a groundbreaking methodology for the construction of chiral indoxazinone compounds, a class of molecules that has garnered attention for their potential as antitumor agents. This specific innovation addresses the critical need for efficient access to enantiomerically pure indole-fused ring systems, which are often challenging to synthesize using traditional methods. The disclosed technology utilizes a sophisticated organocatalytic system involving chiral phosphoric acid and hexafluoroisopropanol to drive the reaction between 2-indolemethanol and nitrones. 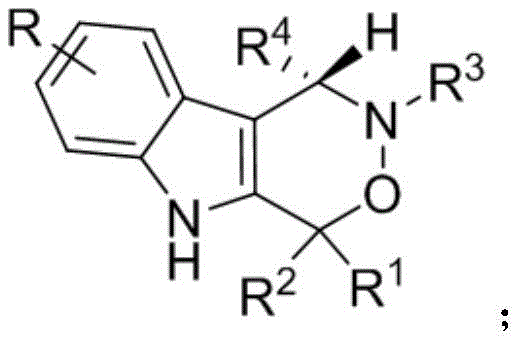 This approach not only ensures high stereocontrol but also operates under remarkably mild conditions, avoiding the harsh environments typically associated with heterocycle formation. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable chiral indoxazinone supplier capable of delivering high-purity intermediates essential for oncology drug discovery pipelines. The structural diversity achievable through this method allows for the rapid generation of analog libraries, accelerating the lead optimization phase in new drug development projects.
This approach not only ensures high stereocontrol but also operates under remarkably mild conditions, avoiding the harsh environments typically associated with heterocycle formation. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable chiral indoxazinone supplier capable of delivering high-purity intermediates essential for oncology drug discovery pipelines. The structural diversity achievable through this method allows for the rapid generation of analog libraries, accelerating the lead optimization phase in new drug development projects.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral indole-fused ring compounds has been plagued by significant technical hurdles that impede efficient commercial production. Traditional pathways often rely on transition metal catalysts which introduce the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet stringent pharmaceutical standards. Furthermore, many existing protocols require extreme reaction conditions, such as high temperatures or strong acidic environments, which can lead to substrate decomposition and poor reproducibility on a larger scale. These violent reaction conditions not only pose safety risks in a manufacturing setting but also result in lower yields and reduced enantioselectivity, ultimately increasing the cost of goods sold. The inability to consistently produce single enantiomers with high optical purity is a major bottleneck, as the biological activity of these compounds is often strictly dependent on their stereochemical configuration. Consequently, manufacturers face difficulties in scaling these processes without compromising quality or incurring prohibitive costs.
The Novel Approach
In stark contrast, the methodology described in CN112209947A offers a transformative solution by leveraging dual organocatalysis to overcome these longstanding limitations. By employing a chiral phosphoric acid catalyst in conjunction with hexafluoroisopropanol, the reaction proceeds through a highly organized transition state that favors the formation of the desired enantiomer with exceptional selectivity. This metal-free approach eliminates the concern of residual metal impurities, thereby simplifying the downstream processing and reducing the overall environmental footprint of the manufacturing process. The reaction conditions are notably mild, typically occurring between 20-30°C, which significantly lowers energy consumption and enhances operational safety within the plant. Moreover, the use of toluene as a solvent and anhydrous sodium sulfate as an additive creates a robust system that tolerates a wide range of substrate variations. This flexibility allows for the synthesis of structurally diverse products with high yields, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing where efficiency and purity are paramount.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Cyclization
The success of this synthetic route hinges on the intricate interplay between the chiral phosphoric acid catalyst and the hydrogen-bonding donor hexafluoroisopropanol. The chiral phosphoric acid acts as a Brønsted acid, activating the nitrone electrophile through hydrogen bonding interactions, while simultaneously organizing the spatial arrangement of the reacting species within its chiral pocket. 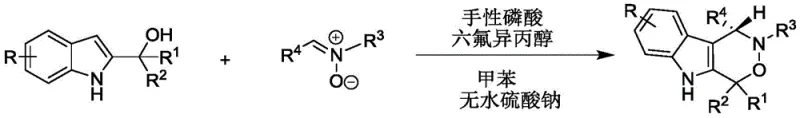 The presence of hexafluoroisopropanol further enhances this activation by stabilizing the developing charge in the transition state through a cooperative hydrogen-bonding network. This dual activation strategy ensures that the nucleophilic attack of the 2-indolemethanol occurs with precise stereocontrol, leading to the formation of the chiral indoxazinone core with high enantiomeric excess. The specific structure of the catalyst, often derived from binaphthyl or spiro skeletons as shown in related embodiments, provides the necessary steric bulk to differentiate between the prochiral faces of the substrate.
The presence of hexafluoroisopropanol further enhances this activation by stabilizing the developing charge in the transition state through a cooperative hydrogen-bonding network. This dual activation strategy ensures that the nucleophilic attack of the 2-indolemethanol occurs with precise stereocontrol, leading to the formation of the chiral indoxazinone core with high enantiomeric excess. The specific structure of the catalyst, often derived from binaphthyl or spiro skeletons as shown in related embodiments, provides the necessary steric bulk to differentiate between the prochiral faces of the substrate. 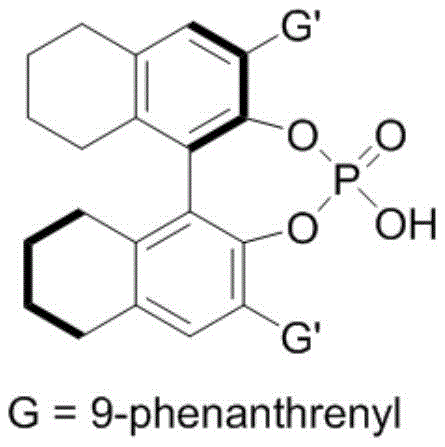 For technical teams, understanding this mechanism is crucial for troubleshooting and optimizing the process, as slight modifications to the catalyst substituents can fine-tune the selectivity for different substrate classes. This level of mechanistic clarity provides confidence in the reproducibility of the process across different batches and scales.
For technical teams, understanding this mechanism is crucial for troubleshooting and optimizing the process, as slight modifications to the catalyst substituents can fine-tune the selectivity for different substrate classes. This level of mechanistic clarity provides confidence in the reproducibility of the process across different batches and scales.
Regarding impurity control, the mild nature of this organocatalytic system inherently suppresses the formation of side products that are common in harsher chemical environments. The high chemoselectivity of the catalyst ensures that only the desired cyclization pathway is favored, minimizing the generation of regioisomers or over-reacted byproducts. The use of anhydrous sodium sulfate helps to sequester water produced during the reaction or present in the reagents, driving the equilibrium towards product formation and preventing hydrolysis of sensitive intermediates. This clean reaction profile translates directly to simplified purification protocols, often requiring only standard silica gel column chromatography to achieve pharmaceutical-grade purity. For quality assurance teams, this means a more consistent impurity profile and easier validation of the cleaning procedures, which are critical factors for regulatory compliance in GMP manufacturing facilities.
How to Synthesize Chiral Indoxazinone Efficiently
Implementing this synthesis in a practical setting requires careful attention to the stoichiometry and reaction parameters outlined in the patent data. The process begins with the preparation of the reaction mixture, where 2-indolemethanol and nitrone are combined in toluene with a molar ratio ranging from 1:1 to 2:1, depending on the specific substrate reactivity. The addition of the chiral phosphoric acid catalyst and hexafluoroisopropanol must be done under controlled conditions to ensure proper mixing and activation before the reaction is allowed to proceed at ambient temperatures. Monitoring the reaction progress via TLC is essential to determine the optimal endpoint, preventing over-reaction which could lead to degradation. Once the conversion is complete, the workup involves a straightforward filtration to remove the solid additive, followed by concentration and purification. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 2-indolemethanol and nitrone substrates in toluene solvent with anhydrous sodium sulfate.
- Introduce the chiral phosphoric acid catalyst and hexafluoroisopropanol additive to the mixture under stirring.
- Maintain the reaction temperature between 20-30°C until completion, then filter, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthetic methodology offers substantial strategic advantages that go beyond mere technical performance. The elimination of transition metal catalysts removes a significant cost center associated with metal scavenging and specialized waste disposal, leading to direct cost savings in the overall production budget. The mild reaction temperatures reduce the energy load required for heating or cooling reactors, contributing to a more sustainable and economically efficient manufacturing process. Furthermore, the robustness of the reaction conditions implies a lower risk of batch failures due to operational variances, enhancing the reliability of supply for downstream drug formulation teams. This stability is crucial for maintaining continuous production schedules and meeting tight delivery windows for clinical trial materials.
- Cost Reduction in Manufacturing: The organocatalytic nature of this process fundamentally alters the cost structure by removing the need for expensive noble metal catalysts and the associated rigorous purification steps required to meet residual metal limits. By utilizing readily available organic acids and solvents, the raw material costs are stabilized and less susceptible to the volatility often seen in the precious metals market. Additionally, the high yields and selectivity reduce the amount of starting material wasted on side products, maximizing the atom economy of the process. These factors combine to create a leaner manufacturing model that supports significant cost reduction in pharmaceutical intermediate manufacturing without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: The simplicity of the reaction setup and the use of common industrial solvents like toluene ensure that the supply chain is resilient to disruptions in specialty chemical availability. Since the process does not rely on exotic reagents or complex equipment, it can be easily replicated across multiple manufacturing sites, providing redundancy and security of supply. The mild conditions also extend the lifespan of reactor equipment by reducing corrosion and stress, leading to less downtime for maintenance. This operational reliability translates to reducing lead time for high-purity pharmaceutical intermediates, allowing partners to respond more agilely to market demands and clinical trial timelines.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of exothermic hazards typically associated with aggressive chemical transformations. The benign nature of the reagents simplifies the handling of effluent streams, making it easier to comply with increasingly strict environmental regulations regarding waste discharge. The high selectivity minimizes the generation of hazardous byproducts, reducing the burden on waste treatment facilities. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key criterion for vendor selection in the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral compounds. The answers are derived directly from the experimental data and beneficial effects reported in the patent literature, ensuring accuracy and relevance for decision-makers. Understanding these details helps in assessing the feasibility of integrating this technology into existing drug development portfolios. We encourage technical teams to review these points when evaluating potential partnerships for custom synthesis projects.
Q: What represents the key advantage of this chiral indoxazinone synthesis method?
A: The primary advantage lies in the use of a chiral phosphoric acid organocatalyst system which achieves extremely high enantioselectivity without requiring toxic transition metals, ensuring a cleaner product profile suitable for pharmaceutical applications.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process operates under mild temperatures (20-30°C) and uses common solvents like toluene, which significantly reduces energy consumption and safety risks, making it highly adaptable for commercial scale-up.
Q: What are the biological applications of the synthesized compounds?
A: The resulting chiral indoxazinone compounds have demonstrated cytotoxic activity against PC-3 human prostate cancer cells, indicating strong potential for development into novel antitumor therapeutic agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a partner who can translate complex academic innovations into commercially viable realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN112209947A can be realized on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral indoxazinone meets the exacting standards required for oncology research. Our commitment to quality and consistency makes us a trusted ally for pharmaceutical companies navigating the complexities of modern drug synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this organocatalytic method. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Contact us today to secure a stable supply of high-quality intermediates for your next breakthrough therapy.
