Advanced Copper-Catalyzed Synthesis of Azetidino-1,4-Benzodiazepine Scaffolds for Commercial API Production
Advanced Copper-Catalyzed Synthesis of Azetidino-1,4-Benzodiazepine Scaffolds for Commercial API Production
The pharmaceutical landscape has long relied on the 1,4-benzodiazepine scaffold as a cornerstone for central nervous system therapeutics, ranging from anxiolytics like Diazepam to anticonvulsants. However, the efficient construction of novel derivatives within this class often faces significant synthetic hurdles, including harsh reaction conditions and limited functional group tolerance. Patent CN112341468A introduces a groundbreaking methodology that addresses these challenges through a streamlined, copper-catalyzed approach. This innovation not only simplifies the assembly of the azetidino-1,4-benzodiazepine core but also provides a versatile platform for downstream functionalization. For R&D teams seeking reliable pharmaceutical intermediate supplier partnerships, this technology represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols. By leveraging intramolecular C-N bond coupling followed by strategic ring-opening, the process delivers high-purity intermediates with exceptional structural diversity.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-benzodiazepine derivatives, as illustrated by classic structures in earlier literature, has been plagued by inefficiencies that hinder large-scale production. Traditional routes often require multiple protection and deprotection steps, leading to poor overall atom economy and increased waste generation. Furthermore, many established methods rely on expensive palladium catalysts or harsh acidic conditions that are incompatible with sensitive functional groups, thereby limiting the chemical space accessible to medicinal chemists. The reliance on solid-phase synthesis or complex homogeneous systems often results in difficult purification processes, where removing trace metal contaminants becomes a bottleneck for regulatory compliance. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, creating substantial friction in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
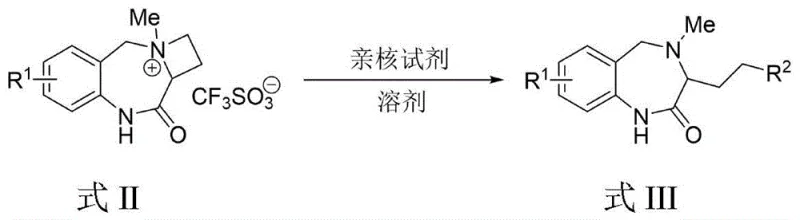
In stark contrast, the methodology disclosed in CN112341468A offers a direct and robust pathway to the target scaffolds. The core innovation lies in the one-step construction of the azetidino-1,4-benzodiazepine skeleton (Formula I) from readily available precursors (Formula IV). This approach eliminates the need for tedious multi-step sequences, drastically simplifying the operational workflow. The subsequent conversion to quaternary ammonium salts (Formula II) and their controlled ring-opening to yield functionalized derivatives (Formula III) provides a modular strategy for diversification. This flexibility is crucial for generating compound libraries for drug screening. By utilizing mild reaction conditions and achieving yields as high as 98% in specific examples, this novel approach ensures cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards required for clinical applications.

Mechanistic Insights into CuI-Catalyzed Intramolecular C-N Coupling
The heart of this synthetic breakthrough is the copper-catalyzed intramolecular C-N bond formation, which efficiently closes the seven-membered diazepine ring fused with the azetidine moiety. The reaction mechanism likely involves the coordination of the amide nitrogen and the aryl halide to the copper center, facilitated by the N,N-dimethylglycine hydrochloride (DMGC) ligand. This ligand acceleration is critical for stabilizing the active catalytic species and promoting the oxidative addition and reductive elimination steps necessary for bond formation. The use of cesium carbonate as a base ensures effective deprotonation without inducing side reactions, while 1,4-dioxane serves as an ideal solvent to maintain solubility and thermal stability during reflux at approximately 105°C. This precise control over reaction parameters minimizes the formation of by-products, ensuring a clean impurity profile that simplifies downstream purification.
Following the core construction, the functionalization strategy relies on the unique reactivity of the strained four-membered azetidine ring. Upon methylation with methyl trifluoromethanesulfonate, the nitrogen atom becomes a potent leaving group precursor within the quaternary ammonium salt structure. This activation renders the adjacent carbon atoms highly electrophilic, allowing for regioselective nucleophilic attack by reagents such as sodium azide, potassium cyanide, or sodium thiophenolate. The ring-opening event relieves the ring strain and installs diverse functional handles (azido, cyano, thiophenol groups) at the C3 position of the diazepine framework. This mechanistic understanding allows process chemists to fine-tune reaction conditions, such as temperature and solvent polarity, to maximize selectivity and yield for specific derivatives, thereby enhancing the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Azetidino-1,4-Benzodiazepine Efficiently
The practical implementation of this synthesis involves a logical sequence of three distinct transformations, each optimized for high efficiency and reproducibility. The initial cyclization step sets the foundation, requiring careful control of stoichiometry and atmosphere to prevent catalyst oxidation. Subsequent quaternization is rapid and exothermic, necessitating temperature management to ensure safety and product integrity. Finally, the nucleophilic substitution offers a wide scope for derivatization, allowing manufacturers to produce a variety of analogues from a common intermediate. For detailed operational parameters, including specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process controls identified in the patent data.
- Perform intramolecular C-N bond coupling of Formula IV precursors using CuI, DMGC, and Cs2CO3 in 1,4-dioxane under reflux to form the azetidino-1,4-benzodiazepine core (Formula I).
- React the Formula I intermediate with methyl trifluoromethanesulfonate in DCM at room temperature to generate the N-methyl quaternary ammonium salt (Formula II).
- Treat the quaternary salt (Formula II) with nucleophiles such as sodium azide, potassium cyanide, or sodium thiophenolate in DMF to effect ring-opening and yield functionalized derivatives (Formula III).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this copper-catalyzed route offers compelling economic benefits that directly impact the bottom line. The substitution of precious metal catalysts with abundant and inexpensive cuprous iodide significantly lowers the raw material input costs. Moreover, the high atom economy of the cyclization step reduces the volume of chemical waste generated, which translates to lower disposal fees and a reduced environmental footprint. The simplicity of the workup procedure, often involving basic filtration and crystallization rather than complex chromatographic separations for every step, further drives down processing costs. These efficiencies collectively contribute to substantial cost savings in API manufacturing, making the final intermediates more competitive in the global market.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the use of cheap bases like cesium carbonate drastically reduce the bill of materials. Additionally, the high yields reported (frequently exceeding 90%) mean less starting material is wasted, optimizing resource utilization. The streamlined process flow reduces labor hours and energy consumption associated with prolonged reaction times or multiple purification stages, leading to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The starting materials, specifically the Formula IV precursors, are synthesized from commercially available building blocks, ensuring a stable and secure supply chain. The robustness of the reaction conditions, which tolerate various substituents on the aromatic ring, means that supply disruptions for specific niche reagents are less likely to halt production. This reliability is critical for maintaining continuous manufacturing schedules and meeting the demanding delivery timelines of multinational pharmaceutical clients.
- Scalability and Environmental Compliance: The use of standard industrial solvents like 1,4-dioxane and DMF, combined with moderate temperatures, facilitates easy translation from laboratory bench scale to multi-ton commercial production. The process generates minimal hazardous by-products, aligning with increasingly stringent environmental regulations. This compliance reduces the risk of regulatory delays and ensures long-term sustainability of the manufacturing operation, securing the supply of high-purity pharmaceutical intermediates for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from a thorough analysis of the patent's experimental data and comparative advantages over legacy methods. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing production portfolios. The answers provide clarity on yield expectations, safety considerations, and the versatility of the final products.
Q: What are the key advantages of the CuI-catalyzed cyclization method over traditional benzodiazepine synthesis?
A: The patented method utilizes inexpensive cuprous iodide instead of precious metal catalysts, operates under mild reflux conditions in 1,4-dioxane, and achieves high yields (up to 98%) in a single step to construct the core skeleton, significantly reducing raw material costs and processing time compared to multi-step conventional routes.
Q: How does the quaternary ammonium salt intermediate facilitate functionalization?
A: By converting the neutral azetidino-benzodiazepine into a quaternary ammonium salt (Formula II) using methyl triflate, the four-membered azetidine ring becomes highly susceptible to nucleophilic attack. This allows for diverse ring-opening reactions with various nucleophiles like azides, cyanides, and thiolates, enabling rapid access to a wide library of functionalized derivatives.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process demonstrates excellent scalability potential due to the use of robust, commercially available reagents (Cs2CO3, CuI), standard solvents (1,4-dioxane, DCM, DMF), and straightforward workup procedures involving filtration and column chromatography, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Benzodiazepine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic strategies outlined in CN112341468A for the next generation of CNS therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab discovery to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,4-benzodiazepine intermediate adheres to the highest international quality standards. We are committed to delivering excellence in both technical execution and supply chain management.
We invite you to collaborate with us to leverage this advanced chemistry for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your timeline to market with reliable, high-quality chemical solutions.
