Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial API Manufacturing
Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanones for Commercial API Manufacturing
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable pathways. A significant breakthrough in this domain is detailed in patent CN112239436A, which discloses a robust preparation method for 4-acyl-2(5H)-furanone compounds. These heterocyclic scaffolds are not merely academic curiosities; they represent a critical structural backbone found in numerous bioactive molecules and natural products. As illustrated in the provided data, compounds featuring this core structure exhibit potent biological activities, ranging from antimicrobial properties seen in agent A to analgesic effects in agent B, and even antifouling capabilities as demonstrated by maculalactone M. The ability to construct these complex cores efficiently is paramount for drug discovery teams aiming to access novel chemical space.
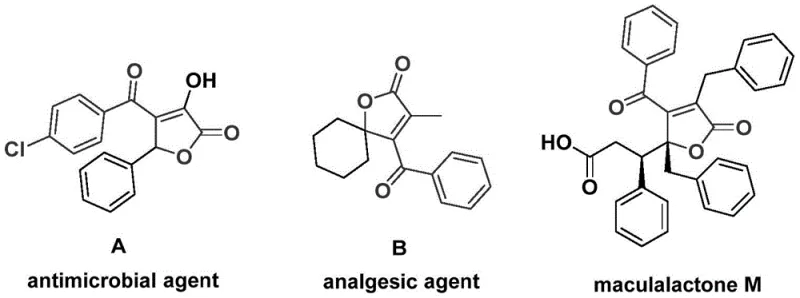
This patent introduces a novel palladium-catalyzed bis-carbonylation strategy that transforms simple starting materials—propiolic alcohols and aryl trifluoromethanesulfonates—into valuable 4-acyl-2(5H)-furanone derivatives. For R&D directors and procurement managers alike, this represents a shift towards high-efficiency manufacturing. The method operates under relatively mild conditions compared to historical precedents, utilizing a catalytic system that ensures high substrate compatibility. By leveraging this technology, manufacturers can potentially reduce the number of synthetic steps required to reach key intermediates, thereby lowering the overall cost of goods sold (COGS) and minimizing waste generation. The strategic importance of such a method cannot be overstated, as it directly impacts the supply chain reliability for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acyl-2(5H)-furanone skeletons has been fraught with challenges that hinder large-scale commercial adoption. Traditional routes often rely on multi-step sequences involving hazardous reagents, extreme temperatures, or expensive stoichiometric oxidants. These conventional methods frequently suffer from poor atom economy, where a significant portion of the starting material ends up as waste rather than incorporated into the final product. Furthermore, the functional group tolerance in older methodologies is often limited, requiring extensive protection and de-protection strategies that add time and cost to the manufacturing process. For supply chain heads, these inefficiencies translate into longer lead times and higher vulnerability to raw material price fluctuations. The reliance on gaseous carbon monoxide in some carbonylation protocols also introduces significant safety hazards and engineering complexities, necessitating specialized high-pressure equipment that many contract manufacturing organizations (CMOs) may lack.
The Novel Approach
In stark contrast, the method disclosed in CN112239436A offers a streamlined, one-step solution that addresses many of these pain points. By employing a palladium-catalyzed bis-carbonylation reaction, the process directly constructs the furanone ring with high precision. The use of aryl trifluoromethanesulfonates (aryl triflates) as electrophiles is particularly advantageous, as these are stable, commercially available, or easily synthesized from phenols. Coupled with propiolic alcohols, which serve as the nucleophilic partners, the reaction achieves excellent convergence. The patent highlights that this approach allows for the efficient and rapid synthesis of the target compounds in a single operational step. This consolidation of steps is a game-changer for process chemistry, as it drastically reduces the solvent usage, energy consumption, and labor hours associated with isolation and purification between steps. The result is a greener, more economical pathway that aligns perfectly with modern principles of sustainable chemical manufacturing.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams looking to optimize the process or adapt it to new substrates. The reaction initiates with the oxidative addition of the palladium catalyst into the aryl triflate bond, generating a reactive aryl-palladium intermediate. This species is the gateway to the subsequent carbonylation events. Uniquely, this protocol utilizes chromium hexacarbonyl (Cr(CO)6) as a solid carbon monoxide surrogate. Upon heating to 100°C in DMF, Cr(CO)6 releases CO in situ, which is then inserted into the aryl-palladium bond to form an acyl-palladium intermediate. This first insertion is critical for establishing the ketone functionality adjacent to the aromatic ring.
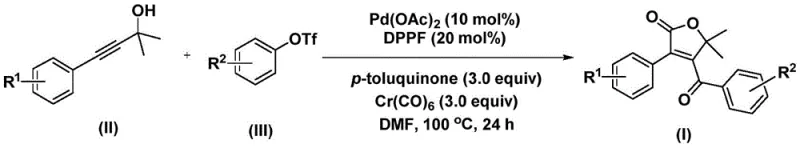
Following the first carbonylation, the propiolic alcohol component enters the catalytic cycle. The acyl-palladium intermediate interacts with the alkyne moiety of the alcohol, leading to the formation of a five-membered ring palladium species. Subsequently, a second molecule of carbon monoxide is inserted into this cyclic intermediate, expanding it to a six-membered ring palladium complex. The final step involves reductive elimination, which releases the desired 4-acyl-2(5H)-furanone product and regenerates the active palladium catalyst. This elegant cascade ensures that two carbonyl groups are installed with high regioselectivity. From an impurity control perspective, the use of DPPF as a ligand and p-toluquinone as an additive helps stabilize the palladium species and suppresses side reactions such as homocoupling of the aryl triflate or polymerization of the alkyne. This mechanistic clarity allows chemists to fine-tune reaction parameters to maximize purity, a key metric for pharmaceutical intermediates.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
The practical execution of this synthesis is designed for reproducibility and scalability. The patent outlines a straightforward procedure where all reagents are combined in a Schlenk tube or reactor. The simplicity of the setup—requiring only standard heating and stirring equipment—makes it accessible for both laboratory-scale optimization and pilot-plant operations. The detailed experimental section provides specific molar ratios, such as the 0.1:0.2:3:3 ratio of Pd(OAc)2 to ligand to Cr(CO)6 to quinone, ensuring that practitioners can replicate the high yields reported. For a comprehensive guide on the standardized operating procedures, please refer to the technical instructions below.
- Combine palladium acetate, DPPF ligand, p-toluquinone, chromium hexacarbonyl, propiolic alcohol, and aryl trifluoromethanesulfonate in DMF solvent.
- Heat the reaction mixture to 100°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this technology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant reduction of manufacturing complexity. By condensing what could be a multi-step synthesis into a single pot reaction, the process inherently lowers the cost of production. There is no need for intermediate isolations, which reduces solvent consumption and waste disposal costs. Furthermore, the starting materials—aryl triflates and propiolic alcohols—are structurally diverse and generally inexpensive, allowing for the cost-effective production of a wide library of analogues. This flexibility is vital for companies needing to pivot quickly between different API candidates during clinical development phases.
- Cost Reduction in Manufacturing: The elimination of gaseous carbon monoxide cylinders and high-pressure reactors in favor of a solid CO source (Cr(CO)6) dramatically lowers capital expenditure and safety compliance costs. Additionally, the high conversion rates observed (with yields reaching up to 95% for certain substrates like compound I-1) mean that less raw material is wasted, directly improving the margin profile for the final intermediate. The use of common solvents like DMF further ensures that supply chains remain robust and unaffected by niche solvent shortages.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against various functional groups (halogens, alkyl, alkoxy) means that supply chains are less fragile. Manufacturers can source a broader range of substituted aryl triflates without fearing reaction failure. This substrate tolerance reduces the risk of batch failures due to minor variations in raw material quality. Moreover, the reaction time of 24 hours is compatible with standard batch processing cycles, facilitating predictable production scheduling and reliable delivery timelines to downstream API manufacturers.
- Scalability and Environmental Compliance: The post-treatment process described is remarkably simple, involving filtration followed by silica gel mixing and column chromatography. While column chromatography is standard in fine chemicals, the initial filtration removes bulk metal salts and insoluble byproducts, easing the load on purification columns. This simplicity translates to easier scale-up potential. From an environmental standpoint, the atom economy of incorporating two CO molecules and the alkyne into the ring structure is superior to many alternative cyclization methods, resulting in a lower E-factor (mass of waste per mass of product) and better alignment with green chemistry initiatives.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation. Understanding these nuances is essential for making informed decisions about process integration and vendor selection.
Q: What are the key advantages of this palladium-catalyzed method over traditional furanone synthesis?
A: This method utilizes readily available aryl triflates and propiolic alcohols, avoiding harsh conditions. It achieves high yields (up to 95%) through a one-step bis-carbonylation process, significantly simplifying the synthetic route compared to multi-step traditional methods.
Q: What is the role of Chromium Hexacarbonyl in this reaction?
A: Chromium hexacarbonyl (Cr(CO)6) serves as a solid carbon monoxide substitute. It releases CO in situ under the reaction conditions (100°C), which is then inserted into the palladium intermediate twice to form the necessary carbonyl groups for the furanone ring construction.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process uses common organic solvents like DMF and standard reagents. The simple post-treatment involving filtration and column chromatography suggests good potential for scale-up, making it suitable for producing complex pharmaceutical intermediates efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
The synthesis of complex heterocycles like 4-acyl-2(5H)-furanones requires a partner with deep technical expertise and a commitment to quality. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced catalytic technologies to deliver high-purity intermediates for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the demanding standards required for GMP API synthesis.
We invite you to explore how our capabilities can enhance your drug development pipeline. Whether you require custom synthesis of specific furanone derivatives or scale-up of the palladium-catalyzed route described in CN112239436A, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your project's unique requirements. Let us be your trusted partner in transforming innovative chemistry into commercial reality.
