Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable 4-Acyl-2(5H)-Furanone Production
Introduction to Next-Generation Furanone Synthesis
The landscape of organic synthesis for bioactive scaffolds is constantly evolving, driven by the need for more efficient and sustainable manufacturing processes. A significant breakthrough in this domain is detailed in patent CN112239436A, which discloses a novel preparation method for 4-acyl-2(5H)-furanone compounds. These heterocyclic structures are not merely academic curiosities; they serve as critical backbones in a wide array of natural products and pharmaceutical agents. As illustrated in the structural diversity of bioactive molecules, this scaffold is prevalent in compounds exhibiting potent antibacterial activity, analgesic properties, and even antifouling capabilities for marine ecosystems. The ability to access these complex architectures efficiently is paramount for drug discovery teams aiming to explore new chemical space.
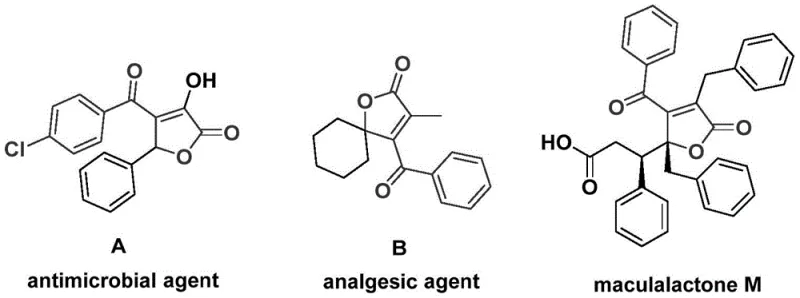
The traditional approaches to constructing such densely functionalized lactones often suffer from low atom economy and tedious multi-step sequences. However, the methodology described in the patent leverages a sophisticated palladium-catalyzed bis-carbonylation strategy. By utilizing readily available starting materials such as propiolic alcohols and aryl trifluoromethanesulfonates, this route offers a direct entry into the target scaffold. For a reliable pharmaceutical intermediate supplier, mastering such transformations is key to providing high-purity building blocks that accelerate the development of next-generation therapeutics. The robustness of this method suggests it can be a cornerstone technology for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acyl-2(5H)-furanones has been fraught with challenges that hinder large-scale production. Conventional routes often rely on the use of gaseous carbon monoxide, which poses significant safety hazards and requires specialized high-pressure equipment that many standard manufacturing facilities lack. Furthermore, existing methods frequently exhibit poor substrate tolerance, meaning that the introduction of diverse functional groups—essential for tuning the biological activity of a drug candidate—can lead to reaction failure or drastically reduced yields. The reliance on harsh reaction conditions or unstable intermediates also complicates the purification process, leading to increased waste generation and higher operational costs. These limitations create a bottleneck in the supply chain, extending lead times for high-purity intermediates and increasing the overall cost of goods sold for downstream API manufacturers.
The Novel Approach
In stark contrast, the innovative process outlined in the patent utilizes a solid carbon monoxide substitute, specifically chromium hexacarbonyl, which releases CO in situ under mild thermal conditions. This eliminates the need for handling toxic CO gas, thereby enhancing workplace safety and simplifying reactor requirements. The reaction proceeds through a palladium-catalyzed cascade involving propiolic alcohol and aryl triflates, as depicted in the general reaction scheme below. This one-pot transformation is remarkably efficient, achieving high conversion rates in polar aprotic solvents like DMF at temperatures between 100°C and 120°C. The operational simplicity allows for a streamlined workflow where the reaction mixture can be directly processed after completion, significantly reducing the time and resources spent on work-up procedures.
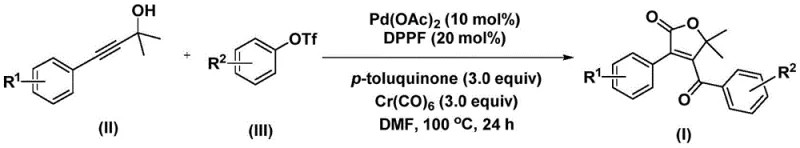
Moreover, this novel approach demonstrates exceptional versatility regarding substrate scope. Whether the aromatic rings bear electron-donating groups like methoxy or electron-withdrawing groups like halogens and trifluoromethyl, the system maintains high reactivity. This broad compatibility is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it allows chemists to access a wide library of analogs from a single, standardized protocol without needing to re-optimize conditions for every new derivative. The combination of safety, efficiency, and versatility makes this method a superior alternative to legacy technologies.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors focused on process optimization and impurity control. The reaction initiates with the oxidative addition of the palladium catalyst into the aryl trifluoromethanesulfonate bond, generating a reactive aryl-palladium intermediate. This species then undergoes the first migratory insertion of carbon monoxide, which is continuously supplied by the decomposition of chromium hexacarbonyl, to form an acyl-palladium complex. This step is critical as it establishes the first carbonyl linkage of the future lactone ring. The presence of the bidentate ligand, 1,1'-bis(diphenylphosphino)ferrocene (DPPP), stabilizes the palladium center throughout this cycle, preventing premature catalyst deactivation and ensuring high turnover numbers.
Subsequently, the acyl-palladium intermediate engages with the propiolic alcohol substrate. This interaction triggers a cyclization event that forms a five-membered ring palladium intermediate, setting the stage for the second carbonyl insertion. A second molecule of carbon monoxide inserts into the palladium-carbon bond, expanding the coordination sphere and forming a six-membered ring intermediate. The cycle concludes with a reductive elimination step that releases the final 4-acyl-2(5H)-furanone product and regenerates the active palladium(0) species. This elegant cascade ensures that the carbon skeleton is constructed with high precision. From an impurity control perspective, the use of methyl p-benzoquinone as an additive plays a vital role in re-oxidizing any inactive palladium species, maintaining the catalytic cycle's integrity and minimizing the formation of side products associated with catalyst death.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocols to maximize yield and purity. The procedure typically involves charging a Schlenk tube with the palladium catalyst, ligand, oxidant, CO source, and substrates in an anhydrous organic solvent. The choice of solvent is critical; N,N-dimethylformamide (DMF) has been identified as the optimal medium due to its ability to dissolve all reagents effectively and stabilize the polar transition states involved in the carbonylation steps. The reaction is heated to 100°C for approximately 24 hours, although this can be extended to 48 hours for sterically hindered substrates to ensure completeness. Following the reaction, the work-up is straightforward, involving filtration to remove metal residues followed by standard chromatographic purification.
- Charge a reaction vessel with palladium acetate, DPPP ligand, methylbenzoquinone, chromium carbonyl, propiolic alcohol, and aryl trifluoromethanesulfonate in DMF solvent.
- Heat the reaction mixture to 100-120°C and maintain stirring for 24 to 48 hours to allow for complete bis-carbonylation and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical elegance. The shift towards this palladium-catalyzed method addresses several critical pain points in the sourcing of complex heterocyclic intermediates. By consolidating what was previously a multi-step synthesis into a single pot, the process drastically reduces the consumption of solvents and reagents, leading to substantial cost savings in raw material procurement. Furthermore, the elimination of gaseous carbon monoxide removes the need for specialized gas handling infrastructure, allowing production to occur in standard multipurpose reactors. This flexibility enhances supply chain reliability, as manufacturing can be distributed across a wider network of qualified contract manufacturing organizations (CMOs) without requiring niche equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by its high atom economy and simplified operational workflow. By avoiding the isolation of unstable intermediates and utilizing a one-pot design, labor costs and processing time are significantly minimized. The use of commercially available starting materials like aryl triflates and propiolic alcohols ensures a stable and competitive pricing structure for inputs. Additionally, the high yields reported (often exceeding 80-90% for various substrates) mean that less raw material is wasted, directly improving the margin profile for the final API. This efficiency allows suppliers to offer more competitive pricing models to their pharmaceutical partners.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis routes. This method mitigates such risks by relying on robust, commodity-grade reagents that are less susceptible to market volatility compared to exotic custom synthons. The tolerance for a wide range of functional groups means that a single manufacturing line can produce a diverse portfolio of furanone derivatives simply by swapping the starting aryl triflate. This modularity allows for rapid response to changing demand patterns, reducing lead times for high-purity intermediates. Suppliers can maintain lower inventory levels of finished goods while still meeting just-in-time delivery requirements, optimizing working capital for both the manufacturer and the buyer.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal intensify, the environmental profile of a synthesis route becomes a key selection criterion. This bis-carbonylation method generates fewer byproducts and avoids the use of hazardous gases, aligning with green chemistry principles. The simplified post-treatment, which primarily involves filtration and chromatography, reduces the volume of aqueous waste streams typically associated with extensive extraction and washing steps. This ease of waste management facilitates smoother regulatory approvals and lowers the cost of environmental compliance. Consequently, scaling this process from kilogram to tonne quantities is technically feasible and environmentally sustainable, ensuring long-term viability for commercial production.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries based on the patent data. These questions address the practical aspects of implementation, safety considerations, and the versatility of the chemical platform. Understanding these details helps stakeholders make informed decisions regarding the integration of this technology into their supply chains.
Q: What are the primary advantages of this Pd-catalyzed method over traditional furanone synthesis?
A: This method utilizes a one-pot bis-carbonylation strategy that eliminates the need for multiple synthetic steps and harsh conditions often required in conventional routes, resulting in higher overall efficiency and simpler post-treatment.
Q: What is the role of Chromium Hexacarbonyl in this reaction system?
A: Chromium hexacarbonyl serves as a safe and effective solid carbon monoxide substitute, releasing CO in situ to facilitate the double carbonyl insertion required to build the furanone skeleton without handling toxic CO gas.
Q: Is this process scalable for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the process demonstrates excellent substrate compatibility and uses commercially available reagents like palladium acetate and aryl triflates, making it highly suitable for scale-up from laboratory to commercial production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the Pd-catalyzed bis-carbonylation described in CN112239436A. Our team of expert process chemists is dedicated to translating such cutting-edge academic and patent literature into robust, GMP-compliant manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to full-scale market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-acyl-2(5H)-furanone intermediate delivered meets the highest industry standards.
We invite you to collaborate with us to leverage this efficient synthesis route for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data for our existing catalog or to discuss route feasibility assessments for your custom targets. Let us help you secure a reliable supply of high-quality intermediates while optimizing your overall manufacturing costs.
