Scalable Production of Bioactive 4-Acyl-2(5H)-Furanones via Novel Palladium-Catalyzed Bis-Carbonylation
Scalable Production of Bioactive 4-Acyl-2(5H)-Furanones via Novel Palladium-Catalyzed Bis-Carbonylation
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to access complex heterocyclic scaffolds that serve as core structures for bioactive molecules. A recent technological breakthrough detailed in patent CN112239436A introduces a highly effective preparation method for 4-acyl-2(5H)-furanone compounds. This specific structural backbone is of immense value, as evidenced by its presence in diverse natural products and therapeutic agents, including potent antimicrobial agents, analgesics, and antifouling compounds like maculalactone M. The ability to synthesize these motifs efficiently opens new doors for drug discovery and process chemistry.
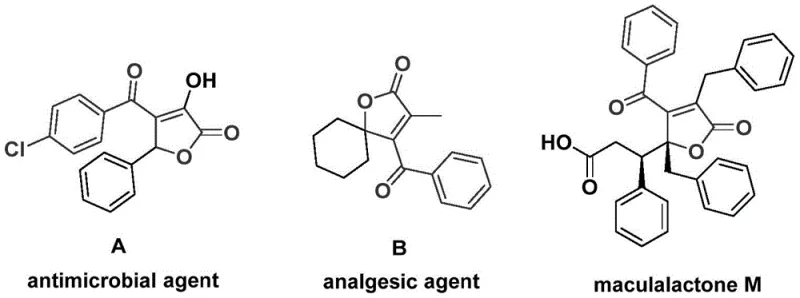
The innovation lies in a palladium-catalyzed bis-carbonylation strategy that utilizes propiolic alcohol and aryl trifluoromethanesulfonate as starting materials. By employing chromium hexacarbonyl as a solid carbon monoxide substitute, the method circumvents the severe safety risks associated with traditional high-pressure CO gas reactions. This approach not only simplifies the operational procedure but also ensures high reaction efficiency and excellent substrate compatibility, making it a superior choice for reliable pharmaceutical intermediate supplier networks aiming to optimize their synthetic portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-containing heterocycles has relied heavily on direct carbonylation reactions using carbon monoxide gas. While chemically effective, this traditional approach presents significant logistical and safety hurdles for large-scale manufacturing. Handling toxic, flammable CO gas requires specialized high-pressure autoclaves and rigorous safety protocols, which drastically increases capital expenditure and operational complexity. Furthermore, controlling the stoichiometry of gaseous reagents can be challenging, often leading to inconsistent reaction rates and difficulties in maintaining uniform mixing throughout the reactor volume. These factors collectively contribute to higher production costs and extended lead times for high-purity intermediates, creating bottlenecks in the supply chain for critical drug substances.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by replacing gaseous CO with a solid surrogate, chromium hexacarbonyl (Cr(CO)₆). This shift allows the reaction to proceed under atmospheric pressure conditions in standard glassware or stainless steel vessels, eliminating the need for expensive high-pressure equipment. The reaction design facilitates a tandem bis-carbonylation and cyclization sequence in a single pot, directly converting simple alkynes and aryl triflates into the complex furanone core. This one-step convergence significantly reduces the number of unit operations, minimizes solvent usage, and streamlines the purification process, thereby achieving substantial cost savings in fine chemical manufacturing while maintaining high purity standards required for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The reaction mechanism involves a sophisticated catalytic cycle driven by palladium species generated in situ from palladium acetate and the dppf ligand. Initially, the oxidative addition of the aryl trifluoromethanesulfonate to the active Pd(0) species forms an aryl-palladium intermediate. Subsequently, carbon monoxide, which is thermally liberated from the chromium hexacarbonyl additive, inserts into the palladium-carbon bond to generate an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propiolic alcohol substrate, initiating the ring-closing process to form a five-membered palladacycle.
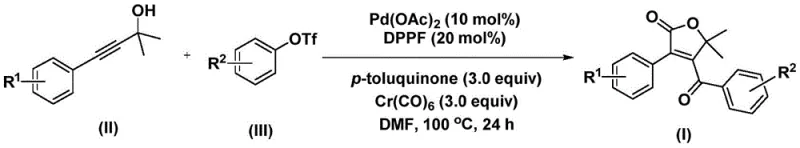
Following the initial cyclization, a second molecule of carbon monoxide inserts into the organopalladium bond, expanding the coordination sphere before a final reductive elimination step releases the 4-acyl-2(5H)-furanone product and regenerates the active catalyst. The presence of methylbenzoquinone as an additive plays a crucial role in re-oxidizing any reduced palladium species or stabilizing the catalytic cycle, ensuring high turnover numbers. This mechanistic pathway explains the broad substrate tolerance observed, as the electronic properties of the aryl ring (R¹ and R² groups) can be modulated without disrupting the fundamental catalytic steps, allowing for the synthesis of diverse derivatives such as those shown in the patent examples.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential. The protocol typically involves charging a reaction vessel with palladium acetate (10 mol%), DPPP ligand (20 mol%), methylbenzoquinone (3.0 equiv), and chromium hexacarbonyl (3.0 equiv) in an anhydrous organic solvent such as DMF. The substrates, specifically the substituted propiolic alcohol and aryl triflate, are then added, and the mixture is heated to temperatures between 100°C and 120°C. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with palladium acetate catalyst, DPPP ligand, methylbenzoquinone additive, and chromium hexacarbonyl in DMF solvent.
- Add propiolic alcohol and aryl trifluoromethanesulfonate substrates to the mixture under inert atmosphere.
- Heat the reaction mixture to 100-120°C for 24-48 hours, then filter and purify via column chromatography to isolate the target furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this solid-state carbonylation method represents a strategic opportunity to de-risk the supply of critical intermediates. By removing the dependency on gaseous carbon monoxide infrastructure, manufacturers can operate in a wider range of facilities, increasing the overall resilience of the supply network. The use of commercially available and inexpensive starting materials, such as aryl triflates and simple alkynols, ensures that raw material costs remain stable and predictable, shielding the project from volatile commodity price fluctuations often seen with specialized gaseous reagents.
- Cost Reduction in Manufacturing: The elimination of high-pressure gas handling equipment results in significant capital expenditure savings, as standard atmospheric reactors can be utilized instead of costly autoclaves. Additionally, the simplified work-up procedure, which involves basic filtration and column chromatography, reduces labor hours and solvent consumption compared to multi-step traditional routes. The high atom economy of the bis-carbonylation process further contributes to lower waste disposal costs, enhancing the overall economic viability of the production process.
- Enhanced Supply Chain Reliability: Utilizing solid reagents like chromium hexacarbonyl simplifies logistics and storage requirements, as these materials do not require specialized pressurized cylinders or hazardous gas transport permits. This ease of handling translates to faster procurement cycles and reduced administrative burdens for the supply chain team. Furthermore, the robust nature of the reaction conditions allows for consistent batch-to-batch reproducibility, ensuring a steady flow of high-quality intermediates to downstream formulation units without unexpected delays.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with various functional groups including halogens, alkoxy, and alkyl substituents. The ability to run the reaction in polar aprotic solvents like DMF, which are common in industrial settings, facilitates easy technology transfer from lab to pilot plant. Moreover, the containment of carbon monoxide within a solid matrix minimizes the risk of atmospheric emissions, aiding in compliance with increasingly stringent environmental regulations regarding volatile organic compounds and toxic gas releases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this technology for their specific pipeline projects. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation.
Q: What is the primary safety advantage of this synthesis method?
A: Unlike traditional carbonylation reactions that require handling toxic and flammable carbon monoxide gas under high pressure, this method utilizes chromium hexacarbonyl as a solid, safe CO surrogate, significantly reducing operational hazards.
Q: What is the typical yield range for this reaction?
A: According to the patent data, the reaction demonstrates high efficiency with isolated yields ranging from 70% to 95% across various substituted substrates, indicating robust substrate compatibility.
Q: Can this process be scaled for industrial production?
A: Yes, the use of standard organic solvents like DMF, commercially available catalysts, and the elimination of high-pressure gas equipment makes this protocol highly amenable to commercial scale-up in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing complex heterocyclic building blocks for modern drug development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-acyl-2(5H)-furanone intermediate meets the highest international standards for pharmaceutical use.
We invite you to contact our technical procurement team to discuss how we can support your specific synthesis requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall project costs. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your timeline from discovery to market.
