Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access bioactive heterocyclic scaffolds, particularly those containing fluorine motifs which enhance metabolic stability and lipophilicity. Patent CN112480015B discloses a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a core structure found in numerous therapeutic agents ranging from antifungals to anticancer drugs. This technology represents a significant leap forward in process chemistry, replacing hazardous high-pressure carbonylation protocols with a safer, palladium-catalyzed cascade reaction that utilizes inexpensive nitro compounds as starting materials. By integrating the reduction of nitro groups and subsequent cyclization into a single operational step, this innovation drastically simplifies the manufacturing workflow for high-purity pharmaceutical intermediates.
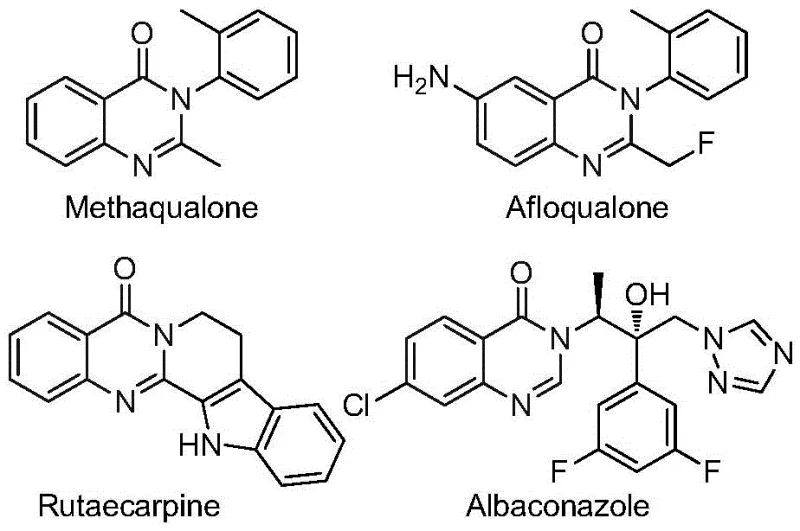
Quinazolinone derivatives are ubiquitous in medicinal chemistry, serving as the backbone for well-known drugs such as Methaqualone and Afloqualone, as illustrated in the structural diversity of bioactive molecules. The introduction of a trifluoromethyl group at the 2-position further augments the pharmacological profile of these compounds, making them highly desirable targets for drug discovery programs. However, traditional synthetic routes often suffer from severe limitations, including the requirement for toxic carbon monoxide gas under high pressure or the use of costly, pre-functionalized precursors that necessitate multiple synthetic steps. The methodology described in CN112480015B addresses these critical pain points by employing a transition metal-catalyzed carbonylation strategy that is both operationally simple and economically viable for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone ring system has relied on methodologies that pose significant safety and economic challenges for industrial application. Conventional approaches frequently involve ruthenium or platinum-catalyzed reductive N-heterocyclization of nitro-substituted benzamides, which strictly require high-pressure carbon monoxide atmospheres, necessitating specialized and expensive reactor equipment. Alternative strategies utilizing iron-catalyzed condensation or palladium-catalyzed cyclization of 2-bromoformylaniline often demand expensive substrates that require prior activation, thereby increasing the overall step count and material costs. Furthermore, these legacy methods typically exhibit narrow substrate scope and lower reaction efficiencies, leading to inconsistent yields and complicated purification processes that hinder the reliable supply of complex polymer additives or electronic chemical precursors derived from similar heterocyclic cores.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed carbonylation cascade reaction that merges multiple transformation steps into a single vessel. By employing trifluoroethylimidoyl chloride and nitro compounds as the primary building blocks, this method bypasses the need for hazardous gaseous CO and expensive pre-activated halides. The reaction proceeds smoothly in common organic solvents like 1,4-dioxane at a moderate temperature of 120°C, utilizing Mo(CO)6 as a convenient solid carbon monoxide surrogate. This strategic shift not only enhances operational safety by eliminating high-pressure gas handling but also significantly broadens the functional group tolerance, allowing for the synthesis of diverse derivatives essential for modern drug development pipelines.
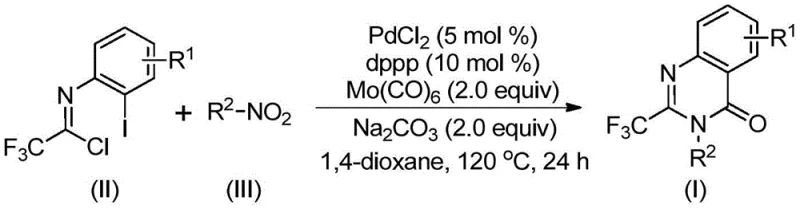
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a sophisticated interplay of reduction, coupling, and cyclization events orchestrated by the palladium catalyst system. The reaction is initiated by the reduction of the nitro compound to the corresponding amine species, facilitated by the decomposition of molybdenum hexacarbonyl under thermal conditions. Subsequently, a base-promoted intermolecular carbon-nitrogen bond coupling occurs between the generated amine and the trifluoroethylimidoyl chloride, yielding a trifluoroacetamidine intermediate. The palladium catalyst then inserts into the carbon-iodine bond of the aromatic ring to form a divalent palladium species, which undergoes CO insertion from the released molybdenum carbonyl to generate an acyl-palladium intermediate. This intricate sequence culminates in the formation of a seven-membered cyclic palladium intermediate, followed by reductive elimination to deliver the final 2-trifluoromethyl substituted quinazolinone product with high regioselectivity.
Understanding the impurity profile is critical for R&D directors focused on purity specifications. The use of a robust ligand system, specifically 1,3-bis(diphenylphosphino)propane (dppp), ensures high catalytic turnover and minimizes side reactions such as homocoupling or incomplete cyclization. The reaction conditions are optimized to favor the desired intramolecular cyclization over intermolecular polymerization, resulting in a clean crude reaction mixture. This high level of chemoselectivity reduces the burden on downstream purification, allowing for the isolation of high-purity OLED material precursors or agrochemical intermediates with minimal chromatographic effort. The compatibility with various substituents on both the nitro compound and the imidoyl chloride further demonstrates the versatility of this catalytic cycle.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory scale-up and commercial manufacturing. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and the two primary organic substrates in a suitable solvent. The mixture is then heated to reflux, allowing the cascade reaction to proceed to completion over a defined period. This streamlined process eliminates the need for intermediate isolation steps, thereby reducing solvent consumption and waste generation. For detailed procedural specifics regarding reagent ratios and workup procedures, please refer to the standardized synthesis guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. By shifting from high-pressure gas-dependent processes to a solution-phase reaction using solid reagents, manufacturers can utilize standard glass-lined or stainless steel reactors without the need for specialized autoclaves rated for high-pressure CO. This capital expenditure avoidance translates into significant cost savings and allows for faster deployment of production capacity. Furthermore, the reliance on nitro compounds, which are commodity chemicals available in vast quantities globally, ensures a stable and continuous supply of raw materials, mitigating the risks associated with sourcing specialized or custom-synthesized precursors.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure and the use of inexpensive, commercially available nitro compounds drastically reduce the overall cost of goods sold. The one-pot nature of the reaction minimizes unit operations, reducing labor costs, energy consumption for heating and cooling between steps, and solvent usage for intermediate washes. Additionally, the high reaction efficiency and yield reported in the patent examples mean less raw material is wasted, further optimizing the economic viability of producing these complex heterocycles for the pharmaceutical market.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials like nitrobenzenes and trifluoroethylimidoyl chloride decouples the supply chain from niche chemical suppliers who might face production bottlenecks. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that variations in raw material quality are less likely to cause batch failures. This reliability is crucial for maintaining consistent lead times for high-purity pharmaceutical intermediates, ensuring that downstream drug formulation schedules are not disrupted by upstream synthesis delays.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from milligram to gram scales with the potential for tonnage production. The absence of toxic gaseous reagents simplifies environmental, health, and safety (EHS) compliance, reducing the regulatory burden and permitting timelines associated with handling hazardous gases. Moreover, the simplified workup procedure involving filtration and chromatography generates less hazardous waste compared to multi-step sequences, aligning with green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, safety, and scalability for potential partners and licensees.
Q: What are the key advantages of this one-pot synthesis method over traditional routes?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates like 2-bromoformylaniline. It utilizes cheap and readily available nitro compounds and trifluoroethylimidoyl chloride, operating under standard atmospheric pressure with high substrate compatibility.
Q: What is the role of Molybdenum Hexacarbonyl (Mo(CO)6) in this reaction?
A: Mo(CO)6 serves a dual function: it acts as a solid carbon monoxide substitute that releases CO in situ under heating conditions for the carbonylation step, and it facilitates the initial reduction of the nitro group to an amine, streamlining the multi-step transformation into a single pot.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states the method can be expanded to the gram level and is suitable for industrial production. The use of stable solid reagents instead of toxic gases and the robustness of the palladium catalyst system support scalable manufacturing of complex heterocycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN112480015B for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee the quality of every batch of 2-trifluoromethyl quinazolinone intermediates we produce.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce time-to-market for your critical pharmaceutical assets.
