Revolutionizing Quinazolinone Production: A Cost-Effective Iron-Catalyzed Strategy for Global Pharma Supply Chains
Revolutionizing Quinazolinone Production: A Cost-Effective Iron-Catalyzed Strategy for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and economically viable synthetic routes for heterocyclic scaffolds that serve as the backbone of modern therapeutics. Patent CN111675662B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in the manufacturing of these high-value intermediates. Quinazolinones are privileged structures found in numerous bioactive molecules, ranging from anticancer agents to antifungal drugs, yet their synthesis has historically been plagued by harsh conditions and expensive reagents. This new methodology leverages a cheap iron catalyst to drive the cyclization of readily available trifluoroethylimidoyl chloride and isatin derivatives, marking a significant departure from traditional precious metal-catalyzed processes. By shifting the paradigm towards base-metal catalysis, this technology not only enhances the economic feasibility of production but also aligns with the growing global demand for greener, more sustainable chemical manufacturing practices.
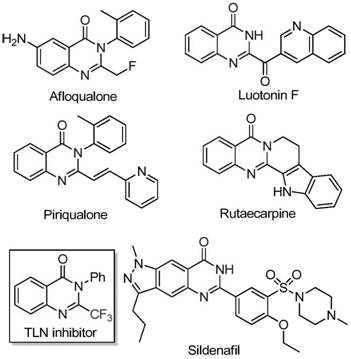
The strategic importance of the trifluoromethyl group in medicinal chemistry cannot be overstated, as its introduction often dramatically improves the metabolic stability, lipophilicity, and bioavailability of drug candidates. However, incorporating this moiety into the quinazolinone core has traditionally required specialized fluorinating agents or multi-step sequences that degrade overall process efficiency. The disclosed invention circumvents these challenges by utilizing a direct cyclization approach that tolerates a wide array of functional groups. For R&D directors evaluating new process technologies, this represents a compelling opportunity to streamline the synthesis of complex nitrogen-containing fused ring systems. The ability to access these structures through a simplified workflow reduces the cumulative time and resource investment typically associated with late-stage functionalization, thereby accelerating the timeline from discovery to clinical supply.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons such as anthranilamide, anthranilic acid, or isatoic anhydride with trifluoroacetic anhydride or ethyl trifluoroacetate. While chemically feasible, these conventional pathways suffer from severe limitations that hinder their application in large-scale industrial settings. The reaction conditions are often excessively harsh, requiring strong acids or bases and elevated temperatures that can compromise sensitive functional groups on the substrate. Furthermore, the starting materials, particularly specialized trifluoromethyl synthons, are frequently expensive and subject to supply chain volatility, creating significant cost uncertainty for procurement managers. Low yields and narrow substrate scope further exacerbate the problem, necessitating extensive purification steps that generate substantial chemical waste and reduce the overall atom economy of the process. These factors collectively contribute to a high cost of goods sold (COGS) and extended lead times, making traditional methods less attractive for commercial manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN111675662B utilizes a synergistic combination of ferric chloride and sodium hydride to catalyze the reaction between trifluoroethylimidoyl chloride and isatin. This method operates under relatively mild conditions, initially at 40°C and subsequently at 120°C in DMF, avoiding the extreme environments that often degrade product quality. The use of inexpensive and abundant iron salts replaces costly transition metals, fundamentally altering the cost structure of the synthesis. Moreover, the reaction demonstrates exceptional functional group tolerance, accommodating substituents such as halogens, alkyl groups, and nitro groups without the need for protecting group strategies. This robustness allows for the direct synthesis of diverse quinazolinone derivatives from commercially available building blocks, significantly simplifying the supply chain logistics. The inclusion of 4A molecular sieves further enhances the reaction efficiency by sequestering water, driving the equilibrium towards product formation and minimizing hydrolysis side reactions.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the iron catalyst and the basic promoter to facilitate carbon-nitrogen bond formation and subsequent ring closure. Initially, the sodium hydride promotes the nucleophilic attack of the isatin nitrogen onto the imidoyl chloride, forming a trifluoroacetamidine intermediate. This step is crucial for establishing the connectivity required for the fused ring system. Following this, the ferric chloride catalyst mediates a decarbonylation and cyclization sequence that rearranges the intermediate into the thermodynamically stable quinazolinone core. The iron center likely coordinates with the carbonyl oxygen and the nitrogen atoms, lowering the activation energy for the decarbonylation step which is often the rate-determining step in similar transformations. This catalytic cycle is highly efficient, allowing the reaction to proceed with only 20 mol% catalyst loading, which is a testament to the turnover number of the iron species in this specific chemical environment.
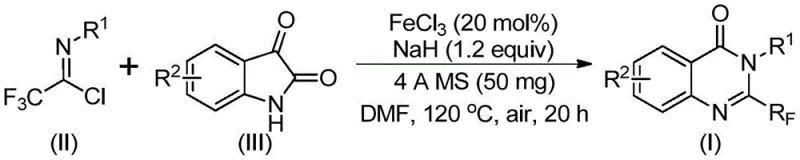
From an impurity control perspective, the mechanism offers distinct advantages over acid-catalyzed alternatives. The basic conditions provided by NaH prevent the formation of acid-sensitive byproducts that often complicate the purification of quinazolinones. Additionally, the specific reactivity of the imidoyl chloride ensures that the trifluoromethyl group is incorporated regioselectively at the 2-position of the quinazolinone ring, eliminating the formation of regioisomers that are difficult to separate. The presence of molecular sieves plays a dual role: not only do they remove water generated during the reaction, but they also adsorb polar impurities, resulting in a cleaner crude reaction mixture. This inherent cleanliness of the reaction profile reduces the burden on downstream processing units, such as crystallization or chromatography, thereby improving the overall throughput of the manufacturing plant. For quality assurance teams, this translates to a more consistent impurity profile and easier validation of the cleaning procedures.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for ease of operation, making it highly accessible for process chemistry teams aiming to scale up production. The protocol involves a straightforward one-pot procedure where all reagents are combined in a polar aprotic solvent, typically DMF, which effectively dissolves both the organic substrates and the inorganic catalyst. The reaction is initiated at a lower temperature of 40°C for roughly 10 hours to allow for the initial coupling, followed by a heating phase at 120°C for 20 hours to drive the cyclization to completion. This two-stage temperature profile optimizes the reaction kinetics, preventing premature decomposition while ensuring full conversion of the starting materials. Upon completion, the workup is remarkably simple, involving filtration to remove the molecular sieves and inorganic salts, followed by standard silica gel column chromatography to isolate the pure product. The detailed standardized synthesis steps for replicating this high-yielding process are outlined in the guide below.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent within a reaction vessel.
- Initiate the reaction by stirring at 40°C for approximately 10 hours to facilitate initial bond formation, then increase temperature to 120°C.
- Maintain heating at 120°C for an additional 20 hours under air atmosphere to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost stability and operational resilience. The primary driver of cost reduction lies in the substitution of expensive precious metal catalysts with commodity-grade ferric chloride, which is available in bulk quantities at a fraction of the price. This switch eliminates the need for complex and costly heavy metal removal steps, such as scavenger resin treatments, which are mandatory when using palladium or platinum catalysts to meet strict regulatory limits for residual metals in APIs. Consequently, the overall manufacturing cost is significantly reduced, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients. Furthermore, the reliance on widely available starting materials like isatin and aromatic amines mitigates the risk of supply disruptions, ensuring a continuous flow of intermediates even during periods of market volatility.
- Cost Reduction in Manufacturing: The elimination of precious metals and the use of inexpensive iron salts drastically lower the direct material costs associated with the catalytic system. Additionally, the high atom economy of the reaction minimizes waste generation, reducing the expenses related to waste disposal and environmental compliance. The simplified workup procedure, which avoids complex extraction or distillation steps, further contributes to operational savings by reducing labor and utility consumption. These cumulative efficiencies result in a leaner manufacturing process that maximizes profit margins while maintaining high product quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including isatin derivatives and trifluoroethylimidoyl chlorides, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. This diversification of the supply base enhances the resilience of the supply chain against geopolitical or logistical shocks. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, allowing for greater flexibility in sourcing. This reliability ensures consistent delivery schedules and helps maintain safety stock levels without the fear of batch failures due to reagent incompatibility.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work efficiently from gram to multi-gram scales in the patent examples, with clear pathways to kilogram and tonne-level production. The use of DMF as a solvent, while requiring careful handling, is well-established in industrial settings with existing recovery and recycling infrastructure. The absence of toxic heavy metals simplifies the environmental permitting process and reduces the regulatory burden associated with effluent treatment. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios. The responses cover catalyst performance, substrate compatibility, and purification strategies, providing a comprehensive overview of the process capabilities.
Q: What are the primary advantages of using ferric chloride over traditional catalysts for quinazolinone synthesis?
A: Ferric chloride offers a drastic reduction in catalyst cost compared to precious metals like palladium or rhodium. Furthermore, it demonstrates excellent functional group tolerance and operates effectively under aerobic conditions, simplifying the operational requirements for large-scale manufacturing.
Q: How does this method improve the impurity profile of the final API intermediate?
A: The use of 4A molecular sieves in the reaction mixture effectively scavenges moisture and byproducts, leading to a cleaner reaction profile. The specific decarbonylation mechanism minimizes side reactions common in traditional condensation methods, resulting in higher purity crude products that require less intensive downstream purification.
Q: Is this synthetic route suitable for diverse substrate scopes beyond the examples provided?
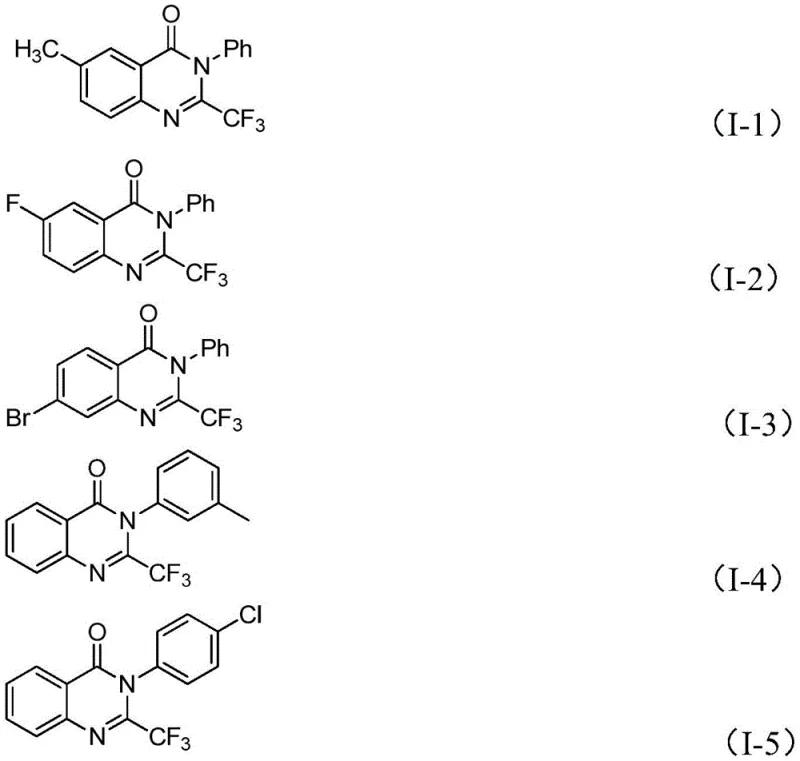
A: Yes, the patent data indicates broad substrate tolerance. The method successfully accommodates various substituents on both the aryl ring of the imidoyl chloride and the isatin core, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups like halogens and nitro groups, ensuring versatility for medicinal chemistry campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone meets the highest industry standards. Our commitment to quality and efficiency makes us the ideal partner for bringing your next-generation therapeutics to market.
We invite you to collaborate with us to leverage this cost-effective synthesis for your specific drug development programs. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your project's unique requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can optimize your supply chain and accelerate your time to market.
