Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Manufacturing
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Manufacturing
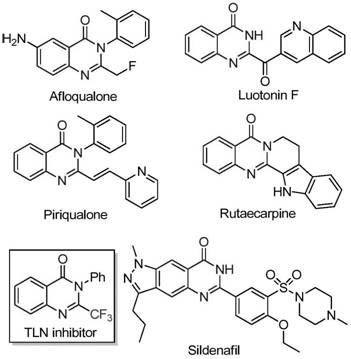
The rapid evolution of medicinal chemistry demands efficient access to fluorinated heterocycles, particularly those containing the quinazolinone scaffold which is prevalent in numerous bioactive natural products and synthetic drugs. Patent CN111675662B introduces a transformative preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current synthetic methodologies. This technology leverages a robust iron-catalyzed cyclization strategy that merges readily available trifluoroethylimidoyl chlorides with isatin derivatives. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates. The introduction of the trifluoromethyl group is known to significantly enhance the metabolic stability, lipophilicity, and bioavailability of drug candidates, making this specific scaffold highly desirable for oncology and anti-inflammatory drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with anthranilamides or isatoic anhydrides. These traditional pathways are frequently plagued by severe reaction conditions that require strict anhydrous environments or cryogenic temperatures, leading to increased operational complexity and energy consumption. Furthermore, the substrates often suffer from limited commercial availability or high costs, creating supply chain vulnerabilities for large-scale production. The narrow substrate scope of these older methods often fails to tolerate diverse functional groups, restricting the chemical space available for medicinal chemists to explore structure-activity relationships. Additionally, low yields and difficult purification processes associated with these conventional routes result in substantial material loss and increased waste generation, negatively impacting both the economic viability and the environmental footprint of the manufacturing process.
The Novel Approach
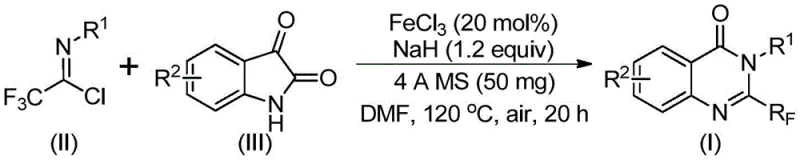
In stark contrast, the methodology disclosed in the patent utilizes a novel combination of trifluoroethylimidoyl chloride and isatin derivatives catalyzed by inexpensive ferric chloride. This approach fundamentally simplifies the synthetic route by employing a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. The reaction proceeds efficiently in polar aprotic solvents like DMF under aerobic conditions, eliminating the need for expensive inert gas protection systems. This new strategy not only broadens the substrate scope to include various electron-donating and electron-withdrawing groups but also achieves superior yields ranging significantly higher than many traditional methods. By utilizing cheap and stable starting materials, this method drastically reduces the raw material costs and simplifies the logistics of sourcing precursors for commercial scale-up.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological breakthrough lies in the dual-role mechanism facilitated by the iron catalyst and the base. Initially, sodium hydride promotes the nucleophilic attack of the isatin nitrogen on the imidoyl chloride, forming a transient trifluoroacetamidine intermediate. This step is critical for establishing the necessary carbon-nitrogen framework. Subsequently, the ferric chloride catalyst activates the system for a decarbonylation event, which is the rate-determining step that drives the aromatization of the quinazolinone ring. The iron center likely coordinates with the carbonyl oxygen, facilitating the extrusion of carbon monoxide and enabling the intramolecular cyclization to close the six-membered heterocyclic ring. This mechanistic pathway is highly efficient because it avoids the formation of stable byproducts that typically hinder reaction progress in non-catalytic thermal cyclizations.
From an impurity control perspective, this mechanism offers distinct advantages. The use of 4A molecular sieves in the reaction mixture plays a crucial role in scavenging trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material or deactivate the catalyst. The high functional group tolerance observed in the experimental data suggests that the catalytic cycle is robust against competitive side reactions such as halogenation or over-oxidation. The specificity of the decarbonylation ensures that the trifluoromethyl group remains intact at the 2-position, preventing the formation of defluorinated impurities that are common in harsher fluorination protocols. This level of control is essential for meeting the stringent purity specifications required for pharmaceutical grade intermediates, reducing the burden on downstream purification steps.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The standardized protocol derived from the patent data outlines a straightforward procedure accessible to most synthetic laboratories. The process begins with the precise weighing of ferric chloride and sodium hydride, followed by the addition of the organic substrates in dry DMF. The reaction profile involves a two-stage temperature program, starting at a mild 40°C to allow for initial coupling, followed by heating to 120°C to drive the cyclization to completion. Detailed operational parameters, including stoichiometry and work-up procedures, are critical for reproducing the high yields reported in the patent examples. For a comprehensive understanding of the exact molar ratios and specific reaction times optimized for different substrates, please refer to the technical guide below.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in anhydrous DMF solvent.
- Stir the reaction mixture at 40°C for 8-10 hours to initiate the alkali-promoted coupling, then heat to 120°C under air atmosphere for 18-20 hours.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed synthesis route presents a compelling value proposition centered on cost stability and operational resilience. The shift from precious metal catalysts or expensive activated reagents to commodity chemicals like ferric chloride and isatins fundamentally alters the cost structure of the intermediate. This transition mitigates the risk of price volatility associated with rare earth metals or specialized fluorinating agents, ensuring more predictable budgeting for long-term projects. Furthermore, the simplicity of the reaction setup, which does not require specialized high-pressure equipment or glovebox techniques, allows for manufacturing in standard multipurpose reactors, thereby maximizing facility utilization rates.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts and the use of widely available starting materials lead to a substantial decrease in the Bill of Materials (BOM) cost. By avoiding complex protection-deprotection sequences often required in alternative syntheses, the overall number of unit operations is reduced, which directly correlates to lower labor and utility costs per kilogram of product. The high atom economy of the decarbonylation-cyclization cascade minimizes waste disposal fees, contributing to a leaner and more profitable manufacturing process without compromising on yield or quality.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved as the key building blocks, such as substituted isatins and aromatic amines (precursors to imidoyl chlorides), are produced by multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks that can halt production lines. The robustness of the reaction conditions, specifically its tolerance to air and moisture when managed correctly, means that the process is less susceptible to minor variations in plant conditions, ensuring consistent batch-to-batch quality and reliable delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from milligram to gram scales with potential for tonnage production. The use of iron, a non-toxic and environmentally benign metal, aligns with green chemistry principles and simplifies regulatory compliance regarding heavy metal residues in the final product. This reduces the need for extensive metal scavenging steps, shortening the production cycle time. Additionally, the simplified work-up procedure involving filtration and standard chromatography facilitates easier technology transfer to CDMO partners, accelerating the timeline from clinical trial material to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the method for industrial partners. Understanding these nuances is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the key advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of Ferric Chloride (FeCl3) offers significant economic and operational benefits compared to precious metal catalysts. It is inexpensive, commercially abundant, and operates effectively under aerobic conditions, eliminating the need for costly inert gas setups while maintaining high functional group tolerance.
Q: Can this synthesis method accommodate diverse substrate substitutions?
A: Yes, the protocol demonstrates excellent substrate scope. It tolerates various substituents on both the isatin ring (such as methyl, fluoro, bromo, and methoxy groups) and the imidoyl chloride aryl ring, allowing for the modular synthesis of a wide library of bioactive analogues.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The method utilizes cheap, readily available starting materials like isatins and avoids sensitive reagents. The reaction conditions are robust (open to air), and the work-up involves standard filtration and chromatography, making it highly amenable to scale-up from gram to multi-kilogram levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient fluorination technologies in modern drug discovery. Our team of expert chemists has thoroughly analyzed the potential of this iron-catalyzed route and is fully prepared to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthesis method for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
