Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
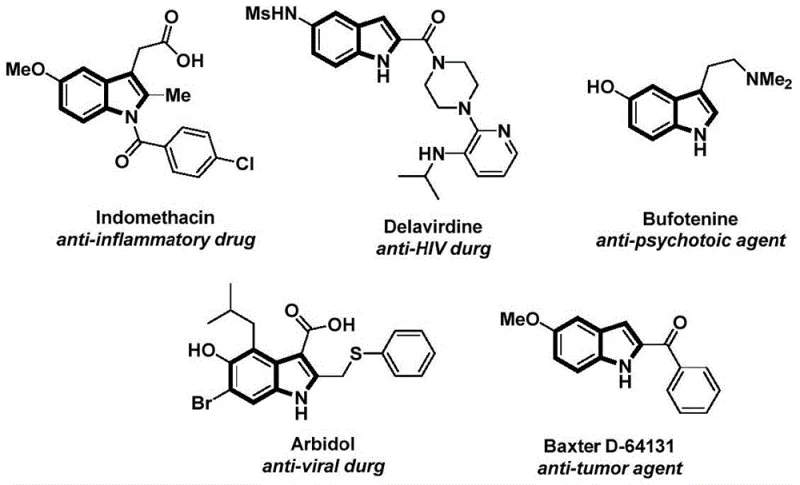
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to anti-HIV drugs such as Delavirdine. As illustrated in the diverse pharmacological landscape shown below, the ability to efficiently functionalize the indole nitrogen is critical for drug discovery and process development. Patent CN112898192B introduces a groundbreaking preparation method for N-acyl indole compounds that addresses long-standing challenges in synthetic efficiency and operational safety. This technology leverages a sophisticated palladium-catalyzed carbonylation cyclization strategy, transforming simple 2-alkynyl anilines and aryl iodides into complex N-acyl indoles in a streamlined manner.
For research directors and process chemists, the significance of this patent lies in its departure from traditional, multi-step acylation protocols. By utilizing a carbon monoxide substitute (TFBen) instead of toxic gaseous CO, the method significantly enhances laboratory safety while maintaining high atom economy. The reaction proceeds through a tandem sequence involving oxidative addition, CO insertion, and subsequent cyclization, all facilitated by a robust palladium catalyst system. This one-pot approach not only reduces the number of isolation steps but also minimizes waste generation, aligning perfectly with modern green chemistry principles required by top-tier pharmaceutical manufacturers seeking reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles has relied on classical acylation reactions using acid chlorides or anhydrides, often requiring harsh conditions and generating stoichiometric amounts of acidic waste. Traditional methods frequently suffer from poor regioselectivity, necessitating tedious purification processes to separate N-acylated products from C-acylated byproducts. Furthermore, direct carbonylation strategies using gaseous carbon monoxide, while effective, pose severe safety hazards due to the toxicity and flammability of CO gas, requiring specialized high-pressure equipment that is not universally available in standard R&D facilities. These limitations result in increased operational costs, extended lead times for high-purity pharmaceutical intermediates, and significant barriers to scaling up production for commercial applications.
The Novel Approach
The methodology disclosed in patent CN112898192B revolutionizes this landscape by employing phenyl 1,3,5-tricarboxylate (TFBen) as a safe and solid carbon monoxide surrogate. This innovation eliminates the need for handling dangerous gases, allowing the reaction to proceed under atmospheric pressure in standard glassware. The process integrates the formation of the amide bond and the construction of the indole ring into a single operational sequence, drastically simplifying the workflow. As demonstrated in the general reaction scheme below, the convergence of 2-alkynyl aniline and aryl iodide precursors under mild thermal conditions (60°C) enables the rapid assembly of the target scaffold with impressive yields, often exceeding 70% across a broad range of substrates.
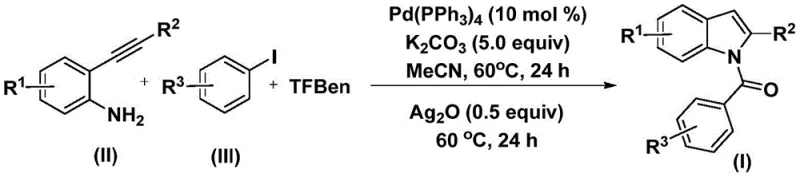
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis. The cycle initiates with the oxidative addition of the aryl iodide to the active Pd(0) species, generating an aryl-palladium(II) intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of TFBen, inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack by the amine group of the 2-alkynyl aniline, followed by reductive elimination to yield an intermediate amide. The final stage involves a silver oxide-mediated intramolecular cyclization, where the alkyne moiety attacks the activated amide or interacts with the metal center to close the five-membered pyrrole ring, regenerating the catalyst and delivering the N-acyl indole product.
From an impurity control perspective, this mechanism offers distinct advantages. The use of silver oxide as a promoter not only facilitates the cyclization step but also helps in scavenging halide ions, driving the equilibrium towards product formation and minimizing the accumulation of dehalogenated side products. The mild reaction temperature of 60°C prevents thermal degradation of sensitive functional groups, ensuring a clean impurity profile which is crucial for regulatory compliance in API manufacturing. The compatibility with various substituents (R1, R2, R3) such as halogens, alkyls, and alkoxy groups allows for the late-stage diversification of the indole core, providing medicinal chemists with a versatile toolbox for structure-activity relationship (SAR) studies without compromising yield or purity.
How to Synthesize N-Acyl Indole Efficiently
To implement this synthesis effectively, precise control over reagent stoichiometry and reaction timing is essential. The protocol dictates a two-stage heating process: an initial 24-hour period for the carbonylation and amidation, followed by the addition of silver oxide and a second 24-hour period for cyclization. Acetonitrile is identified as the optimal solvent due to its ability to dissolve both organic substrates and inorganic bases effectively. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and maximum yield for your specific batch requirements.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits regarding cost stability and sourcing reliability. The reliance on commercially available starting materials, such as substituted aryl iodides and 2-alkynyl anilines, ensures a robust supply chain that is not dependent on exotic or custom-synthesized reagents. The elimination of high-pressure equipment and toxic gas handling infrastructure significantly lowers the capital expenditure (CAPEX) required for production facilities, allowing for more flexible manufacturing setups. Furthermore, the high conversion rates and simplified post-treatment procedures reduce the consumption of solvents and silica gel during purification, leading to substantial cost savings in raw material usage and waste disposal.
- Cost Reduction in Manufacturing: The replacement of gaseous carbon monoxide with a solid surrogate like TFBen removes the need for expensive gas containment systems and specialized safety protocols, directly lowering operational overheads. Additionally, the high atom economy of the carbonylation step means that a greater proportion of the input mass is converted into the final product, reducing the cost per kilogram of the active pharmaceutical ingredient. The use of a standard palladium catalyst, which can potentially be recovered or used in low loadings (10 mol%), further optimizes the cost structure compared to methods requiring precious metals in stoichiometric quantities.
- Enhanced Supply Chain Reliability: Since the key reagents (aryl iodides, alkynes, TFBen) are commodity chemicals available from multiple global vendors, the risk of supply disruption is minimized. This diversity in sourcing options empowers procurement teams to negotiate better terms and maintain continuous production schedules even during market fluctuations. The robustness of the reaction conditions (60°C, ambient pressure) also means that the process can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without extensive re-validation, ensuring consistent supply continuity.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of benign inorganic salts and spent catalyst residues that are easier to treat than the acidic waste streams from traditional Friedel-Crafts acylations. The absence of volatile organic compounds associated with gas handling and the use of acetonitrile, a solvent with well-established recovery protocols, supports stricter environmental compliance standards. This green profile facilitates smoother regulatory approvals and aligns with the sustainability goals of major pharmaceutical companies, making the commercial scale-up of complex pharmaceutical intermediates more feasible and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-acyl indole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, safety, and scalability for potential partners and licensees.
Q: What are the key advantages of this N-acyl indole synthesis method?
A: The method described in patent CN112898192B offers high reaction efficiency, excellent substrate compatibility, and utilizes cheap, readily available starting materials like 2-alkynyl anilines and aryl iodides.
Q: What catalyst system is used for this transformation?
A: The process employs a palladium catalyst, specifically tetrakis(triphenylphosphine)palladium, along with potassium carbonate as a base and silver oxide as an oxidant/promoter.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the reaction operates under mild conditions (60°C) in common solvents like acetonitrile, uses stable solid reagents, and avoids hazardous gaseous carbon monoxide, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has extensively evaluated the carbonylation cyclization route described in CN112898192B and confirmed its potential for delivering high-purity intermediates with exceptional consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acyl indole meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a competitive edge in the global pharmaceutical market.
