Advanced Palladium-Catalyzed Carbonylation for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct privileged scaffolds such as the indole nucleus, which serves as a critical backbone in numerous bioactive molecules ranging from anti-inflammatory agents to antiviral drugs. A recent technological breakthrough documented in patent CN112898192B introduces a highly efficient preparation method for N-acyl indole compounds, leveraging a sophisticated palladium-catalyzed carbonylation strategy. This innovation addresses long-standing challenges in organic synthesis by utilizing a solid carbon monoxide surrogate, thereby bypassing the logistical and safety hazards associated with gaseous CO. The significance of this development cannot be overstated for a reliable pharmaceutical intermediates supplier, as it opens new avenues for accessing complex heterocyclic structures with improved safety profiles and operational simplicity. By integrating readily available starting materials like 2-alkynyl aniline and aryl iodides, this protocol ensures a streamlined pathway that is both economically viable and chemically elegant.
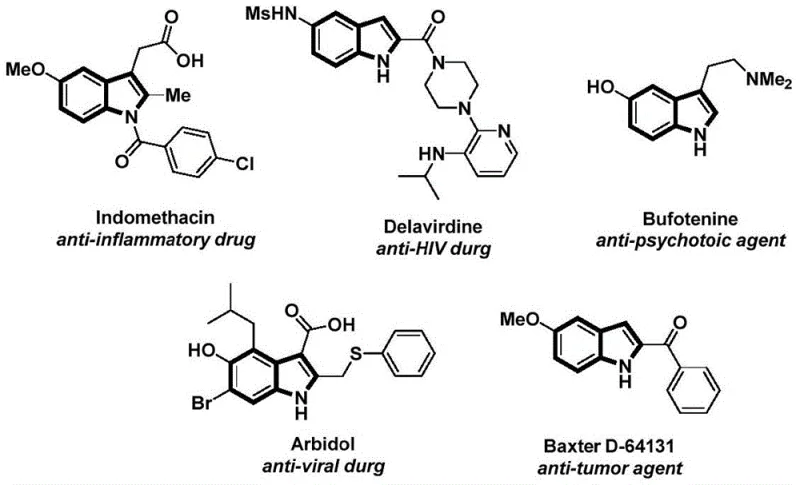
Furthermore, the versatility of this synthetic approach allows for the generation of diverse N-acyl indole derivatives, which are pivotal in the discovery and development of next-generation therapeutics. The ability to synthesize these compounds in a single pot with high atom economy represents a paradigm shift from multi-step, low-yielding traditional processes. For research and development teams focused on expanding their chemical libraries, this method offers a powerful tool to rapidly access novel analogues for biological evaluation. The underlying chemistry not only enhances the speed of synthesis but also aligns with modern green chemistry principles by reducing waste and energy consumption. As we delve deeper into the technical specifics, it becomes evident that this patent provides a foundational technology that can be leveraged for cost reduction in API manufacturing, ensuring that high-quality intermediates are accessible for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indole compounds has been fraught with significant technical hurdles that impede efficient commercial production. Traditional carbonylation reactions often rely on the direct use of carbon monoxide gas, which poses severe safety risks due to its high toxicity and the requirement for specialized high-pressure equipment to maintain reaction efficacy. These stringent safety protocols inevitably lead to inflated capital expenditures and operational complexities, making the process less attractive for large-scale manufacturing environments. Moreover, conventional methods frequently suffer from poor substrate compatibility, where sensitive functional groups on the aromatic rings may degrade under the harsh thermal or acidic conditions required to drive the reaction to completion. This limitation restricts the chemical space that can be explored, forcing chemists to employ cumbersome protecting group strategies that add unnecessary steps and reduce overall yield. Additionally, the use of stoichiometric amounts of hazardous reagents in older protocols generates substantial chemical waste, creating environmental compliance burdens that modern enterprises strive to minimize.
The Novel Approach
In stark contrast, the methodology outlined in CN112898192B revolutionizes the landscape by employing phenol 1,3,5-tricarboxylate (TFBen) as a safe and effective solid carbon monoxide surrogate. This strategic substitution eliminates the need for handling toxic gases, thereby drastically simplifying the reactor setup and enhancing workplace safety for production staff. The reaction proceeds under remarkably mild conditions, typically at 60°C, which preserves the integrity of sensitive functional groups and allows for a broader scope of substrate applicability without the need for extensive protection-deprotection sequences. This one-pot tandem process seamlessly integrates carbonylation and cyclization steps, significantly reducing the processing time and solvent consumption compared to stepwise traditional approaches. For a procurement manager, this translates to tangible benefits such as reduced raw material costs and lower waste disposal fees, directly impacting the bottom line. The robustness of this novel approach ensures consistent product quality and reliability, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, initiating with the oxidative addition of the aryl iodide to the tetrakis(triphenylphosphine)palladium(0) complex. This critical step generates an aryl-palladium intermediate, which subsequently undergoes migratory insertion of carbon monoxide released in situ from the thermal decomposition of TFBen. The resulting acyl-palladium species is highly reactive and poised for nucleophilic attack by the amine functionality of the 2-alkynyl aniline. This sequence forms an amide intermediate, setting the stage for the subsequent intramolecular cyclization. The precision of this catalytic cycle ensures high regioselectivity and minimizes the formation of undesired by-products, which is crucial for maintaining high purity standards required in pharmaceutical applications. Understanding this mechanism allows process chemists to fine-tune reaction parameters such as ligand choice and base concentration to optimize yields further.
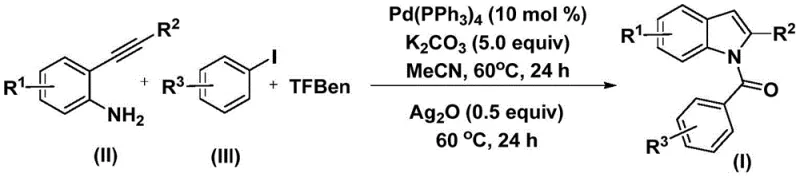
Following the formation of the amide intermediate, the addition of silver oxide plays a pivotal role in promoting the final cyclization step to yield the N-acyl indole core. Silver oxide acts as a mild oxidant and base, facilitating the activation of the alkyne moiety and enabling the intramolecular nucleophilic attack that closes the five-membered ring. This dual-function reagent ensures that the cyclization proceeds smoothly under the same mild thermal conditions, avoiding the need for harsh acids or Lewis acids that could compromise product stability. The impurity profile is tightly controlled throughout this process, as the specific catalytic system favors the desired pathway over potential side reactions such as homocoupling or polymerization. For R&D directors, this level of control over the impurity spectrum is invaluable, as it simplifies downstream purification and ensures that the final high-purity N-acyl indole compounds meet stringent regulatory specifications. The mechanistic clarity provided by this patent empowers manufacturers to replicate the process with confidence and consistency.
How to Synthesize N-Acyl Indole Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring to ensure optimal outcomes. The process begins by combining the palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in an organic solvent such as acetonitrile, which provides excellent solubility for all components. The mixture is then heated to 60°C for approximately 24 hours to allow the carbonylation and coupling steps to reach completion. Subsequently, silver oxide is introduced to the reaction vessel, and heating is continued for another 24 hours to drive the cyclization to full conversion. Detailed standardized synthesis steps see the guide below.
- Combine palladium catalyst, potassium carbonate, TFBen (CO source), 2-alkynyl aniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling steps.
- Add silver oxide to the mixture and continue heating at 60°C for another 24 hours to promote cyclization, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that resonate deeply with procurement managers and supply chain heads looking to optimize their vendor networks. The elimination of high-pressure carbon monoxide equipment significantly lowers the barrier to entry for manufacturing partners, allowing for a more diversified and resilient supply base. This flexibility ensures that production can be scaled up rapidly in response to market demand without being bottlenecked by specialized infrastructure limitations. Furthermore, the use of commercially available and inexpensive starting materials reduces the volatility associated with sourcing exotic or proprietary reagents, leading to more stable pricing models over time. The simplified work-up procedure, which involves standard filtration and chromatography, minimizes labor costs and processing time, contributing to overall operational efficiency.
- Cost Reduction in Manufacturing: The substitution of toxic CO gas with a solid surrogate like TFBen removes the need for expensive gas handling systems and rigorous safety monitoring, resulting in substantial capital expenditure savings. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high efficiency of the catalyst minimizes the amount of precious metal required per batch. These factors collectively drive down the cost of goods sold, allowing for more competitive pricing in the global market without sacrificing margin. The reduction in waste generation also lowers disposal costs, further enhancing the economic viability of the process for large-scale operations.
- Enhanced Supply Chain Reliability: By relying on widely available commodity chemicals such as aryl iodides and 2-alkynyl anilines, the risk of supply disruption is significantly mitigated compared to processes dependent on niche reagents. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This reliability is critical for maintaining continuous manufacturing schedules and meeting tight delivery deadlines for downstream pharmaceutical clients. The ability to produce high-purity intermediates consistently strengthens the trust between suppliers and buyers, fostering long-term partnerships.
- Scalability and Environmental Compliance: The inherent safety of using solid reagents and mild temperatures makes this process exceptionally easy to scale from kilogram to tonne quantities without encountering the engineering challenges typical of high-pressure gas reactions. This scalability ensures that supply can grow in tandem with clinical or commercial demand. Moreover, the reduced environmental footprint aligns with increasingly strict global regulations on emissions and waste, positioning manufacturers as responsible corporate citizens. The simplified purification process reduces solvent usage, contributing to greener manufacturing practices that are highly valued by environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical scope for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the safety advantages of using TFBen over carbon monoxide gas?
A: Using phenol 1,3,5-tricarboxylate (TFBen) as a solid carbon monoxide surrogate eliminates the need for handling toxic, high-pressure CO gas, significantly improving operational safety and reducing infrastructure costs for manufacturing facilities.
Q: How does this method improve substrate compatibility compared to traditional routes?
A: The mild reaction conditions (60°C) and the specific palladium catalytic system allow for excellent tolerance of various functional groups, including halogens and alkoxy groups, which might decompose under harsher traditional cyclization conditions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of commercially available reagents, mild temperatures, and a simplified one-pot procedure makes this method highly scalable and cost-effective for industrial applications without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN112898192B and are fully equipped to leverage them for your project needs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are designed to handle complex catalytic reactions with precision, supported by rigorous QC labs that guarantee stringent purity specifications for every batch. We understand that in the fast-paced pharmaceutical industry, time and quality are paramount, and our dedicated team is committed to delivering excellence at every stage of the value chain.
We invite you to collaborate with us to unlock the full commercial potential of N-acyl indole compounds for your pipeline. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us be your trusted partner in driving innovation and efficiency in your supply chain.
