Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The rapid evolution of medicinal chemistry demands robust, safe, and scalable methodologies for constructing privileged heterocyclic scaffolds. Among these, the 1,2,3-triazole ring system stands out due to its exceptional metabolic stability and ability to act as a bioisostere for amide bonds and other linkers. However, introducing electron-withdrawing groups like the trifluoromethyl moiety at the 5-position has historically presented significant synthetic challenges, often relying on hazardous reagents or expensive transition metal catalysts. Patent CN113121462B introduces a transformative approach to this problem, detailing a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds that circumvents these traditional bottlenecks. This technology leverages a base-promoted cyclization strategy between readily available trifluoroethylimidoyl chlorides and diazo compounds, offering a pathway that is not only chemically efficient but also inherently safer for industrial environments. For R&D directors and procurement specialists alike, this represents a critical opportunity to streamline the supply chain for fluorinated heterocycles, reducing reliance on volatile azide chemistry while maintaining high yields and structural diversity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted triazoles has been dominated by two primary strategies, both of which carry substantial operational liabilities for commercial manufacturing. The first and most common method involves the copper-catalyzed [3+2] cycloaddition of alkynes with organic azides to form a triazole-metal complex, which subsequently reacts with various trifluoromethylating reagents. While effective on a small scale, this route is plagued by the inherent instability and explosive nature of organic azides, posing severe safety risks during scale-up. Furthermore, the requirement for copper catalysts introduces the necessity for rigorous downstream purification to remove trace heavy metals, a step that is both costly and time-consuming, particularly for pharmaceutical intermediates destined for clinical use. The second conventional approach utilizes organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones. Although metal-free, this method still necessitates the handling of toxic and potentially explosive azide precursors, creating significant regulatory and safety hurdles for process chemistry teams aiming for green manufacturing standards.
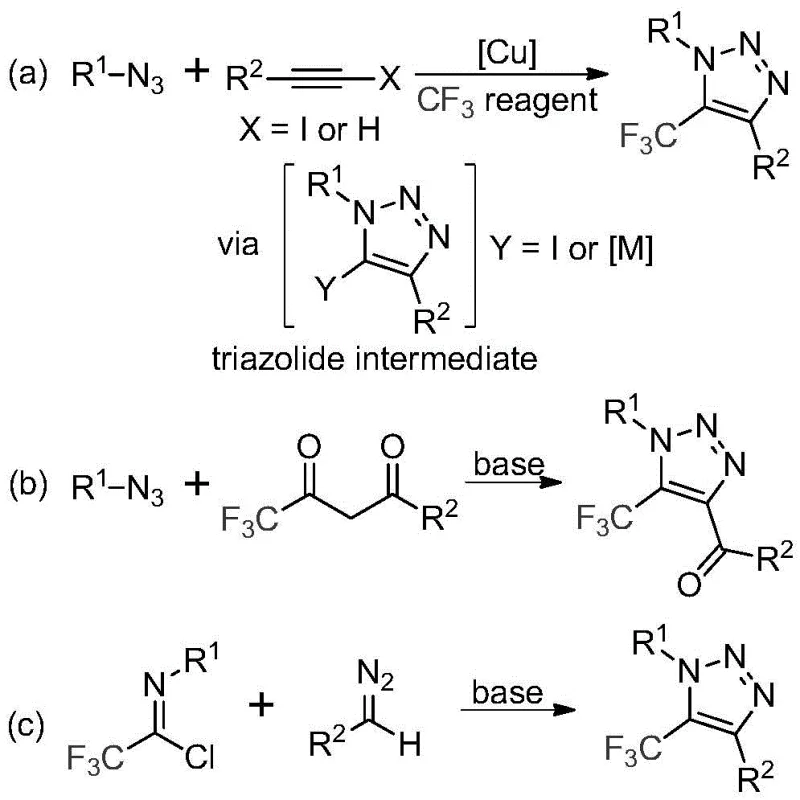
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN113121462B offers a paradigm shift by completely eliminating the need for both transition metal catalysts and explosive azide reagents. The core innovation lies in the direct coupling of trifluoroethylimidoyl chlorides with diazo compounds under mild basic conditions. This metal-free protocol utilizes inexpensive inorganic bases, such as cesium carbonate, to promote the reaction in common polar aprotic solvents like acetonitrile. By avoiding copper, the process inherently produces a cleaner crude product profile, drastically simplifying the workup procedure and eliminating the need for specialized metal scavenging resins. Moreover, the substitution of hazardous azides with more stable diazo precursors significantly enhances the safety profile of the operation, allowing for safer handling in larger reactor volumes. This approach not only aligns with modern principles of green chemistry but also provides a cost-effective route for the commercial scale-up of complex pharmaceutical intermediates, ensuring a reliable supply of high-value fluorinated building blocks.
Mechanistic Insights into Base-Promoted Cyclization
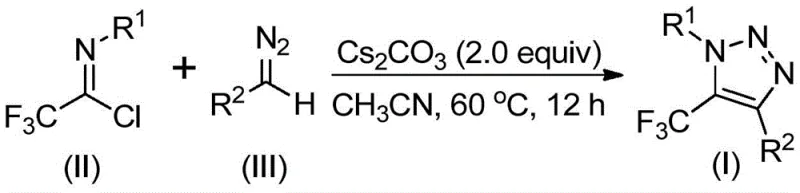
The mechanistic pathway proposed for this transformation offers fascinating insights into the reactivity of fluorinated imines and diazo species under basic conditions. The reaction is believed to initiate with a base-promoted intermolecular nucleophilic addition-elimination process. Specifically, the diazo compound, acting as a nucleophile, attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step is facilitated by the strong electron-withdrawing nature of the trifluoromethyl group, which activates the imine carbon towards nucleophilic assault. The elimination of the chloride ion generates a key intermediate, effectively forging the new carbon-carbon bond that links the diazo fragment to the trifluoromethyl-imine scaffold. This initial coupling is critical, as it sets the stage for the subsequent ring closure without requiring external activation from a metal center.
Following the formation of the linear intermediate, the system undergoes an intramolecular 5-endo-dig cyclization to generate the final 1,2,3-triazole ring. This cyclization step is driven by the nucleophilic attack of the terminal nitrogen of the diazo group onto the adjacent electrophilic center, closing the five-membered heterocyclic ring. The use of cesium carbonate is particularly advantageous here; as a mild yet effective base, it likely assists in the deprotonation events required to drive the equilibrium towards the aromatic triazole product. This mechanism explains the broad substrate tolerance observed in the patent examples, where various electron-donating and electron-withdrawing groups on the aromatic rings of both the imidoyl chloride and the diazo compound are well-tolerated. The result is a highly versatile synthetic platform capable of generating diverse libraries of high-purity pharmaceutical intermediates with precise control over the substitution pattern at the 1 and 4 positions of the triazole ring.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, making it an ideal candidate for technology transfer from the laboratory to pilot plant operations. The protocol typically involves charging a reaction vessel with the trifluoroethylimidoyl chloride, the specific diazo compound, and cesium carbonate in a molar ratio optimized for maximum conversion, often utilizing a slight excess of the diazo species to drive the reaction to completion. The mixture is suspended in anhydrous acetonitrile, often with the addition of 4Å molecular sieves to sequester trace moisture that could hydrolyze the sensitive imidoyl chloride starting material. Heating the reaction to moderate temperatures, typically around 60°C, for a period of 12 hours allows the cyclization to proceed smoothly. Upon completion, the workup is straightforward, involving simple filtration to remove inorganic salts followed by standard purification techniques.
- Combine trifluoroethylimidoyl chloride, diazo compound, and cesium carbonate in anhydrous acetonitrile with 4Å molecular sieves.
- Heat the reaction mixture to 60°C and stir for 12 hours under inert atmosphere to ensure complete conversion.
- Filter the mixture, concentrate the filtrate, and purify the crude residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free, azide-free methodology translates directly into tangible operational efficiencies and risk mitigation. The elimination of copper catalysts removes a significant cost center associated with catalyst procurement and, more importantly, the downstream removal of heavy metal residues, which often requires expensive scavenging agents and extended processing times. Furthermore, the avoidance of explosive azide reagents dramatically lowers the safety compliance burden, reducing the need for specialized blast-proof infrastructure and lowering insurance premiums associated with hazardous chemical storage. This streamlined process flow ensures a more predictable production schedule, minimizing the risk of batch failures due to catalyst poisoning or azide instability.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the use of commodity-grade reagents and the simplification of the purification workflow. By utilizing cesium carbonate instead of precious metal catalysts like copper or ruthenium, the raw material costs are significantly lowered. Additionally, the absence of metal contaminants means that the purification process does not require specialized chromatography columns or scavenging resins, leading to substantial cost savings in consumables and solvent usage. The high atom economy of the cyclization further contributes to reduced waste generation, lowering the overall cost of goods sold (COGS) for the final triazole intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Trifluoroethylimidoyl chlorides and diazo compounds are commercially accessible from multiple global suppliers, reducing the risk of single-source dependency that often plagues specialized catalyst supply chains. The robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. This reliability ensures consistent delivery timelines for reliable pharmaceutical intermediate suppliers serving the global market, allowing downstream drug manufacturers to maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers distinct advantages for large-volume production. The reaction generates minimal hazardous waste compared to traditional azide-based routes, simplifying effluent treatment and disposal protocols. The mild thermal requirements allow for the use of standard glass-lined or stainless steel reactors without the need for high-pressure autoclaves, facilitating a smoother transition from kilogram to ton-scale manufacturing. This ease of scale-up supports the growing demand for fluorinated heterocycles in the agrochemical and pharmaceutical sectors, ensuring that supply can meet market needs without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined within the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential licensees or manufacturing partners.
Q: Does this synthesis method involve hazardous azide reagents?
A: No, unlike traditional copper-catalyzed click chemistry, this patented method utilizes stable diazo compounds and imidoyl chlorides, completely eliminating the safety risks associated with handling explosive organic azides.
Q: Is the process suitable for large-scale manufacturing?
A: Yes, the reaction operates under mild thermal conditions (50-70°C) using inexpensive inorganic bases like cesium carbonate, making it highly amenable to scale-up from gram to multi-kilogram production without complex pressure vessels.
Q: What is the typical purity profile of the resulting triazoles?
A: The method demonstrates high functional group tolerance and clean reaction profiles, typically yielding products that can be purified to >98% purity via standard silica gel chromatography, suitable for API intermediate specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global fine chemicals market. Our team of expert process chemists has thoroughly evaluated the methodology described in patent CN113121462B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient route to full industrial maturity. We are committed to delivering stringent purity specifications for all our fluorinated intermediates, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to ensure every batch meets the highest quality standards demanded by the pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative synthesis for your next drug discovery program or commercial campaign. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our triazole library and discuss route feasibility assessments for your custom synthesis projects, ensuring a seamless partnership from gram-scale development to commercial supply.
