Scalable Metal-Free Production of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Production of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Drug Discovery
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with fluorine atoms to enhance metabolic stability and bioavailability. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical bottlenecks in current synthetic workflows. This technology leverages a base-promoted cyclization strategy that bypasses the need for toxic azides and expensive transition metal catalysts, offering a safer and more economically viable pathway for producing high-value intermediates. For R&D directors and procurement managers alike, this innovation represents a significant leap forward in process chemistry, enabling the efficient assembly of complex molecular scaffolds that are pivotal in modern drug design. The ability to access these structures without the baggage of heavy metal contamination or hazardous reagents positions this method as a superior choice for GMP manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloadditions or organocatalytic reactions involving trifluoromethyl ketones. These traditional pathways, while effective in academic settings, present substantial challenges when translated to industrial scale. The primary concern is the reliance on organic azides, which are inherently unstable and pose severe explosion risks during handling and storage, necessitating specialized equipment and rigorous safety protocols that drive up operational costs. Furthermore, the use of copper catalysts introduces a persistent impurity profile that requires extensive downstream purification to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients. The combination of hazardous reagents and difficult purification steps creates a fragile supply chain that is prone to delays and quality deviations, making these conventional routes less attractive for commercial production.
The Novel Approach
In stark contrast, the methodology disclosed in CN113121462B utilizes cheap and readily available diazo compounds and trifluoroethylimidoyl chloride as starting materials, completely eliminating the need for azides or metal catalysts. This metal-free, base-promoted approach not only enhances process safety by removing explosive hazards but also streamlines the purification workflow by avoiding heavy metal scavenging steps. The reaction proceeds efficiently under mild thermal conditions, demonstrating excellent functional group tolerance and high yields across a diverse range of substrates. By shifting the paradigm from metal-catalyzed coupling to nucleophilic cyclization, this novel route offers a cleaner, greener, and more cost-effective solution for synthesizing these valuable heterocycles. 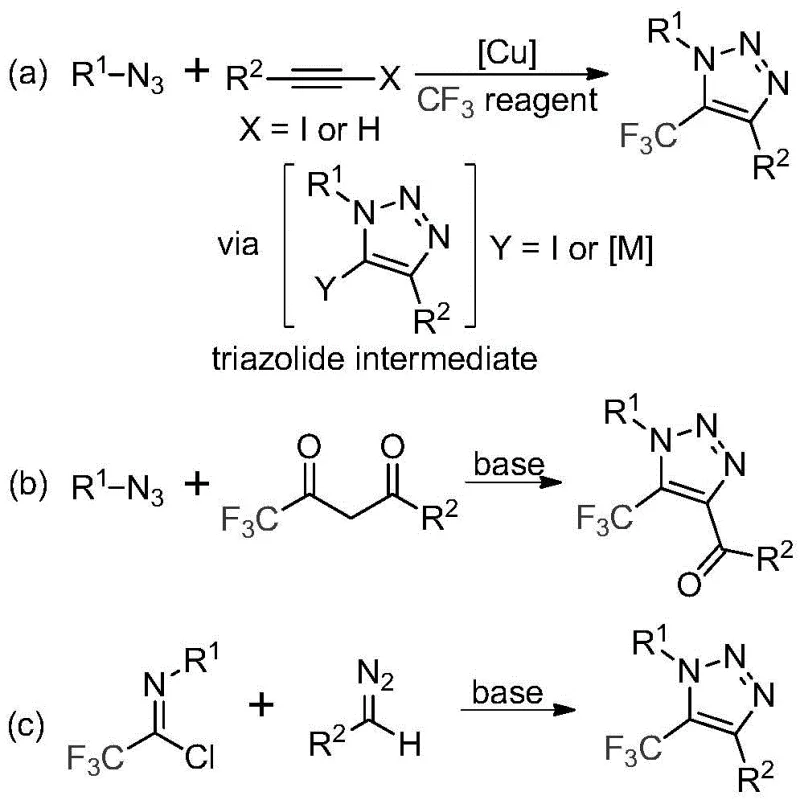
Mechanistic Insights into Base-Promoted Cyclization
The core of this innovative synthesis lies in a sophisticated base-promoted mechanism that facilitates the formation of the triazole ring through a sequence of nucleophilic additions and eliminations. The reaction initiates with the activation of the diazo compound by the base, likely cesium carbonate, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This intermolecular nucleophilic addition-elimination process is crucial for establishing the initial carbon-carbon bond, setting the stage for the subsequent ring closure. The choice of base and solvent plays a pivotal role in modulating the reactivity of the diazo species, ensuring that the reaction proceeds with high regioselectivity to favor the desired 5-trifluoromethyl isomer over potential byproducts. Understanding these mechanistic nuances allows chemists to fine-tune reaction parameters for optimal performance across different substrate classes.
Following the initial bond formation, the intermediate undergoes an intramolecular 5-endo-dig cyclization to close the triazole ring, driven by the thermodynamic stability of the aromatic system. This cyclization step is facilitated by the electron-withdrawing nature of the trifluoromethyl group, which activates the adjacent positions for ring closure. The entire process is remarkably clean, minimizing the formation of polymeric side products often associated with diazo chemistry. The result is a high-purity product that requires minimal purification, typically achievable through standard silica gel chromatography. This mechanistic clarity provides confidence in the reproducibility of the process, a key factor for technology transfer and scale-up activities. 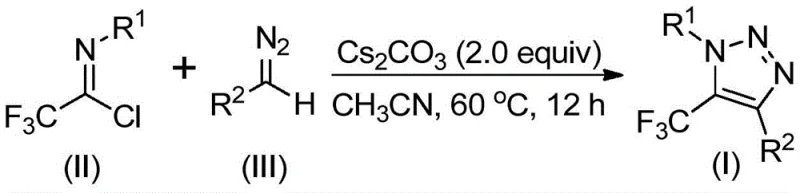
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward, requiring standard glassware and commonly available reagents. The protocol involves mixing the trifluoroethylimidoyl chloride and diazo compound in an aprotic solvent such as acetonitrile, followed by the addition of cesium carbonate as the promoter. The reaction mixture is then heated to a moderate temperature, typically around 60°C, and stirred for a period of 8 to 16 hours to ensure complete conversion. Workup is simplified to filtration and silica gel treatment, followed by column chromatography to isolate the pure triazole derivative. This operational simplicity makes the method accessible even to facilities with limited specialized equipment.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, diazo compound, and an organic solvent such as acetonitrile in a reaction vessel.
- Heat the reaction mixture to a temperature between 50°C and 70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Upon completion, filter the mixture, treat with silica gel, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible improvements in cost structure and supply reliability. The elimination of copper catalysts removes the need for expensive metal scavengers and the associated waste disposal costs, directly impacting the bottom line. Moreover, the avoidance of hazardous azides reduces insurance premiums and safety compliance costs, further enhancing the economic attractiveness of the process. The use of commercially available starting materials ensures a stable supply chain, reducing the risk of raw material shortages that can plague specialized reagent markets. This robustness allows for better forecasting and inventory management, critical components of a resilient supply chain strategy.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts significantly lowers the cost of goods sold by eliminating expensive catalyst purchases and the complex purification steps required to remove trace metals. Additionally, the use of inexpensive bases like cesium carbonate and common solvents like acetonitrile keeps raw material costs low, while the high reaction efficiency minimizes waste generation and maximizes yield per batch. These factors combine to create a highly economical process that offers substantial cost savings compared to traditional metal-catalyzed routes.
- Enhanced Supply Chain Reliability: By relying on stable, non-explosive starting materials that are widely available from multiple chemical suppliers, this method mitigates the risk of supply disruptions. The absence of specialized or hazardous reagents simplifies logistics and storage requirements, allowing for larger inventory buffers without safety concerns. This stability ensures consistent production schedules and reliable delivery timelines, which are essential for maintaining uninterrupted manufacturing operations in the pharmaceutical sector.
- Scalability and Environmental Compliance: The mild reaction conditions and simple workup procedures make this process highly scalable from gram to multi-kilogram quantities without significant re-optimization. The reduction in hazardous waste and the absence of heavy metals align with green chemistry principles, facilitating easier regulatory approval and environmental compliance. This scalability ensures that the method can grow with demand, supporting the transition from clinical trials to commercial production seamlessly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for their own pipelines.
Q: What are the safety advantages of this synthesis method compared to traditional routes?
A: This method eliminates the use of organic azides, which are known for their toxicity and explosive hazards, and avoids transition metal catalysts like copper, simplifying purification and enhancing operational safety.
Q: How does this process impact the cost of manufacturing pharmaceutical intermediates?
A: By utilizing inexpensive bases like cesium carbonate and readily available starting materials while removing the need for expensive metal catalysts and complex removal steps, the overall production cost is significantly reduced.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the reaction operates under mild thermal conditions (50-70°C) in common solvents like acetonitrile and involves simple workup procedures, making it highly amenable to commercial scale-up from gram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for producing high-purity 5-trifluoromethyl-1,2,3-triazoles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest industry standards for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this process for your specific needs, delivering both quality and quantity with precision.
We invite you to engage with our technical procurement team to discuss how this innovative methodology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free route for your specific API intermediates. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, technical excellence, and mutual success.
