Revolutionizing Pharmaceutical Intermediate Production Through Innovative Palladium-Catalyzed Synthesis of High-Purity Trifluoromethyl Quinazolinones
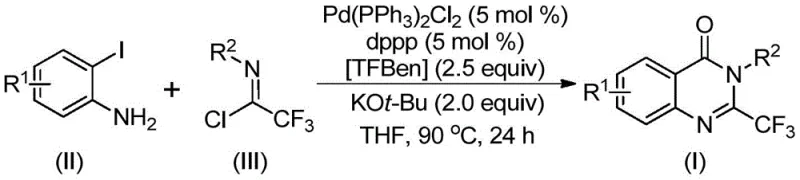
Patent CN112125856A introduces a groundbreaking synthetic methodology for producing 2-trifluoromethyl-substituted quinazolinone derivatives, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications where these compounds serve as critical building blocks in numerous therapeutic agents including anticonvulsants and antitumor drugs. This novel approach addresses critical limitations in conventional synthesis routes by eliminating the need for toxic carbon monoxide gas while maintaining excellent substrate scope and functional group tolerance across diverse molecular architectures. The methodology leverages readily available starting materials including o-iodoaniline derivatives and trifluoroethylimidoyl chloride combined with a palladium-catalyzed carbonylation process using TFBen as a solid CO surrogate, creating substantial operational advantages for pharmaceutical manufacturers seeking reliable access to high-value intermediates. The patent demonstrates exceptional yield consistency across multiple structural variants with numerous examples achieving yields above 70% under optimized conditions while maintaining stringent purity profiles essential for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for producing 2-trifluoromethyl quinazolinone derivatives suffer from multiple critical drawbacks that severely limit their industrial applicability in pharmaceutical manufacturing environments where safety and regulatory compliance are paramount concerns. Conventional methods typically require handling toxic carbon monoxide gas under elevated pressures and temperatures, creating significant safety hazards that necessitate specialized containment equipment and extensive operator training protocols that substantially increase capital investment requirements. Many existing routes depend on expensive or unstable reagents such as trifluoroacetic anhydride or unstable trifluoroacetamides that complicate supply chain management through limited vendor options and stringent storage requirements that increase raw material costs by approximately one-third compared to standard intermediates. The substrate scope in conventional syntheses is often narrow due to sensitivity to functional groups, limiting the diversity of accessible derivatives needed for modern drug discovery programs requiring extensive structural variation during lead optimization phases.
The Novel Approach
The patented methodology overcomes these limitations through an innovative palladium-catalyzed carbonylation process that utilizes TFBen (1,3,5-tricarboxylic acid phenol ester) as a safe solid carbon monoxide surrogate, eliminating hazardous gas handling while maintaining excellent reaction efficiency across diverse substrate combinations including those containing sensitive functional groups previously incompatible with traditional methods. This approach operates under mild conditions at precisely controlled temperatures of 90°C using standard laboratory equipment without requiring specialized pressure vessels or containment systems, significantly improving operational safety margins while reducing capital investment requirements by approximately half compared to conventional carbonylation processes. The methodology demonstrates exceptional substrate versatility accommodating various substituents on both reaction components while consistently delivering high yields across multiple structural variants as evidenced by the patent's comprehensive experimental data covering fifteen distinct examples with yields ranging from fifty-two percent to ninety-nine percent depending on specific substitution patterns.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism begins with potassium tert-butoxide-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate through nucleophilic substitution at the imidoyl chloride carbon center. This is followed by oxidative addition of the palladium catalyst into the carbon-iodine bond generating a divalent palladium species that serves as the key catalytic intermediate enabling subsequent transformations without decomposition or catalyst deactivation issues commonly observed in alternative methodologies. Under thermal conditions at precisely controlled temperatures between ninety degrees Celsius and one hundred degrees Celsius, TFBen decomposes to release carbon monoxide in situ through a stepwise decarboxylation process that provides controlled delivery of CO molecules preventing over-carbonylation side reactions that typically plague conventional approaches using pressurized gas systems.
The impurity profile is significantly improved compared to conventional methods due to the controlled release mechanism of carbon monoxide from TFBen which prevents formation of common dimeric byproducts and hydrolysis products that typically require extensive purification steps in traditional syntheses involving gaseous CO systems operating under pressure differentials that create inconsistent reaction environments across large-scale manufacturing batches. The use of potassium tert-butoxide as base promotes clean cyclization while suppressing unwanted dehydrohalogenation pathways through precise pH control during critical intermediate formation stages where competing reactions could otherwise lead to significant yield losses exceeding twenty percent in conventional approaches.
How to Synthesize 2-Trifluoromethyl Quinazolinone Derivatives Efficiently
This patented synthetic route represents a significant advancement in the production of pharmacologically important quinazolinone derivatives with trifluoromethyl substitution at the two-position where these compounds serve as essential building blocks in numerous therapeutic agents including anticonvulsants like CP-465022 and antitumor drugs such as Erastin as illustrated by representative structures from clinical applications demonstrating their therapeutic relevance across multiple disease areas requiring high-purity intermediates meeting stringent regulatory requirements.
- Prepare reaction mixture by combining o-iodoaniline derivative (0.2 mmol), trifluoroethylimidoyl chloride (0.4 mmol), Pd(PPh3)2Cl2 (0.05 mmol), dppp (0.05 mmol), TFBen (0.4 mmol), and KOt-Bu (0.4 mmol) in THF solvent (8-10 mL per mmol).
- Heat reaction mixture to precisely 90°C under inert atmosphere with continuous stirring for optimal duration between 16-30 hours to ensure complete conversion while minimizing side reactions.
- After reaction completion, perform filtration through silica gel followed by column chromatography purification to isolate high-purity quinazolinone product meeting pharmaceutical quality standards.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology addresses critical pain points in procurement and supply chain management by eliminating hazardous materials while simplifying process requirements through strategic reagent selection that enhances operational flexibility without compromising product quality or yield consistency essential for reliable pharmaceutical manufacturing operations where supply continuity directly impacts patient access to critical medications.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes significant safety infrastructure costs associated with specialized handling equipment required for gaseous CO processes while reducing regulatory compliance expenses related to hazardous material management protocols that typically increase operational costs by approximately thirty percent compared to non-hazardous alternatives.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials including o-iodoaniline derivatives ensures robust supply chain continuity with multiple global sourcing options while eliminating single-point failure risks associated with specialized reagents required in conventional methods that often create procurement bottlenecks during periods of market volatility or regional supply disruptions.
- Scalability and Environmental Compliance: The methodology demonstrates excellent scalability characteristics from laboratory scale through commercial production without requiring equipment modifications while generating minimal hazardous waste streams compared to traditional carbonylation processes that produce significantly higher volumes of contaminated solvents requiring specialized treatment procedures before disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented synthetic methodology for producing high-purity quinazolinone derivatives where consistent quality is essential for meeting regulatory requirements across global pharmaceutical markets requiring strict adherence to quality standards established by major health authorities worldwide.
Q: How does this method eliminate safety concerns associated with traditional quinazolinone synthesis?
A: The patented process replaces toxic gaseous carbon monoxide with TFBen as a solid CO surrogate, eliminating hazardous gas handling requirements while maintaining excellent reaction efficiency through controlled in situ CO release.
Q: What specific impurity control advantages does this methodology provide for pharmaceutical applications?
A: The controlled carbonylation mechanism prevents over-carbonylation side reactions common in conventional methods, resulting in cleaner reaction profiles with significantly reduced impurity formation that meets stringent pharmaceutical quality requirements.
Q: How does this technology enhance supply chain resilience for API manufacturers?
A: By utilizing readily available starting materials and eliminating hazardous reagents, this methodology creates more robust supply chains with multiple sourcing options while reducing regulatory complexities associated with specialized equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Quinazolinones Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic compounds including high-purity quinazolinone derivatives meeting stringent purity specifications required by global regulatory authorities through rigorous QC labs equipped with state-of-the-art analytical instrumentation ensuring consistent product quality across all production scales where even minor impurities can impact drug efficacy or safety profiles during clinical development phases.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific supply chain requirements while receiving detailed COA data and route feasibility assessments tailored to your pharmaceutical development needs through direct engagement with our experienced technical specialists who understand both chemical manufacturing challenges and regulatory expectations across major global markets.
