Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. Patent CN112239456B introduces a groundbreaking preparation method for substituted 2,3-dihydroquinolone compounds, utilizing a transition metal palladium-catalyzed carbonylation reaction. This technology addresses critical challenges in the synthesis of these valuable intermediates by employing N-pyridine sulfonyl-o-iodoaniline and olefins as starting materials in the presence of a solid carbon monoxide substitute. The significance of this chemical class cannot be overstated, as the 2,3-dihydroquinolone skeleton is prevalent in numerous bioactive molecules, including those with demonstrated antitumor and analgesic properties. As illustrated in the structural examples of known bioactive agents, the versatility of this core structure allows for diverse functionalization to tune pharmacological profiles.
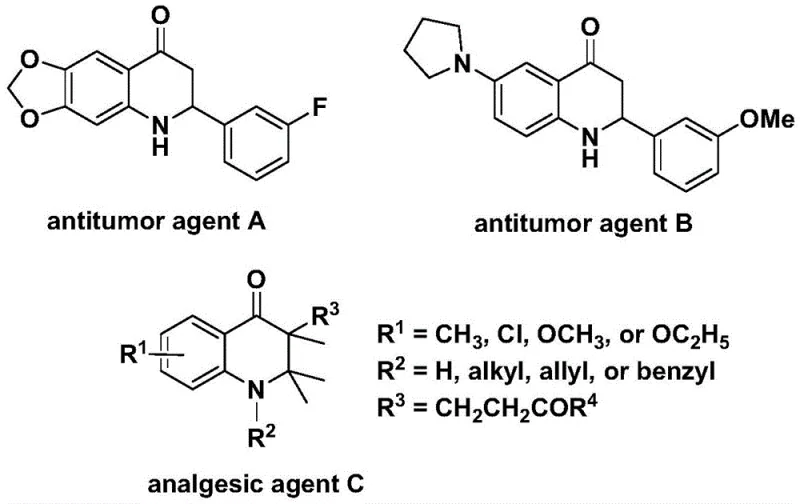
For R&D directors and process chemists, the ability to access these complex heterocycles through a robust and scalable methodology is paramount. The disclosed invention not only simplifies the operational procedure but also enhances reaction efficiency and substrate compatibility, making it a highly attractive candidate for the manufacturing of high-purity pharmaceutical intermediates. By leveraging this patented approach, organizations can secure a reliable pharmaceutical intermediate supplier pathway that mitigates the risks associated with traditional, more hazardous carbonylation techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-containing heterocyclic rings like 2,3-dihydroquinolones has often relied on direct carbonylation using carbon monoxide gas. While effective in principle, the use of gaseous CO presents severe safety hazards, requiring specialized high-pressure equipment and rigorous safety protocols to prevent toxicity and explosion risks. Furthermore, conventional methods frequently suffer from limited substrate scope, where sensitive functional groups on the aromatic ring or the olefin partner may degrade under harsh reaction conditions. Many existing protocols also struggle with regioselectivity issues or require expensive and difficult-to-remove catalysts, leading to complex purification workflows that drive up manufacturing costs. These limitations create significant bottlenecks in cost reduction in API manufacturing, as the need for specialized infrastructure and extensive waste treatment increases the overall process burden.
The Novel Approach
The methodology described in patent CN112239456B offers a transformative solution by replacing toxic CO gas with 1,3,5-trimesic acid phenol ester, a solid and stable carbon monoxide surrogate. This strategic substitution fundamentally alters the safety profile of the reaction, allowing it to be conducted under standard atmospheric pressure conditions without the need for specialized gas handling infrastructure. The reaction proceeds via a palladium-catalyzed cycle where the catalyst inserts into the carbon-nitrogen bond of the iodoaniline derivative, followed by CO insertion and subsequent olefin coordination. As depicted in the general reaction scheme, this process efficiently couples N-pyridine sulfonyl-o-iodoaniline with various olefins to yield the target 2,3-dihydroquinolone derivatives with high precision.
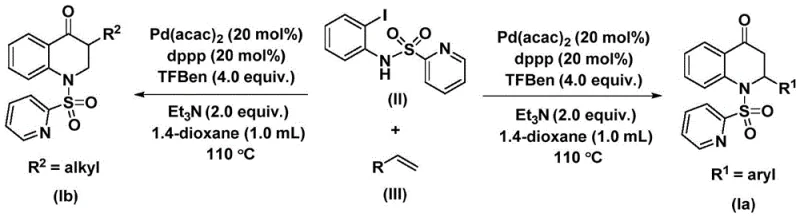
This novel approach significantly broadens the scope of accessible chemical space, enabling the synthesis of both 2-aryl and 3-alkyl substituted variants through simple substrate design. The use of readily available starting materials and a straightforward post-treatment procedure involving filtration and column chromatography ensures that the process is not only chemically elegant but also practically viable for industrial application. For procurement teams, this translates to a supply chain that is less dependent on hazardous gases and more resilient to regulatory changes regarding safety and environmental compliance.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
A deep understanding of the catalytic cycle is essential for optimizing reaction parameters and ensuring consistent product quality. The mechanism initiates with the oxidative addition of the palladium catalyst, specifically bis(acetylacetone)palladium coordinated with the dppp ligand, into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is the key species driving the subsequent transformations. The presence of the pyridine sulfonyl group on the nitrogen atom plays a crucial role in stabilizing the intermediate and directing the regioselectivity of the cyclization. Following oxidative addition, the carbon monoxide molecule, released in situ from the thermal decomposition of the 1,3,5-trimesic acid phenol ester, inserts into the palladium-carbon bond to form an acyl-palladium species.

Subsequently, the olefin substrate coordinates to the palladium center and undergoes migratory insertion into the acyl-palladium bond, forming a new carbon-carbon bond and generating a palladium-alkyl intermediate. The final step involves reductive elimination, which releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst to continue the cycle. This mechanistic pathway explains the high tolerance for various functional groups observed in the experimental data, as the mild conditions prevent side reactions such as polymerization of the olefin or decomposition of sensitive substituents. For quality control purposes, understanding this mechanism allows for better prediction of potential impurities, such as those arising from incomplete CO insertion or beta-hydride elimination, facilitating the development of robust purification strategies to meet stringent purity specifications.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-value intermediates in a laboratory or pilot plant setting. The procedure involves combining the palladium catalyst, ligand, base, CO surrogate, and substrates in an aprotic organic solvent like dioxane, followed by heating to promote the reaction. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during scale-up operations.
- Charge a reaction vessel with bis(acetylacetone)palladium, dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and the selected olefin substrate in an organic solvent such as dioxane.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits that extend beyond mere chemical yield. The elimination of gaseous carbon monoxide removes a major logistical and safety hurdle, significantly reducing the capital expenditure required for reactor setup and maintenance. This shift towards safer reagents aligns perfectly with modern green chemistry principles, minimizing the environmental footprint of the manufacturing process and simplifying regulatory compliance. For supply chain heads, the reliance on solid, shelf-stable reagents like 1,3,5-trimesic acid phenol ester ensures greater supply continuity and reduces the risk of production stoppages due to gas delivery failures.
- Cost Reduction in Manufacturing: The use of inexpensive and commercially available starting materials, such as simple olefins and iodoanilines, combined with a catalytic system that operates at relatively low loading, drives down the raw material costs significantly. Furthermore, the simplified post-treatment process, which avoids complex aqueous workups or hazardous gas scrubbing systems, reduces labor and waste disposal expenses. The high reaction efficiency and yield reported in the patent examples mean that less raw material is wasted, directly contributing to improved overall process economics and cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: By utilizing a solid CO surrogate, the process decouples production from the supply of hazardous industrial gases, which can be subject to strict transportation regulations and availability fluctuations. The robustness of the reaction across a wide range of substrates, including those with electron-withdrawing and electron-donating groups, allows for flexible sourcing of raw materials. This flexibility ensures that procurement managers can adapt to market changes without compromising the quality or timeline of the final product delivery, thereby enhancing the reliability of the supply chain for complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: The patent explicitly mentions the successful expansion of the reaction to the gram level, demonstrating its potential for kilogram and ton-scale production. The absence of toxic gas emissions and the use of standard organic solvents make the waste stream easier to manage and treat, facilitating compliance with increasingly stringent environmental regulations. This scalability ensures that the method can grow with demand, supporting the commercial scale-up of complex drug candidates without the need for fundamental process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity for potential partners and stakeholders.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: This method utilizes 1,3,5-trimesic acid phenol ester as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous high-pressure CO gas, thereby significantly improving operational safety.
Q: What types of substituents are compatible with this palladium-catalyzed reaction?
A: The process demonstrates excellent substrate compatibility, tolerating various functional groups including methyl, tert-butyl, methoxy, fluorine, and chlorine on the aryl ring, as well as linear, branched alkyl, and silyl groups on the olefin.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the patent explicitly states that the method is simple to operate, uses cheap and easily obtainable raw materials, and has been validated for gram-level synthesis, indicating strong potential for industrial large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to accelerate drug development pipelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into full-scale manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet rigorous quality standards, supported by our state-of-the-art rigorous QC labs and comprehensive analytical capabilities. Our dedication to process optimization allows us to offer competitive pricing without compromising on the integrity or safety of the final product.
We invite you to collaborate with us to leverage this innovative palladium-catalyzed carbonylation technology for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
