Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazole Intermediates for Commercial Scale
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazole Intermediates for Commercial Scale
The pharmaceutical industry continuously demands efficient routes to fluorinated heterocycles, which serve as critical scaffolds in modern drug design. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing the long-standing challenges of harsh reaction conditions and limited substrate scope found in traditional methodologies. This novel approach leverages a synergistic molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. The significance of this chemical transformation cannot be overstated, as the 1,2,4-triazole core is a privileged structure present in numerous bioactive molecules, including prominent drugs like Sitagliptin and various anticonvulsants. By enabling the construction of these motifs under mild conditions with high efficiency, this technology offers a reliable pharmaceutical intermediate supplier pathway for developing next-generation therapeutics with enhanced metabolic stability and lipophilicity.
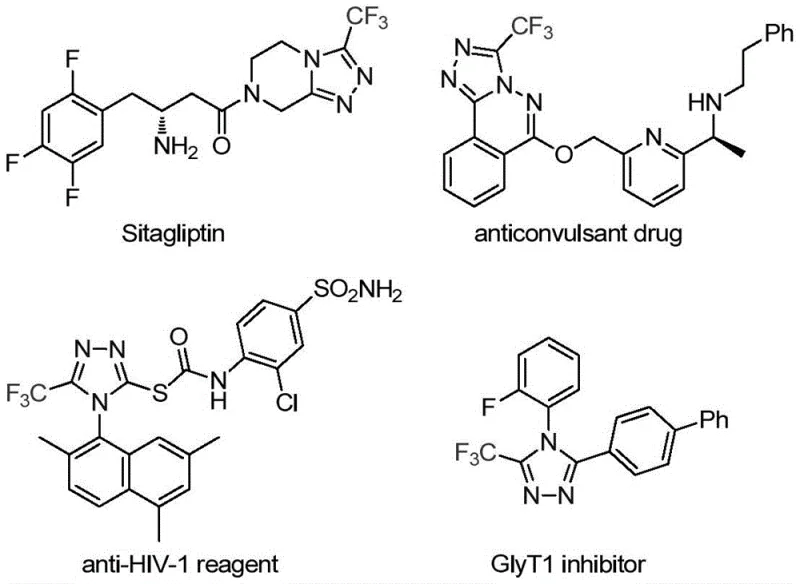
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has relied on cyclization reactions involving trifluoroacetyl hydrazine and amidine compounds, or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These traditional routes often suffer from significant drawbacks, including the requirement for harsh reaction conditions that can degrade sensitive functional groups, leading to lower overall yields and complex impurity profiles. Furthermore, alternative multi-component reactions utilizing diazonium salts or trifluorodiazoethane frequently involve hazardous reagents and generate substantial waste, posing safety risks and environmental compliance challenges for large-scale manufacturing. The lack of versatility in these older methods restricts the ability of chemists to rapidly explore structure-activity relationships, thereby slowing down the drug discovery pipeline and increasing the cost reduction in API manufacturing efforts due to inefficient processes.
The Novel Approach
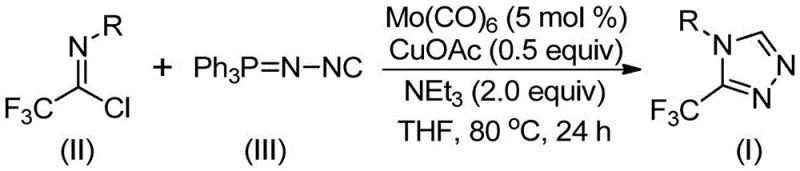
In stark contrast, the methodology disclosed in the patent utilizes a transition metal-catalyzed [3+2] cycloaddition strategy that operates under remarkably mild conditions, typically between 70°C and 90°C. By employing commercially available trifluoroethylimidoyl chloride and functionalized isonitriles as starting materials, this route bypasses the need for unstable or dangerous precursors. The use of molybdenum hexacarbonyl as a metal activator alongside cuprous acetate creates a unique catalytic environment that promotes the formation of the five-membered triazole ring with high regioselectivity. This innovative process not only simplifies the operational procedure but also expands the applicability of the synthesis to a wide range of substrates, allowing for the introduction of diverse functional groups such as alkyl, alkoxy, and halogen substituents without compromising reaction efficiency or product purity.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic pathway of this transformation involves a sophisticated interplay between the molybdenum and copper species. Initially, molybdenum hexacarbonyl interacts with the functionalized isonitrile to form a reactive metal-isocyanide complex, which activates the carbon-nitrogen triple bond for subsequent nucleophilic attack. Concurrently, the cuprous acetate promoter facilitates the coordination of the trifluoroethylimidoyl chloride, lowering the activation energy barrier for the cycloaddition step. This dual-catalyst system ensures that the reaction proceeds through a concerted mechanism to form the key five-membered ring intermediate efficiently. Following the ring closure, the system undergoes a hydrolysis step where water present in the reaction mixture aids in the elimination of triphenylphosphine oxide, driving the equilibrium towards the final 3-trifluoromethyl-substituted 1,2,4-triazole product. This elegant mechanism highlights the importance of precise catalyst loading and solvent choice in achieving optimal conversion rates.
Controlling the impurity profile is paramount for any pharmaceutical intermediate, and this catalytic system offers inherent advantages in this regard. The high selectivity of the Mo/Cu co-catalysis minimizes the formation of side products such as oligomerized isonitriles or hydrolyzed imidoyl chlorides, which are common issues in non-catalyzed thermal reactions. The mild temperature range of 70-90°C further prevents thermal decomposition of the sensitive trifluoromethyl group and the triazole ring, ensuring that the final product maintains its structural integrity. Additionally, the use of triethylamine as a base scavenges the hydrogen chloride byproduct generated during the reaction, preventing acid-catalyzed degradation pathways. This results in a crude reaction mixture that is significantly cleaner, thereby reducing the burden on downstream purification processes and enhancing the overall throughput of the manufacturing operation.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, one must adhere to the optimized stoichiometry and reaction parameters outlined in the patent data. The process begins with the precise weighing of molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), and triethylamine (2.0 equiv) into a reaction vessel containing the appropriate organic solvent, preferably THF. The substrates, trifluoroethylimidoyl chloride and the functionalized isonitrile, are then added, and the mixture is heated to 80°C for approximately 24 hours. Detailed standardized synthesis steps see the guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), trifluoroethylimidoyl chloride, and functionalized isonitrile in THF solvent.
- Heat the reaction mixture to 80°C and maintain stirring for 24 hours under inert atmosphere to ensure complete conversion.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents compelling economic and logistical benefits. The reliance on readily available starting materials, such as aromatic amines and trifluoroacetic acid derivatives, ensures a stable and continuous supply chain, mitigating the risks associated with sourcing exotic or controlled reagents. Furthermore, the operational simplicity of the reaction, which does not require cryogenic conditions or high-pressure equipment, allows for execution in standard glass-lined reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure significantly reduces the capital expenditure required for technology transfer and scale-up, facilitating a faster time-to-market for new drug candidates.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like diazonium salts or trifluorodiazoethane leads to substantial cost savings in raw material procurement. Additionally, the high reaction efficiency and yield observed across a broad substrate scope mean that less starting material is wasted, directly improving the atom economy of the process. The simplified workup procedure, involving basic filtration and chromatography, reduces the consumption of solvents and silica gel, further driving down the operational expenses associated with production.
- Enhanced Supply Chain Reliability: Since the key reagents including molybdenum hexacarbonyl and cuprous acetate are commodity chemicals available from multiple global suppliers, the risk of supply disruption is minimized. The robustness of the reaction conditions allows for flexible scheduling and batch processing, ensuring that production targets can be met consistently even during periods of high demand. This reliability is crucial for maintaining the continuity of API synthesis pipelines and avoiding costly delays in clinical trial material production.
- Scalability and Environmental Compliance: The method has been demonstrated to be scalable to gram levels and beyond, with the potential for ton-scale production given the mild thermal requirements. The use of THF as a solvent, which is easily recoverable and recyclable, aligns with green chemistry principles and helps facilities meet stringent environmental regulations regarding volatile organic compound emissions. The absence of heavy metal waste streams, aside from the catalytic amounts of Mo and Cu which can be recovered, simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this triazole synthesis method. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, offering practical guidance for process chemists and engineers looking to adopt this technology.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, utilizing Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method exhibits excellent substrate tolerance, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, and nitro substituents.
Q: Is the purification process complex for large-scale production?
A: The post-treatment is straightforward, involving simple filtration followed by standard column chromatography, making it highly suitable for scale-up without requiring exotic separation techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay every batch. By leveraging our expertise in transition metal catalysis and heterocyclic chemistry, we can help you optimize this Mo/Cu co-catalyzed route to meet your specific cost and timeline objectives.
We invite you to contact our technical procurement team to discuss your specific requirements for 3-trifluoromethyl-1,2,4-triazole derivatives. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for a new candidate, our experts are ready to provide the support you need. Partner with us to secure a reliable supply of these valuable building blocks and accelerate your drug discovery journey with confidence.
