Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles: A Breakthrough in Pharmaceutical Intermediate Manufacturing
The pharmaceutical and agrochemical industries continuously seek robust, scalable methodologies for constructing nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine motifs which are known to enhance metabolic stability and bioavailability. Patent CN114920707B, published in April 2023, introduces a transformative approach to synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds, a core structural element found in numerous high-value bioactive molecules. As illustrated in the structural diversity of modern therapeutics, the 1,2,4-triazole ring serves as a pivotal pharmacophore in drugs ranging from antidiabetic agents like Sitagliptin to potent Factor IXa inhibitors and GlyT1 inhibitors. 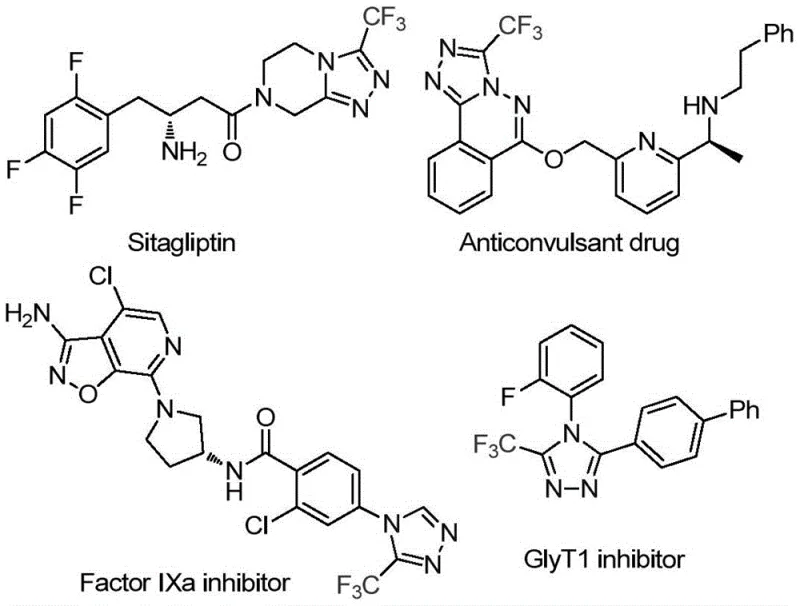 This patent discloses a novel preparation method that utilizes the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a reaction medium, but fundamentally as a carbon source, thereby streamlining the synthetic route and offering substantial advantages for industrial scale-up.
This patent discloses a novel preparation method that utilizes the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a reaction medium, but fundamentally as a carbon source, thereby streamlining the synthetic route and offering substantial advantages for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on multi-step sequences involving the condensation of hydrazides with specific nitriles or amidines, often requiring harsh reaction conditions and expensive, moisture-sensitive reagents. Conventional protocols frequently necessitate the use of strong dehydrating agents or transition metal catalysts that introduce significant challenges in downstream purification, particularly regarding the removal of toxic metal residues to meet stringent pharmaceutical standards. Furthermore, many existing methods suffer from limited substrate scope, failing to tolerate sensitive functional groups or requiring complex protecting group strategies that increase the overall step count and reduce the final yield. The reliance on specialized C1 sources, distinct from the solvent system, adds to the material costs and complicates the supply chain logistics for large-scale manufacturing operations.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in CN114920707B leverages a clever tandem cyclization strategy where DMF serves a dual role as both the polar aprotic solvent and the essential one-carbon synthon. 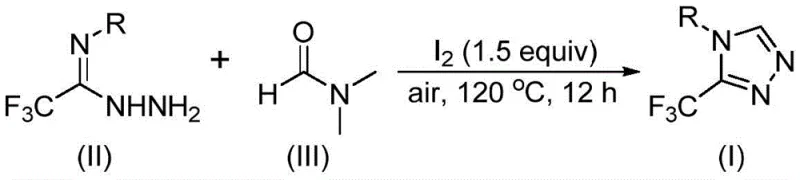 This innovative process employs molecular iodine as a promoter to activate the DMF molecule, facilitating a direct cyclization with trifluoroethyliminohydrazide under remarkably mild conditions. The reaction proceeds efficiently at temperatures between 110°C and 130°C in an open air atmosphere, completely eliminating the need for energy-intensive anhydrous or anaerobic environments. By integrating the carbon source directly into the solvent matrix, this approach drastically simplifies the reaction setup, reduces the volume of chemical waste generated, and enhances the overall atom economy, making it an ideal candidate for cost reduction in API manufacturing.
This innovative process employs molecular iodine as a promoter to activate the DMF molecule, facilitating a direct cyclization with trifluoroethyliminohydrazide under remarkably mild conditions. The reaction proceeds efficiently at temperatures between 110°C and 130°C in an open air atmosphere, completely eliminating the need for energy-intensive anhydrous or anaerobic environments. By integrating the carbon source directly into the solvent matrix, this approach drastically simplifies the reaction setup, reduces the volume of chemical waste generated, and enhances the overall atom economy, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a fascinating example of solvent-participatory synthesis, where the N-methyl and formyl groups of DMF are activated to participate in the ring closure. When the formyl group acts as the carbon source, it undergoes a condensation reaction with the trifluoroethyliminohydrazide to generate a hydrazone intermediate, which subsequently cyclizes intramolecularly with the elimination of dimethylamine to yield the target triazole. Alternatively, when the N-methyl group serves as the carbon source, molecular iodine first reacts with DMF to generate an reactive amine salt species. This activated intermediate then undergoes nucleophilic addition with the hydrazide, followed by the elimination of N-methylformamide to form an azadiene intermediate, which finally undergoes oxidative aromatization to furnish the stable 3-trifluoromethyl-1,2,4-triazole product.  This dual-pathway capability ensures high conversion rates across a wide range of substrates, providing flexibility in optimizing reaction conditions for specific derivative synthesis.
This dual-pathway capability ensures high conversion rates across a wide range of substrates, providing flexibility in optimizing reaction conditions for specific derivative synthesis.
From an impurity control perspective, the use of molecular iodine as a promoter rather than a stoichiometric oxidant helps minimize the formation of over-oxidized byproducts. The reaction conditions are sufficiently selective to tolerate various substituents on the aromatic ring of the starting hydrazide, including electron-donating alkyl and alkoxy groups as well as electron-withdrawing halogens and trifluoromethyl moieties. The robustness of the catalytic cycle ensures that side reactions such as polymerization or decomposition of the sensitive trifluoromethyl group are minimized, resulting in a cleaner crude reaction profile that simplifies the subsequent purification steps required to achieve high-purity pharmaceutical intermediates.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The operational simplicity of this protocol makes it highly accessible for laboratory and pilot plant settings, requiring only standard glassware and heating equipment without the need for specialized inert gas lines or gloveboxes. The procedure involves simply combining the trifluoroethyliminohydrazide substrate with molecular iodine in DMF, heating the mixture to the specified temperature range, and allowing the tandem cyclization to proceed to completion. Detailed standardized synthesis steps, including precise molar ratios, quenching procedures, and purification protocols, are outlined in the technical guide below to ensure reproducible results for process chemists.
- Combine molecular iodine and trifluoroethyliminohydrazide in an organic solvent system primarily composed of DMF.
- Heat the reaction mixture to 110-130°C under an air atmosphere for 10-15 hours to facilitate the tandem cyclization.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DMF-based synthesis route offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver of cost efficiency is the substitution of expensive, specialized C1 reagents with DMF, a commodity chemical that is produced on a massive global scale and is readily available from multiple suppliers at a fraction of the cost. This shift not only lowers the raw material expenditure but also mitigates supply risk, as the dependency on niche reagents with long lead times is effectively eliminated. Furthermore, the ability to run the reaction under air atmosphere removes the capital and operational expenses associated with maintaining strict inert gas systems, leading to significant overhead reductions in facility management.
- Cost Reduction in Manufacturing: The integration of the solvent and reactant functions into a single chemical entity dramatically improves the process mass intensity, reducing the total volume of materials required per kilogram of product. By avoiding the use of precious metal catalysts or complex ligand systems, the process eliminates the costly and time-consuming steps associated with metal scavenging and residual metal testing, which are critical bottlenecks in API production. The simplified workup procedure, which typically involves filtration and standard chromatography, further reduces labor costs and solvent consumption during the isolation phase, contributing to substantial overall cost savings.
- Enhanced Supply Chain Reliability: The reliance on DMF and molecular iodine, both of which are established bulk chemicals with stable supply chains, ensures consistent production continuity even during periods of market volatility. Unlike specialized reagents that may face shortages or quality fluctuations, these commoditized inputs offer predictable availability and pricing, allowing for more accurate long-term planning and inventory management. The robustness of the reaction conditions also means that the process is less susceptible to variations in environmental factors, ensuring reliable batch-to-batch consistency which is essential for maintaining regulatory compliance and customer trust.
- Scalability and Environmental Compliance: The absence of hazardous reagents and the use of a common solvent system facilitate easier scale-up from gram to ton quantities without the need for extensive process re-engineering. The reaction generates fewer hazardous byproducts compared to traditional methods, simplifying waste treatment and disposal procedures and aligning with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental footprint of the manufacturing process but also enhances the corporate sustainability metrics, which are becoming increasingly important criteria for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology, derived directly from the experimental data and beneficial effects reported in the patent documentation. These insights are intended to clarify the operational parameters and potential applications for R&D and production teams evaluating this methodology for their specific pipeline needs.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF acts as both the reaction solvent and the critical C1 carbon source, eliminating the need for separate, expensive formylating agents and simplifying the workup process.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, the process operates efficiently under standard air atmosphere conditions without the need for rigorous drying or inert gas protection, significantly reducing operational complexity.
Q: What types of substituents are tolerated on the aromatic ring?
A: The method demonstrates broad substrate scope, successfully accommodating electron-donating groups like alkyl and alkoxy, as well as electron-withdrawing groups such as halogens and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthetic routes in the development of next-generation therapeutics and agrochemicals. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like the DMF-mediated triazole synthesis can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities to guarantee the quality and consistency of every batch supplied to our global partners.
We invite you to collaborate with us to leverage this advanced technology for your specific project requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your development timelines and optimize your supply chain efficiency.
