Revolutionizing Quinazolinone Production: A Cost-Effective Iron-Catalyzed Route for Global Supply Chains
Revolutionizing Quinazolinone Production: A Cost-Effective Iron-Catalyzed Route for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize complex heterocyclic scaffolds that serve as the backbone for modern therapeutics. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These nitrogen-containing fused ring systems are ubiquitous in medicinal chemistry, known for their potent biological activities ranging from anti-cancer and anticonvulsant to antifungal properties. The strategic introduction of a trifluoromethyl group into these heterocycles further enhances their metabolic stability, lipophilicity, and bioavailability, making them highly desirable targets for drug discovery programs. This patent presents a transformative approach that shifts away from traditional, resource-intensive methods towards a streamlined, iron-catalyzed protocol that promises to redefine the economic and operational landscape for producing these critical pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has been fraught with significant chemical and economic challenges. Traditional literature methods predominantly rely on the cyclization of synthons containing the trifluoromethyl group, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide, anthranilic acid, or isatoic anhydride. While chemically valid, these conventional routes suffer from severe limitations that hinder their utility in large-scale manufacturing. The reaction conditions are often excessively harsh, requiring stringent anhydrous environments or extreme temperatures that pose safety risks and energy burdens. Furthermore, the starting materials, particularly specialized trifluoromethylating agents, can be prohibitively expensive and difficult to source in bulk quantities. Perhaps most critically, these older methods frequently exhibit narrow substrate scope and low yields, leading to substantial waste generation and inefficient use of raw materials, which directly impacts the cost of goods sold (COGS) for downstream API manufacturers.
The Novel Approach
In stark contrast to these legacy techniques, the methodology described in patent CN111675662B introduces a highly efficient and versatile synthetic strategy. By utilizing readily available trifluoroethylimidoyl chloride and isatin derivatives as the primary building blocks, this new route circumvents the need for costly anhydrides. The core innovation lies in the use of a cheap, earth-abundant iron catalyst (ferric chloride) to drive the cyclization. This approach not only drastically reduces the raw material costs but also significantly broadens the functional group tolerance, allowing for the synthesis of a diverse array of substituted quinazolinones. As illustrated in the structural diversity below, this method accommodates various substituents on the aromatic rings, enabling medicinal chemists to rapidly access a wide library of analogues for structure-activity relationship (SAR) studies without being constrained by synthetic feasibility.
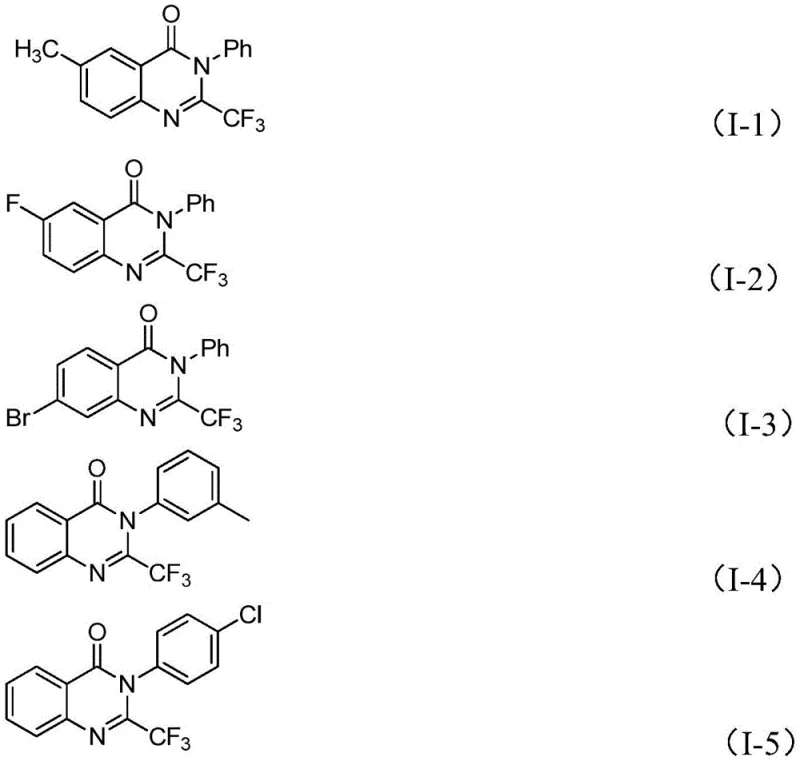
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The success of this transformation hinges on a sophisticated yet operationally simple catalytic cycle mediated by ferric chloride. The reaction mechanism initiates with an alkali-promoted formation of carbon-nitrogen bonds between the trifluoroethylimidoyl chloride and the isatin substrate. This initial step generates a trifluoroacetamidine intermediate, setting the stage for the subsequent ring closure. The presence of sodium hydride acts as a crucial base to deprotonate the isatin nitrogen, facilitating nucleophilic attack. Following this, the iron catalyst plays a pivotal role in promoting a decarbonylation event. This step is energetically demanding in the absence of a catalyst but proceeds smoothly under the influence of the Lewis acidic iron species. The final stage involves an intramolecular cyclization and isomerization to yield the thermodynamically stable 2-trifluoromethyl substituted quinazolinone core. This mechanistic pathway is robust enough to proceed even under air atmosphere, a rare feature for organometallic transformations that typically demand inert gas protection.
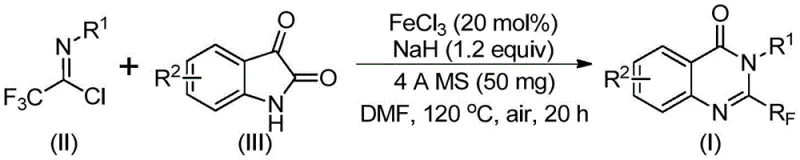
From an impurity control perspective, this mechanism offers distinct advantages. The high selectivity of the iron catalyst minimizes the formation of side products that often plague uncatalyzed thermal cyclizations. The use of 4A molecular sieves in the reaction mixture serves a dual purpose: it acts as a water scavenger to protect the moisture-sensitive imidoyl chloride and sodium hydride, and it helps drive the equilibrium towards product formation by removing trace water generated during the process. This results in a cleaner crude reaction profile, which simplifies the downstream purification workup. For R&D directors, understanding this mechanism is vital as it highlights the process's robustness; the tolerance for air and the use of common solvents like DMF suggest that the process is less sensitive to minor variations in operational parameters, thereby ensuring consistent batch-to-batch quality.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and purity. The process is designed to be operationally straightforward, avoiding the need for specialized high-pressure reactors or cryogenic conditions. The standard protocol involves a two-stage heating profile: an initial period at a moderate temperature to allow for the formation of the intermediate, followed by a higher temperature phase to drive the cyclization to completion. The choice of solvent is critical, with polar aprotic solvents like DMF proving superior in dissolving the ionic intermediates and facilitating the catalytic cycle. For those looking to replicate or scale this chemistry, the following guide outlines the standardized steps derived from the patent data to ensure optimal outcomes.
- Prepare the reaction mixture by adding ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative into an organic solvent such as DMF.
- Initiate the reaction at a lower temperature of 40°C for 8-10 hours to facilitate initial bond formation, then increase temperature to 120°C.
- Maintain the reaction at 120°C for 18-20 hours under air atmosphere to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this iron-catalyzed methodology represents a strategic opportunity to optimize the supply base for quinazolinone intermediates. The primary driver for cost reduction here is the substitution of expensive noble metal catalysts or specialized fluorinating reagents with commodity chemicals. Ferric chloride is produced on a massive industrial scale for water treatment and etching, making it incredibly inexpensive and universally available. This eliminates the supply chain volatility associated with precious metals like palladium or rhodium, whose prices can fluctuate wildly based on geopolitical factors. Furthermore, the starting materials—isatins and aromatic amines used to make the imidoyl chlorides—are bulk chemicals with established global supply chains, ensuring long-term availability and price stability for your manufacturing operations.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of high-cost inputs. By replacing expensive trifluoroacetic anhydride with cheaper imidoyl chlorides and utilizing a base metal catalyst, the direct material costs are significantly lowered. Additionally, the reaction's ability to proceed under air atmosphere removes the capital expenditure and operational cost associated with maintaining strict inert gas lines and glovebox conditions. The simplified post-treatment, which involves standard filtration and chromatography rather than complex metal scavenging steps required for Pd-catalyzed reactions, further reduces processing time and consumable costs, leading to substantial overall savings in the cost of goods.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available reagents. Unlike specialized fluorinating agents that may have single-source suppliers or long lead times, the key components of this reaction are produced by multiple chemical manufacturers globally. This multi-sourcing capability mitigates the risk of supply disruptions. Moreover, the robustness of the reaction conditions means that the synthesis is less prone to failure due to minor environmental variances, ensuring higher reliability in meeting production schedules. The scalability from gram to kilogram levels without changing the fundamental chemistry allows for a seamless transition from R&D to commercial supply, reducing the lead time for scaling up new drug candidates.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The use of iron, a non-toxic metal, reduces the burden of heavy metal waste disposal and lowers the risk of residual metal contamination in the final product, a critical specification for pharmaceutical APIs. The reaction efficiency and high yields mean less solvent waste per kilogram of product. While DMF is a regulated solvent, its high boiling point allows for efficient recovery and recycling in a closed-loop system. The overall process generates fewer hazardous byproducts compared to traditional methods, simplifying wastewater treatment and helping facilities maintain compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this route. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing pipeline.
Q: What are the primary advantages of using ferric chloride over traditional catalysts for quinazolinone synthesis?
A: Ferric chloride offers a substantial cost advantage as it is an abundant, non-precious metal catalyst compared to expensive palladium or rhodium systems. Furthermore, it demonstrates excellent functional group tolerance and eliminates the need for complex ligand systems, simplifying the post-reaction purification process and reducing heavy metal contamination risks in the final API intermediate.
Q: How does this new method improve upon conventional trifluoroacetic anhydride routes?
A: Conventional routes often rely on expensive and corrosive reagents like trifluoroacetic anhydride or ethyl trifluoroacetate, which can require harsh conditions and yield limited substrate scope. The patented method utilizes readily available trifluoroethylimidoyl chlorides and isatins, operating under milder conditions with higher yields and broader applicability across diverse aromatic substitutions.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the protocol is explicitly designed for scalability. The use of common solvents like DMF, inexpensive catalysts, and robust reaction conditions (tolerating air atmosphere) makes the transition from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial production highly feasible without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
The technological advancements detailed in patent CN111675662B underscore the immense potential of 2-trifluoromethyl quinazolinones as key building blocks for next-generation therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent chemistry into reliable commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the specific thermal profiles and solvent systems required for this iron-catalyzed cyclization. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of intermediate meets the exacting standards required for pharmaceutical development, guaranteeing consistency and quality for your drug discovery programs.
We invite you to leverage our technical expertise to accelerate your project timelines. Whether you require custom synthesis of specific analogues listed in the patent or need to scale up the core scaffold for clinical trials, our team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall development costs.
