Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds for High-Value Pharmaceutical Intermediates
Introduction to Novel Pyrrol-2-One Synthesis Technology
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a critical pharmacophore found in numerous bioactive natural products and therapeutic agents, necessitating robust and efficient synthetic methodologies for its construction. As detailed in patent CN112694430B, a groundbreaking preparation method has been developed that leverages palladium-catalyzed bis-carbonylation to assemble this complex heterocyclic core in a single operational step. This technology addresses the longstanding challenges associated with synthesizing these scaffolds, which are prevalent in molecules exhibiting significant antibacterial, hypoglycemic, and anticancer activities, such as Althiomycin, Glimepiride, and Isomalyngamide A. 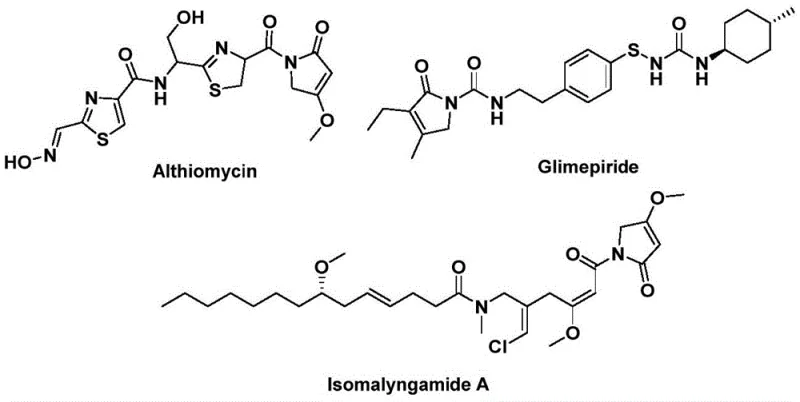 By utilizing readily available starting materials like propargyl amines and benzyl chlorides, this innovation not only streamlines the synthetic route but also significantly enhances the safety profile by avoiding the direct use of toxic carbon monoxide gas, thereby establishing a new benchmark for the reliable pharmaceutical intermediate supplier seeking to optimize their pipeline.
By utilizing readily available starting materials like propargyl amines and benzyl chlorides, this innovation not only streamlines the synthetic route but also significantly enhances the safety profile by avoiding the direct use of toxic carbon monoxide gas, thereby establishing a new benchmark for the reliable pharmaceutical intermediate supplier seeking to optimize their pipeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-containing heterocycles often relied on traditional carbonylation reactions that required the handling of high-pressure carbon monoxide gas, posing severe safety risks and requiring specialized equipment that increases capital expenditure. Furthermore, conventional multi-step syntheses to access the 1,5-dihydro-2H-pyrrol-2-one skeleton frequently suffered from low overall yields due to the accumulation of losses at each isolation stage, alongside the generation of substantial chemical waste. Many existing protocols also exhibited poor functional group tolerance, limiting the diversity of analogues that could be explored during the drug discovery phase, thus hindering the rapid iteration required in modern medicinal chemistry programs. The reliance on harsh reaction conditions often led to decomposition of sensitive substrates, resulting in complex impurity profiles that were difficult and costly to remove during downstream processing.
The Novel Approach
In stark contrast, the methodology disclosed in CN112694430B introduces a transformative one-pot strategy that utilizes phenyl 1,3,5-tricarboxylate as a safe and effective solid carbon monoxide substitute, effectively eliminating the hazards associated with gaseous CO. This novel approach enables the direct coupling of propargyl amines and benzyl chlorides under relatively mild thermal conditions (100-120°C) to achieve high conversion rates and excellent selectivity for the target pyrrol-2-one products. The reaction system demonstrates remarkable substrate compatibility, successfully accommodating a wide array of electronic and steric environments on both the amine and chloride components, which is crucial for generating diverse chemical libraries. 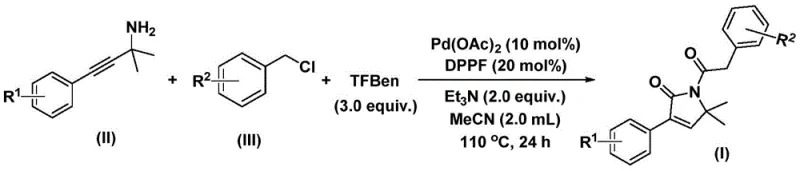 By consolidating multiple bond-forming events into a single vessel, this process drastically reduces solvent consumption, labor hours, and purification burdens, offering a streamlined pathway that is ideally suited for cost reduction in pharmaceutical intermediate manufacturing.
By consolidating multiple bond-forming events into a single vessel, this process drastically reduces solvent consumption, labor hours, and purification burdens, offering a streamlined pathway that is ideally suited for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle underpinning this transformation is a sophisticated sequence of organometallic steps initiated by the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride substrate. Following this activation, the first molecule of carbon monoxide, released in situ from the thermal decomposition of the phenyl 1,3,5-tricarboxylate additive, inserts into the benzyl-palladium intermediate to generate a reactive acyl-palladium species. This acyl intermediate then undergoes a nucleophilic attack or coordination with the propargyl amine, facilitating the formation of a five-membered ring palladium complex through an intramolecular cyclization event. Subsequently, a second carbon monoxide insertion occurs, expanding the metallacycle to a six-membered ring intermediate, which finally undergoes reductive elimination to release the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerate the active palladium catalyst for the next turnover. 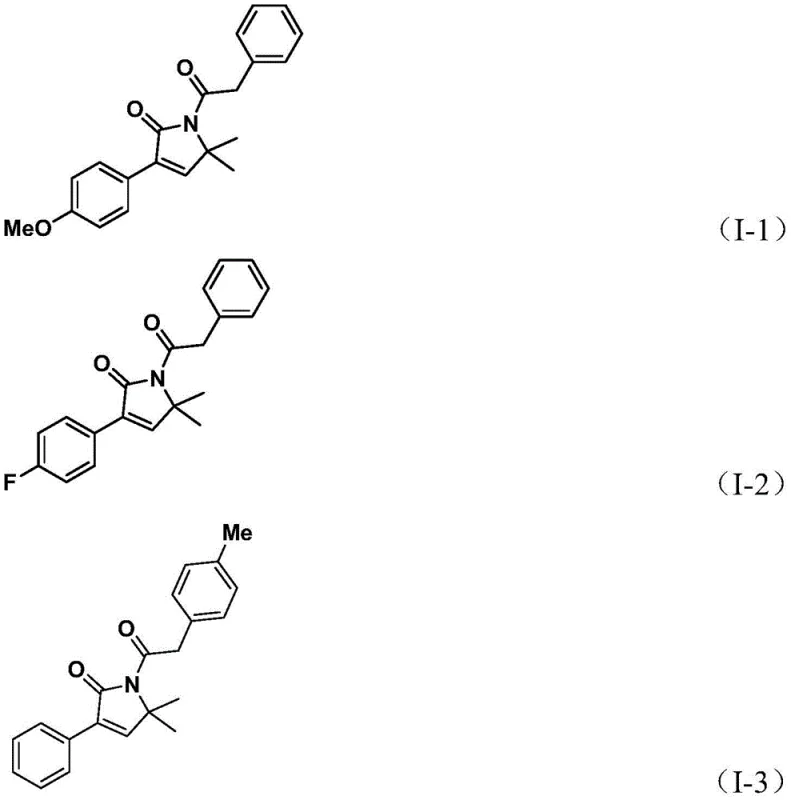 This intricate mechanism ensures high atom economy and precise control over the regioselectivity of the carbonyl groups within the final heterocyclic structure.
This intricate mechanism ensures high atom economy and precise control over the regioselectivity of the carbonyl groups within the final heterocyclic structure.
From an impurity control perspective, the use of a well-defined palladium catalyst system combined with the specific ligand 1,1'-bis(diphenylphosphino)ferrocene (DPPP) plays a pivotal role in suppressing side reactions such as homocoupling of the benzyl chloride or polymerization of the alkyne moiety. The choice of acetonitrile as the solvent further optimizes the solubility of all reaction components, ensuring a homogeneous reaction environment that promotes consistent kinetics and minimizes the formation of insoluble byproducts. The mild basicity provided by triethylamine is sufficient to neutralize the hydrochloric acid byproduct without promoting unwanted base-catalyzed degradation of the sensitive lactam ring. Consequently, the crude reaction mixture typically exhibits a clean profile, allowing for straightforward purification via standard silica gel chromatography to achieve high-purity 1,5-dihydro-2H-pyrrole-2-one compounds suitable for biological evaluation.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The practical execution of this synthesis involves charging a reaction vessel with palladium acetate, the DPPP ligand, triethylamine, the solid CO source, and the respective amine and chloride substrates in acetonitrile. The mixture is then heated to the specified temperature range and maintained for a duration sufficient to drive the reaction to completion, followed by a simple workup procedure involving filtration and chromatographic purification.
- Combine palladium acetate, DPPP ligand, triethylamine, phenyl 1,3,5-tricarboxylate (CO source), propargyl amine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 100-120°C and stir for 24 to 48 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers substantial strategic benefits by fundamentally altering the cost structure and risk profile of producing these valuable intermediates. The reliance on commodity chemicals such as benzyl chlorides and propargyl amines, which are abundantly available from global chemical suppliers, ensures a stable and resilient raw material supply chain that is less susceptible to market volatility compared to exotic or custom-synthesized precursors. Furthermore, the elimination of high-pressure gas infrastructure and the use of standard laboratory glassware for reaction setup significantly lowers the barrier to entry for manufacturing, allowing for flexible production scheduling across multiple facilities without the need for specialized reactor installations.
- Cost Reduction in Manufacturing: The consolidation of multiple synthetic steps into a single one-pot operation inherently reduces the consumption of solvents, reagents, and energy, leading to a significantly simplified process flow that drives down overall production costs. By avoiding the use of expensive and hazardous carbon monoxide gas cylinders and the associated safety monitoring systems, facilities can realize substantial capital and operational expenditure savings while maintaining a safer working environment. The high reaction efficiency and yield reported in the patent data imply that less raw material is wasted, directly improving the material throughput and reducing the cost per kilogram of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against varying electronic properties of substrates means that supply chains are not bottlenecked by the availability of highly specific, narrowly defined starting materials. Manufacturers can source generic benzyl chloride and amine derivatives from a wide network of vendors, mitigating the risk of single-source dependency and ensuring continuous production even if one supplier faces disruptions. The scalability of the process from milligram to multi-kilogram scales without significant re-optimization allows for seamless transition from clinical trial material supply to commercial manufacturing, guaranteeing timely delivery to downstream partners.
- Scalability and Environmental Compliance: The use of acetonitrile, a common and recyclable organic solvent, aligns well with modern green chemistry initiatives and simplifies waste management protocols compared to processes utilizing chlorinated or highly toxic solvents. The solid nature of the carbon monoxide source eliminates the regulatory burdens and permitting complexities associated with storing and handling large quantities of toxic gases, facilitating faster site approvals and smoother audits. Additionally, the high selectivity of the reaction minimizes the generation of hazardous byproducts, reducing the environmental footprint and disposal costs associated with chemical waste treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route, derived directly from the experimental data and scope defined in the patent literature.
Q: What are the key advantages of this palladium-catalyzed method over traditional carbonylation?
A: This method utilizes a solid carbon monoxide substitute (phenyl 1,3,5-tricarboxylate) instead of toxic CO gas, operates under milder conditions (100-120°C), and achieves high yields in a single step with excellent functional group tolerance.
Q: Which substrates are compatible with this synthesis route?
A: The process demonstrates broad substrate compatibility, accommodating various substituents on both the propargyl amine and benzyl chloride, including alkyl, alkoxy, halogen (F, Cl, Br), phenyl, and trifluoromethyl groups.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the protocol uses commercially available catalysts and solvents (acetonitrile), involves simple post-processing (filtration and chromatography), and avoids hazardous gas handling, making it highly scalable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines and reducing time-to-market for novel therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and robust. We are committed to delivering high-purity 1,5-dihydro-2H-pyrrole-2-one intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite potential partners to engage with our technical procurement team to discuss how this advanced catalytic technology can be integrated into your specific supply chain to achieve a Customized Cost-Saving Analysis. By collaborating with us, you gain access to specific COA data and comprehensive route feasibility assessments that will empower your R&D and operations teams to make informed decisions, ultimately securing a competitive advantage in the global marketplace for high-value pharmaceutical intermediates.
