Advanced Palladium-Catalyzed Synthesis of Bioactive 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112694430B, which discloses a highly efficient preparation method for 1,5-dihydro-2H-pyrrole-2-one compounds. This specific structural motif is not merely an academic curiosity; it is a privileged scaffold found in numerous potent natural products and therapeutic agents. For instance, the antibiotic Althiomycin and the hypoglycemic agent Glimepiride both rely on variations of this core structure for their biological efficacy. The ability to synthesize these cores rapidly and with high purity is therefore of paramount importance to R&D teams aiming to develop next-generation therapeutics. The disclosed technology leverages a palladium-catalyzed bis-carbonylation strategy, transforming simple starting materials into valuable intermediates in a single operational step.
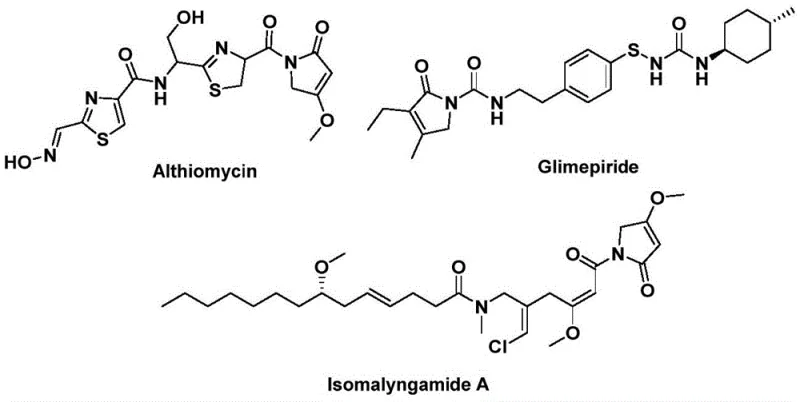
Traditionally, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has been fraught with synthetic challenges that hinder rapid drug discovery and process development. Conventional routes often require multi-step sequences involving harsh reagents, sensitive intermediates, or difficult purification protocols that degrade overall yield. Many existing methods suffer from poor atom economy or require specialized equipment to handle toxic carbon monoxide gas directly, posing significant safety and environmental hazards in a manufacturing setting. Furthermore, the substrate scope in older methodologies is frequently limited, failing to accommodate diverse functional groups that are essential for tuning the pharmacological properties of the final drug candidate. These limitations create bottlenecks in the supply chain, increasing lead times and costs for procurement managers who struggle to source high-quality intermediates reliably.
In stark contrast, the novel approach described in the patent utilizes a sophisticated yet operationally simple palladium-catalyzed system that overcomes these historical barriers. By employing a combination of palladium acetate and a bidentate phosphine ligand, specifically 1,1'-bis(diphenylphosphino)ferrocene (DPPF), the reaction achieves remarkable efficiency. The process uses a solid carbon monoxide substitute, eliminating the need for handling hazardous CO gas cylinders, which significantly enhances workplace safety and simplifies reactor setup. This method demonstrates exceptional functional group tolerance, successfully accommodating electron-donating and electron-withdrawing substituents on both the amine and chloride components. The result is a versatile platform technology that allows chemists to generate a wide library of derivatives for structure-activity relationship (SAR) studies without redesigning the synthetic route for each new analog.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation Cyclization
The success of this transformation lies in the intricate catalytic cycle that orchestrates the assembly of the five-membered lactam ring. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. This step is crucial as it activates the electrophile for subsequent carbonylation. Following this activation, the carbon monoxide surrogate releases CO in situ, which inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargyl amine, setting the stage for ring closure. The unique aspect of this mechanism is the bis-carbonylation event, where a second molecule of carbon monoxide is inserted before the final reductive elimination occurs. This double insertion is what constructs the specific carbonyl arrangement found in the 1,5-dihydro-2H-pyrrole-2-one core, distinguishing it from simpler carbonylation reactions.
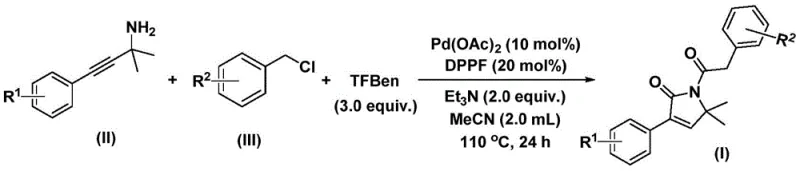
Controlling impurities in such complex catalytic cycles is a primary concern for quality assurance teams. The use of triethylamine as a base plays a dual role: it neutralizes the hydrochloric acid byproduct generated during the oxidative addition, preventing catalyst deactivation, and it helps drive the equilibrium towards product formation. The choice of acetonitrile as the solvent is also mechanistically significant, as it provides the ideal polarity to solubilize the ionic intermediates while maintaining the stability of the palladium complex. The reaction conditions, typically maintained at 110°C for 24 hours, are optimized to ensure complete consumption of the starting materials, thereby minimizing the presence of unreacted halides or amines in the crude mixture. This high conversion rate simplifies the downstream purification process, allowing for the isolation of the target compound with high purity specifications through standard silica gel chromatography.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, CO surrogate, base, and substrates in the correct stoichiometric ratios. The detailed standardized synthesis steps, including precise weighing, addition order, and workup procedures, are outlined below to guide process chemists in replicating these high-yielding results.
- Combine palladium catalyst, ligand, CO surrogate, base, propargyl amine, and benzyl chloride in an organic solvent.
- Heat the reaction mixture to 100-120°C and stir for 24-48 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material portfolio. By utilizing benzyl chlorides and propargyl amines, which are commodity chemicals available from multiple global suppliers, the risk of supply disruption is significantly mitigated. This reliance on abundant feedstocks ensures a stable and continuous supply of the critical intermediate, protecting the manufacturing schedule from the volatility often associated with exotic or custom-synthesized reagents. Furthermore, the elimination of gaseous carbon monoxide from the process removes a major logistical and safety hurdle, reducing the need for specialized gas handling infrastructure and lowering the overall capital expenditure required for production facilities.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by its high atom efficiency and reduced operational complexity. Because the reaction proceeds in a single pot with high conversion rates, the need for intermediate isolation and purification steps is eliminated, leading to substantial savings in solvent usage, labor hours, and waste disposal costs. The high yields reported across a broad range of substrates mean that less raw material is wasted, directly improving the cost of goods sold (COGS). Additionally, the use of a solid CO surrogate instead of high-pressure gas equipment lowers the barrier to entry for manufacturing, allowing for production in standard glass-lined reactors rather than expensive high-pressure autoclaves.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system translates directly into supply chain resilience. The tolerance for various functional groups means that the same core process can be used to manufacture a diverse array of derivatives without requiring extensive process re-validation for each new product. This flexibility allows manufacturers to respond quickly to changing market demands or clinical trial requirements. The simplicity of the post-processing workflow, involving basic filtration and chromatography, ensures that batch turnover times are minimized, enabling faster delivery of materials to downstream formulation teams and reducing inventory holding costs.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The avoidance of toxic CO gas and the use of relatively benign solvents like acetonitrile reduce the environmental footprint of the manufacturing process. The high selectivity of the reaction minimizes the formation of hazardous byproducts, simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations. As the process scales from kilogram to tonne quantities, these safety and environmental advantages become even more pronounced, facilitating a smoother path to commercial validation and regulatory approval.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries from our industry partners. These responses address specific concerns regarding reaction optimization, substrate scope, and scalability, providing a clear picture of what clients can expect when adopting this technology for their own pipelines.
Q: What are the key advantages of this Pd-catalyzed carbonylation method?
A: This method offers a one-step synthesis from readily available starting materials (propargyl amines and benzyl chlorides) with high reaction efficiency and excellent substrate compatibility, avoiding complex multi-step sequences.
Q: What represents the optimal reaction temperature and time?
A: According to the patent data, the optimal conditions involve heating the reaction mixture to 110°C for approximately 24 hours in acetonitrile solvent to achieve high yields.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available catalysts and simple post-processing steps like filtration and column chromatography, making it highly amenable to scale-up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new pharmaceuticals. Our team of expert process chemists has thoroughly analyzed the methodology described in patent CN112694430B and is fully prepared to leverage this advanced palladium-catalyzed route for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,5-dihydro-2H-pyrrole-2-one derivative we deliver meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your trusted partner in turning complex chemical challenges into commercial realities.
