Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
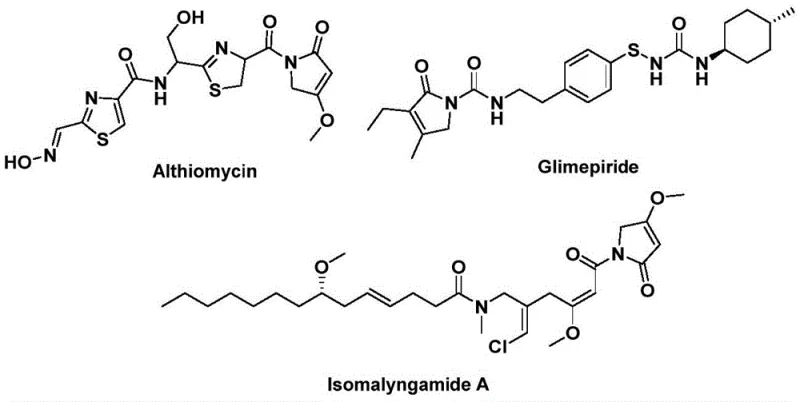
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for a myriad of bioactive natural products and therapeutic agents. As illustrated in the biological context, compounds such as Althiomycin exhibit significant antibacterial properties, while Glimepiride functions as a potent hypoglycemic agent, and Isomalyngamide A displays promising anticancer activity. Recognizing the critical importance of this heterocyclic system, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing propargyl amines and benzyl chlorides as accessible starting materials to construct the five-membered lactam ring with remarkable efficiency. By replacing hazardous gaseous carbon monoxide with a solid surrogate and employing mild reaction conditions, this technology not only enhances laboratory safety but also provides a robust platform for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines for complex bioactive molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has been fraught with synthetic inefficiencies that hinder large-scale manufacturing. Traditional routes often rely on multi-step sequences involving harsh cyclization conditions, the use of toxic reagents, or the handling of high-pressure carbon monoxide gas, which poses significant safety risks and infrastructure costs for industrial facilities. Furthermore, many existing methodologies suffer from limited substrate scope, failing to accommodate diverse functional groups without extensive protection-deprotection strategies, which drastically increases waste generation and reduces overall atom economy. These conventional processes frequently result in moderate yields and difficult purification profiles due to the formation of complex byproduct mixtures, creating bottlenecks in the supply chain for high-value active pharmaceutical ingredients. Consequently, there has been an urgent demand within the industry for a more direct, safe, and versatile synthetic route that can bridge the gap between academic discovery and commercial viability.
The Novel Approach
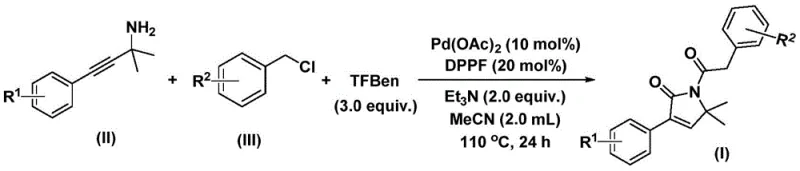
The innovative methodology disclosed in the patent data revolutionizes this landscape by introducing a one-pot palladium-catalyzed bis-carbonylation reaction that streamlines the synthesis into a single, highly efficient step. By utilizing benzyl chlorides and propargyl amines in the presence of a palladium catalyst and a solid carbon monoxide substitute, this process eliminates the need for dangerous gas cylinders and specialized high-pressure equipment. The reaction operates under relatively mild thermal conditions, typically between 100°C and 120°C, ensuring energy efficiency while maintaining high conversion rates. This approach exhibits exceptional functional group tolerance, allowing for the seamless integration of electron-rich and electron-deficient substituents on the aromatic rings, thereby expanding the chemical space accessible to process chemists. The simplicity of the operation, combined with the use of cheap and readily available starting materials, positions this technology as a superior alternative for the cost reduction in pharmaceutical intermediate manufacturing, enabling faster time-to-market for new drug candidates.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensures high regioselectivity and yield. The cycle initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzyl-palladium intermediate. Subsequently, a molecule of carbon monoxide, liberated in situ from the phenol 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This electrophilic acyl intermediate then undergoes nucleophilic attack by the propargyl amine, facilitating the formation of a five-membered ring palladium complex. A second insertion of carbon monoxide follows, expanding the coordination sphere to a six-membered ring palladium intermediate, which ultimately undergoes reductive elimination to release the final 1,5-dihydro-2H-pyrrol-2-one product and regenerate the active catalyst. This elegant cascade avoids the accumulation of unstable intermediates and drives the reaction to completion with high fidelity.
From an impurity control perspective, the mechanism inherently minimizes side reactions through the rapid consumption of reactive intermediates. The use of a solid CO source ensures a steady, controlled release of carbon monoxide, preventing the formation of homocoupling byproducts that often plague gas-phase carbonylations. Furthermore, the choice of ligand, specifically 1,1'-bis(diphenylphosphino)ferrocene (DPPF), stabilizes the palladium center throughout the catalytic cycle, preventing catalyst decomposition and metal precipitation which could contaminate the final product. The reaction conditions are optimized to favor the desired bis-carbonylation pathway over competing mono-carbonylation or polymerization routes, resulting in a clean crude profile that simplifies downstream purification. This level of mechanistic control is crucial for producing high-purity pharmaceutical intermediates that meet stringent regulatory standards for residual metals and organic impurities.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The practical execution of this synthesis is designed for scalability and ease of handling in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, solid CO surrogate, and the two primary substrates in an organic solvent such as acetonitrile. The mixture is then heated to the optimal temperature range and stirred for a defined period to ensure full conversion. Following the reaction, a straightforward workup procedure involving filtration and silica gel treatment allows for the isolation of the target compound. For detailed operational parameters and specific stoichiometric ratios required to replicate these results, please refer to the standardized synthesis guide below.
- Combine palladium acetate, DPPP ligand, solid CO surrogate, base, propargyl amine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to ensure complete conversion.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards this catalytic system addresses critical pain points related to raw material availability, operational safety, and process economics. By relying on commodity chemicals like benzyl chlorides and propargyl amines, manufacturers can mitigate supply risks associated with exotic or custom-synthesized reagents. Moreover, the elimination of high-pressure gas infrastructure reduces capital expenditure and maintenance costs, while the simplified one-pot nature of the reaction decreases labor hours and utility consumption per batch. These factors collectively contribute to a more resilient and cost-effective supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available starting materials and the avoidance of costly high-pressure equipment. The high atom efficiency of the bis-carbonylation reaction minimizes waste disposal costs, while the high yields reported reduce the cost of goods sold by maximizing output per unit of input. Additionally, the simplified purification workflow lowers the consumption of chromatography media and solvents, further driving down operational expenses without compromising quality.
- Enhanced Supply Chain Reliability: Sourcing stability is significantly improved as the key reagents, including the palladium catalyst and benzyl chloride derivatives, are standard industrial chemicals with robust global supply networks. The process does not depend on fragile cold chains or specialized gas delivery services, ensuring continuous production capability even during logistical disruptions. This reliability allows for better inventory planning and reduces the risk of production stoppages due to material shortages.
- Scalability and Environmental Compliance: The transition from bench scale to commercial production is facilitated by the mild reaction conditions and the absence of hazardous gaseous reagents. The process generates less hazardous waste compared to traditional methods, aligning with increasingly strict environmental regulations and sustainability goals. The ability to run the reaction in common solvents like acetonitrile simplifies solvent recovery and recycling, supporting green chemistry initiatives and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational details. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing a realistic overview of what can be achieved. Understanding these nuances is essential for effective process development and risk assessment prior to scale-up.
Q: What is the primary advantage of this carbonylation method over traditional cyclization?
A: This method utilizes a solid carbon monoxide surrogate and readily available benzyl chlorides, avoiding the need for high-pressure CO gas and complex multi-step sequences, thereby enhancing operational safety and efficiency.
Q: Does this protocol tolerate diverse functional groups on the aromatic rings?
A: Yes, the process demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens and trifluoromethyl moieties.
Q: What are the typical purification requirements for the crude product?
A: The post-treatment is streamlined; the crude reaction mixture typically requires filtration followed by standard silica gel column chromatography to achieve high-purity isolates suitable for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to multi-ton manufacturing is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,5-dihydro-2H-pyrrole-2-one intermediate meets the highest industry standards for potency and impurity profiles. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including efficient metal scavenging and solvent recovery systems.
We invite potential partners to engage with our technical procurement team to discuss how this innovative carbonylation technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations.
