Advanced Palladium Catalysis for Efficient Production of Bioactive Lactone Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly alpha-methylene-gamma-butyrolactones, which serve as critical pharmacophores in numerous bioactive molecules exhibiting anti-tumor and antibacterial properties. A groundbreaking development detailed in Chinese patent CN111635437B introduces a novel palladium complex featuring an ortho-carborane benzimidazole structure that fundamentally alters the synthetic landscape for these valuable intermediates. This innovation addresses the longstanding challenges of harsh reaction conditions and expensive precursor materials by enabling a highly efficient, one-pot multicomponent reaction under remarkably mild circumstances. As a leading entity in the chemical sector, understanding the implications of such catalytic advancements is crucial for maintaining a competitive edge in the production of high-purity pharmaceutical intermediates. The technology not only streamlines the synthetic route but also opens new avenues for cost-effective manufacturing processes that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the alpha-methylene-gamma-butyrolactone skeleton has relied heavily on transition metal-catalyzed cyclization reactions of intramolecular enynes or Rh-catalyzed processes involving 1,6-enynes. While these methods have been effective in academic settings, they present significant hurdles for large-scale industrial application due to the high cost and limited availability of specialized starting materials like enynes and alkynoic acids. Furthermore, these conventional protocols typically necessitate elevated temperatures and rigorous reaction conditions, which increase energy consumption and complicate process safety management. The reliance on precious metal catalysts that often lack robustness under varied substrate conditions further exacerbates the economic burden, making the final API intermediates prohibitively expensive for widespread therapeutic use. Additionally, the multi-step nature of many traditional syntheses leads to cumulative yield losses and generates substantial chemical waste, conflicting with the industry's push towards more sustainable manufacturing practices.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN111635437B utilizes a uniquely designed palladium complex to catalyze the direct coupling of simple, commercially abundant alkynes, carboxylic acids, and alkenes. This approach eliminates the need for pre-functionalized, expensive substrates, allowing manufacturers to utilize basic feedstocks such as phenylacetylene, formic acid, and styrene derivatives. The reaction proceeds efficiently at room temperature in common solvents like toluene, drastically reducing the energy footprint associated with heating and cooling cycles. 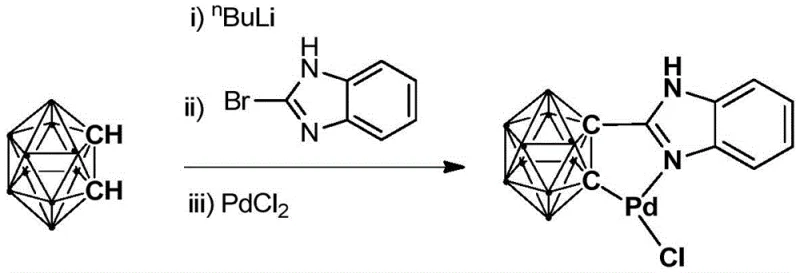 The versatility of this catalytic system is demonstrated by its ability to tolerate various functional groups, including electron-donating and electron-withdrawing substituents on the aromatic rings, without compromising yield or selectivity. By consolidating multiple bond-forming events into a single operational step, this novel methodology significantly simplifies the workflow, reduces solvent usage, and minimizes the generation of by-products, thereby offering a superior alternative for the cost reduction in fine chemical manufacturing.
The versatility of this catalytic system is demonstrated by its ability to tolerate various functional groups, including electron-donating and electron-withdrawing substituents on the aromatic rings, without compromising yield or selectivity. By consolidating multiple bond-forming events into a single operational step, this novel methodology significantly simplifies the workflow, reduces solvent usage, and minimizes the generation of by-products, thereby offering a superior alternative for the cost reduction in fine chemical manufacturing.
Mechanistic Insights into Carborane-Stabilized Palladium Catalysis
The exceptional performance of this catalytic system can be attributed to the unique electronic and steric properties imparted by the ortho-carborane benzimidazole ligand framework. The carborane cage, known for its three-dimensional aromaticity and electron-withdrawing characteristics, stabilizes the palladium center against decomposition and aggregation, which are common failure modes in homogeneous catalysis. This stabilization allows the catalyst to maintain high turnover numbers even under the relatively mild conditions employed in the reaction. The benzimidazole moiety acts as a robust chelating group, ensuring the metal remains coordinated throughout the catalytic cycle, facilitating the precise activation of the alkyne and alkene substrates. 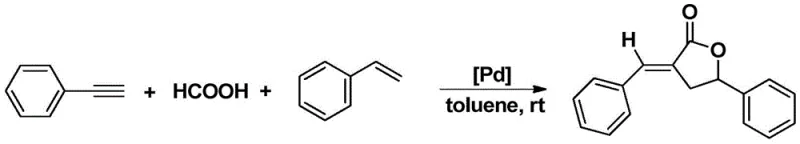 Mechanistically, the palladium complex likely initiates the reaction through oxidative addition or coordination with the alkyne, followed by insertion of the alkene and subsequent carboxylation with the carboxylic acid source. The rigid geometry of the ligand environment helps control the regioselectivity of the addition, ensuring the formation of the desired alpha-methylene-gamma-butyrolactone isomer with high fidelity. This level of control is essential for pharmaceutical applications where isomeric purity is a critical quality attribute, as it reduces the burden on downstream purification processes.
Mechanistically, the palladium complex likely initiates the reaction through oxidative addition or coordination with the alkyne, followed by insertion of the alkene and subsequent carboxylation with the carboxylic acid source. The rigid geometry of the ligand environment helps control the regioselectivity of the addition, ensuring the formation of the desired alpha-methylene-gamma-butyrolactone isomer with high fidelity. This level of control is essential for pharmaceutical applications where isomeric purity is a critical quality attribute, as it reduces the burden on downstream purification processes.
Furthermore, the impurity profile generated by this process is notably cleaner compared to traditional high-temperature methods. The mild reaction conditions prevent the thermal degradation of sensitive functional groups and minimize side reactions such as polymerization of the alkene or oligomerization of the alkyne. The high selectivity observed, with yields reaching up to 95% in optimized examples, indicates that the catalyst effectively suppresses competing pathways. For R&D teams focused on process development, this means a more predictable and reproducible synthesis that requires less extensive optimization of work-up procedures. The ability to achieve such high purity directly from the reaction mixture simplifies the isolation of the final product, often requiring only standard column chromatography or crystallization, which translates to significant time and resource savings during the scale-up phase.
How to Synthesize Ortho-Carborane Benzimidazole Palladium Complex Efficiently
The preparation of this advanced catalyst is designed to be straightforward and scalable, utilizing readily available reagents and standard laboratory techniques. The synthesis begins with the lithiation of ortho-carborane at low temperatures, followed by reaction with bromobenzimidazole to install the organic ligand framework. Subsequent treatment with palladium chloride completes the metallation process, yielding the active catalyst species. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during preparation.
- Dropwise addition of n-BuLi solution to a carborane solution at low temperature (-80 to -75°C) followed by stirring and warming to room temperature.
- Addition of bromobenzimidazole to the reaction mixture and continuing the reaction at room temperature for 6 to 8 hours.
- Introduction of PdCl2 to the system, reacting for 3 to 5 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers compelling strategic benefits that extend beyond mere technical novelty. The shift from expensive, specialized precursors to commodity chemicals like styrene and phenylacetylene fundamentally alters the cost structure of producing these lactone intermediates. By leveraging widely available raw materials, manufacturers can mitigate the risks associated with supply chain disruptions for niche reagents and negotiate better pricing due to the high market liquidity of the inputs. This transition supports a more resilient supply chain capable of withstanding market volatility while ensuring continuous production schedules for critical drug substances.
- Cost Reduction in Manufacturing: The elimination of costly enyne substrates and the removal of energy-intensive heating requirements result in substantial operational expenditure savings. The one-pot nature of the reaction reduces the number of unit operations, lowering labor costs and minimizing solvent consumption, which directly impacts the overall cost of goods sold. Furthermore, the high catalytic efficiency means that lower loading of the precious metal palladium is required, optimizing the utilization of this expensive resource and reducing the financial burden of metal recovery or disposal.
- Enhanced Supply Chain Reliability: Utilizing common chemical feedstocks ensures that production is not bottlenecked by the availability of exotic starting materials. The robustness of the catalyst allows for flexible manufacturing windows, as the reaction does not require strict temperature control infrastructure beyond ambient conditions. This reliability facilitates better inventory management and shorter lead times for high-purity pharmaceutical intermediates, enabling faster response to market demand fluctuations and reducing the need for large safety stocks of finished goods.
- Scalability and Environmental Compliance: The mild conditions and simplified work-up procedures make this process highly amenable to scale-up from kilogram to multi-ton production without significant engineering challenges. The reduction in waste generation and energy usage aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential liabilities associated with hazardous waste disposal. This green chemistry profile enhances the corporate sustainability image and ensures long-term viability of the manufacturing process in a regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed technology. These insights are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on its practical application.
Q: What are the primary advantages of this palladium complex over traditional catalysts?
A: Unlike traditional methods requiring expensive enyne substrates and heating, this complex enables one-pot synthesis from simple alkynes, carboxylic acids, and alkenes under mild room temperature conditions with yields up to 95%.
Q: Is the catalyst stable for long-term storage and industrial use?
A: Yes, the patent highlights that the ortho-carborane benzimidazole structure imparts excellent physical, chemical, and thermal stability to the palladium center, ensuring consistent performance.
Q: What types of substrates are compatible with this catalytic system?
A: The system demonstrates broad substrate scope, successfully catalyzing reactions with various substituted phenylacetylenes and styrenes, including those with methyl, methoxy, and chloro groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Methylene-Gamma-Butyrolactone Supplier
The technological breakthrough represented by this ortho-carborane palladium complex underscores the rapid evolution of synthetic methodologies available for producing complex pharmaceutical building blocks. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is backed by rigorous QC labs and stringent purity specifications, ensuring that every batch of intermediate meets the exacting standards required by global regulatory bodies. We understand that transitioning to a new catalytic process requires confidence in the supplier's ability to deliver consistent quality and volume.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your production volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for cost efficiency and supply security. Partner with us to leverage this cutting-edge chemistry for your next generation of therapeutic products.
