Advanced Catalytic Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A recent technological breakthrough disclosed in patent CN113307778A introduces a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are ubiquitous in medicinal chemistry, renowned for their ability to enhance metabolic stability, lipophilicity, and bioavailability when incorporated into drug candidates. Notable examples include blockbuster drugs such as Sitagliptin, where the triazole core plays a pivotal role in biological activity. The introduction of the trifluoromethyl group further amplifies these physicochemical properties, making the development of streamlined synthetic access to these motifs a high priority for R&D teams globally.
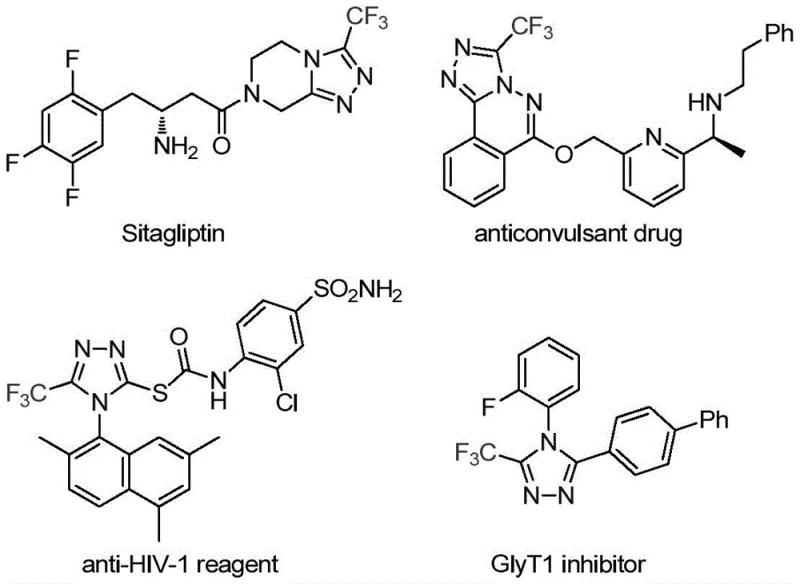
This novel methodology addresses the longstanding challenges associated with constructing fully substituted or specifically 3,4-disubstituted 1,2,4-triazole rings. By leveraging a unique dual-catalytic system involving molybdenum hexacarbonyl and cuprous acetate, the process achieves high reaction efficiency under remarkably mild thermal conditions. For procurement managers and supply chain directors, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity scaffolds with reduced lead times and optimized cost structures. The versatility of the reaction allows for the design of various functionalized derivatives, ensuring that diverse chemical space can be explored without compromising on process safety or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant operational and safety hurdles that hinder large-scale manufacturing. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical pathways often necessitate harsh reaction conditions, including extreme temperatures or the use of strong bases, which can lead to poor atom economy and the generation of substantial chemical waste. Furthermore, alternative multi-component reactions utilizing diazonium salts and trifluorodiazoethane pose severe safety risks due to the explosive nature of diazo compounds, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure. The reliance on such hazardous reagents not only complicates the regulatory approval process but also creates bottlenecks in the supply chain, as the availability of safe, high-quality diazo precursors can be inconsistent.
The Novel Approach
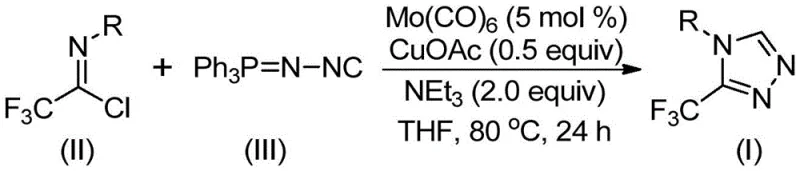
In stark contrast to these legacy methods, the technology described in patent CN113307778A offers a paradigm shift by utilizing a copper-promoted [3+2] cycloaddition strategy co-catalyzed by molybdenum. This innovative route employs trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks, reacting them in the presence of Mo(CO)6 and CuOAc. The reaction proceeds smoothly in common organic solvents like THF at moderate temperatures ranging from 70°C to 90°C. This mild thermal profile drastically reduces energy consumption and minimizes the formation of thermal degradation byproducts, resulting in a cleaner crude reaction profile. The elimination of explosive diazonium salts and the use of commercially available, stable starting materials significantly de-risk the manufacturing process, making it an ideal candidate for cost reduction in API manufacturing where safety and consistency are paramount.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this transformation lies in the synergistic interaction between the molybdenum and copper species, which orchestrates a complex yet efficient cascade of bond-forming events. Mechanistically, the molybdenum hexacarbonyl serves as a crucial metal activator for the functionalized isonitrile, likely forming a transient metal-isocyanide complex that enhances the nucleophilicity or electrophilicity of the carbon center. Simultaneously, the cuprous acetate promotes the initial [3+2] cycloaddition between the activated isonitrile and the trifluoroethylimidoyl chloride. This step constructs the five-membered triazole ring intermediate with high regioselectivity. Following the ring closure, the system facilitates the elimination of triphenylphosphine oxide, driven by the presence of water in the reaction matrix, to yield the final aromatic 3-trifluoromethyl-1,2,4-triazole product. This intricate dance of coordination chemistry ensures that the reaction proceeds with high fidelity, minimizing the formation of regioisomers that are often difficult to separate in traditional triazole syntheses.
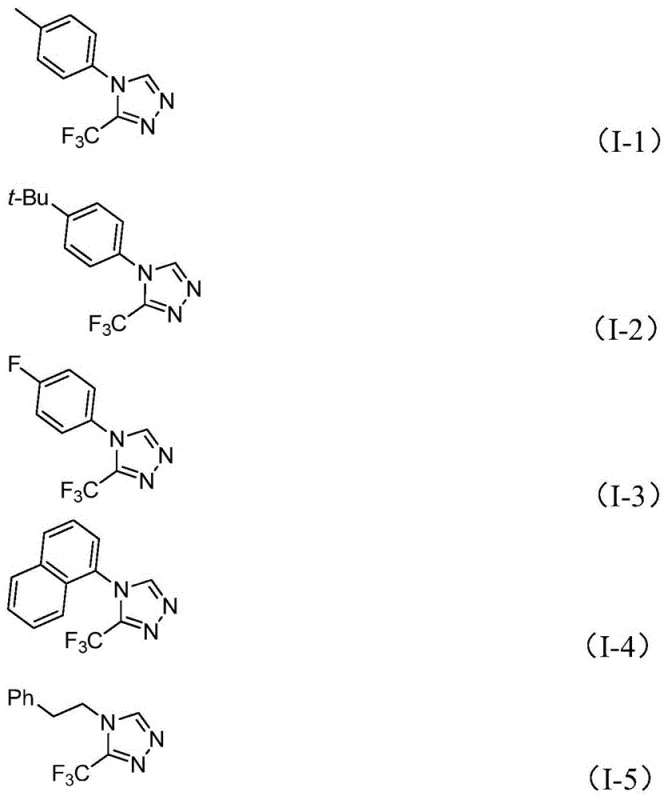
From an impurity control perspective, the mildness of the reaction conditions is a decisive factor in maintaining high product purity. Harsh acidic or basic conditions typical of older methods often lead to hydrolysis of sensitive functional groups or polymerization of reactive intermediates. In this new protocol, the neutral to slightly basic environment provided by triethylamine preserves the integrity of diverse functional groups on the aromatic ring. The patent data indicates excellent tolerance for electron-donating groups like methyl and tert-butyl, as well as electron-withdrawing substituents such as fluoro, chloro, and nitro groups. This broad substrate scope implies that the process generates a consistent impurity profile regardless of the specific R-group employed, simplifying the downstream purification strategy. For R&D directors, this predictability is invaluable, as it reduces the time spent on method development for impurity identification and control during the scale-up phase.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The operational simplicity of this synthesis makes it accessible for both laboratory discovery and pilot plant operations. The procedure involves a straightforward one-pot setup where all reagents are combined in a Schlenk tube or standard reactor. The use of molecular sieves helps to manage moisture levels, which is critical for the stability of the imidoyl chloride and the efficiency of the catalytic cycle. After the reaction period of 18 to 30 hours, the workup is uncomplicated, involving simple filtration to remove metal residues and inorganic salts, followed by standard silica gel chromatography. This ease of execution lowers the barrier for adoption, allowing process chemists to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies. Detailed standardized synthesis steps see the guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), trifluoroethylimidoyl chloride, and functionalized isonitrile (NIITP) in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-1,2,4-triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling strategic advantages that extend beyond mere chemical elegance. The primary driver for adoption is the substantial optimization of the cost of goods sold (COGS) through the utilization of commodity chemicals. The catalyst system relies on cuprous acetate and molybdenum hexacarbonyl, which are significantly more affordable than the precious metal catalysts (such as palladium or rhodium) often required in cross-coupling reactions. Furthermore, the base triethylamine and the solvent THF are bulk commodities with stable global supply chains, insulating the manufacturing process from the volatility associated with exotic reagents. This shift to earth-abundant metals and common solvents translates directly into a more resilient supply chain, reducing the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the elimination of expensive and hazardous reagents. By avoiding the use of diazonium salts and complex hydrazine derivatives, manufacturers save significantly on raw material costs and the specialized infrastructure required to handle them safely. The mild reaction temperatures (70-90°C) also imply lower energy consumption compared to high-temperature reflux conditions, contributing to a smaller carbon footprint and reduced utility bills. Additionally, the high reaction efficiency reported in the patent suggests that less solvent and stationary phase are required for purification, further driving down the operational expenses associated with waste disposal and chromatography media.
- Enhanced Supply Chain Reliability: The robustness of the starting materials ensures a steady flow of production. Trifluoroethylimidoyl chlorides and functionalized isonitriles are readily available from multiple global vendors, preventing single-source dependency. The tolerance of the reaction to various functional groups means that a single manufacturing platform can produce a wide array of derivatives without needing to requalify the entire process for each new analog. This flexibility allows supply chain planners to respond more agilely to fluctuating demand for different API intermediates, ensuring continuity of supply even when specific drug candidates enter late-stage clinical trials or commercial launch phases unexpectedly.
- Scalability and Environmental Compliance: The patent explicitly notes that the method can be expanded to gram-level reactions with high efficiency, indicating a clear path toward kilogram and tonne-scale production. The simplified post-treatment process, which avoids complex extractions or crystallizations, facilitates easier scale-up in standard stainless steel reactors. From an environmental standpoint, the avoidance of heavy metal waste streams associated with stoichiometric oxidants or reducers simplifies effluent treatment. The use of THF, a solvent that can be efficiently recovered and recycled, aligns with modern green chemistry principles, helping pharmaceutical companies meet their increasingly stringent sustainability goals and regulatory compliance requirements regarding waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this catalytic system. These insights are derived directly from the experimental data and mechanistic proposals within the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing. Understanding these nuances is critical for assessing the feasibility of integrating this technology into existing production lines.
Q: What are the key advantages of the Mo/Cu co-catalytic system over traditional methods?
A: The Mo/Cu co-catalytic system operates under significantly milder conditions (70-90°C) compared to traditional harsh cyclization methods. It utilizes readily available starting materials like trifluoroethylimidoyl chloride and functionalized isonitriles, avoiding the safety hazards associated with diazonium salts or complex hydrazine derivatives, thereby offering a safer and more efficient pathway for industrial scale-up.
Q: What is the substrate scope for the R group in this synthesis?
A: The methodology demonstrates excellent tolerance for various substituents on the aryl ring of the R group. It successfully accommodates electron-donating groups such as methyl, methoxy, and tert-butyl, as well as electron-withdrawing groups like fluoro, chloro, and nitro. Additionally, naphthyl and phenethyl groups are compatible, allowing for the synthesis of a diverse library of 3,4-disubstituted 1,2,4-triazole derivatives.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly states that the method can be expanded to gram-level reactions with high efficiency. The use of inexpensive catalysts like cuprous acetate and common solvents like THF, combined with simple post-processing steps involving filtration and column chromatography, makes the process highly amenable to commercial scale-up for API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Mo/Cu co-catalyzed synthesis for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facility is equipped with state-of-the-art rigorous QC labs and analytical instrumentation capable of verifying the stringent purity specifications required for GMP-grade intermediates. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process chemistry team is ready to optimize this specific route to maximize yield and minimize impurities for your specific application.
We invite you to collaborate with us to leverage this advanced technology for your pipeline projects. Whether you require custom synthesis of specific 3,4-disubstituted triazole analogs or full-scale commercial manufacturing, our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data for our standard catalog items or to discuss route feasibility assessments for your proprietary molecules. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
