Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazole Intermediates for Commercial Scale
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are pivotal for enhancing metabolic stability and bioavailability in modern drug design. As highlighted in patent CN113307778A, a significant breakthrough has been achieved in the preparation of 3-trifluoromethyl substituted 1,2,4-triazole compounds, a structural core found in prominent medications such as Sitagliptin and various anticonvulsant agents. This novel approach addresses the longstanding challenges associated with traditional synthetic routes by employing a dual-catalytic system that operates under remarkably mild conditions. The strategic integration of molybdenum hexacarbonyl and cuprous acetate facilitates a highly efficient cycloaddition reaction, offering a streamlined pathway that is both economically viable and environmentally considerate for the production of high-value pharmaceutical intermediates.
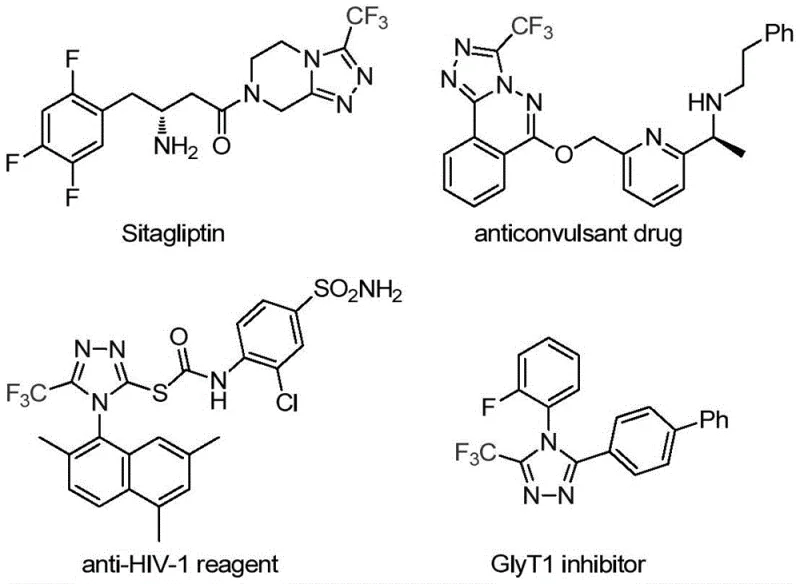
The limitations of conventional methods for synthesizing trifluoromethyl-substituted 1,2,4-triazoles have long hindered efficient commercial manufacturing, often necessitating the use of hazardous reagents or extreme reaction parameters. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones, processes that frequently suffer from poor atom economy and difficult purification protocols. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane introduce significant safety risks due to the explosive nature of diazo compounds, making them less than ideal for large-scale industrial application. These legacy methods often result in complex impurity profiles that require extensive downstream processing, thereby inflating production costs and extending lead times for critical API intermediates.
In stark contrast, the novel approach detailed in the provided patent data utilizes a sophisticated Mo/Cu co-catalytic system that fundamentally reshapes the synthetic landscape for these heterocycles. By reacting trifluoroethylimidoyl chloride with functionalized isonitriles (NIITP) in the presence of molybdenum hexacarbonyl and cuprous acetate, the process achieves high conversion rates at temperatures between 70°C and 90°C. This methodology eliminates the need for unstable diazonium salts and harsh hydrazine derivatives, replacing them with stable, commercially available starting materials that are easy to handle and store. The reaction mechanism involves the formation of a metal complex followed by a copper-promoted [3+2] cycloaddition, which efficiently constructs the five-membered triazole ring with exceptional regioselectivity and minimal byproduct formation.
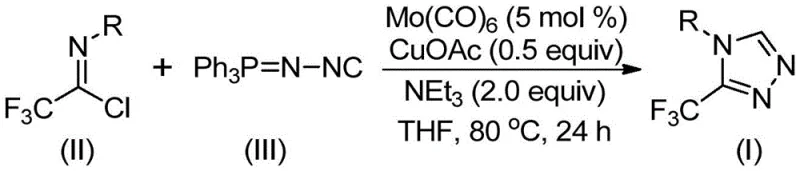
Mechanistic insights into this Mo/Cu co-catalyzed cyclization reveal a synergistic interaction between the two metal centers that is critical for activating the relatively inert isonitrile functionality. The molybdenum hexacarbonyl acts as a potent metal activator, coordinating with the functionalized isonitrile to lower the energy barrier for the subsequent nucleophilic attack. Simultaneously, the cuprous acetate promotes the [3+2] cycloaddition of the activated isonitrile to the trifluoroethylimidoyl chloride, facilitating the rapid assembly of the triazole core. Following the ring closure, the system effectively eliminates triphenylphosphine oxide under the influence of trace water, driving the equilibrium towards the final 3-trifluoromethyl-substituted product. This precise control over the reaction pathway ensures that side reactions, such as polymerization of the isonitrile or hydrolysis of the imidoyl chloride, are kept to an absolute minimum, resulting in a clean crude reaction mixture.
From an impurity control perspective, the mild thermal conditions (70-90°C) play a pivotal role in maintaining the integrity of sensitive functional groups on the aromatic rings. Unlike high-temperature pyrolysis methods that can lead to decomposition or rearrangement, this catalytic protocol preserves the substitution pattern of the starting aryl amines, whether they contain electron-withdrawing nitro groups or electron-donating methoxy groups. The use of triethylamine as a base further aids in scavenging acidic byproducts without promoting unwanted elimination reactions. Consequently, the resulting impurity profile is significantly simplified, allowing for straightforward purification via standard silica gel column chromatography. This level of chemical fidelity is essential for pharmaceutical manufacturers who must adhere to stringent regulatory guidelines regarding genotoxic impurities and residual solvents in final drug substances.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis of these valuable heterocyclic compounds is designed to be operationally simple, requiring standard laboratory equipment and readily accessible reagents to ensure reproducibility across different manufacturing sites. The process begins with the precise weighing of molybdenum hexacarbonyl, cuprous acetate, and triethylamine, which are combined with the specific trifluoroethylimidoyl chloride and functionalized isonitrile substrates in an anhydrous organic solvent such as THF. The reaction mixture is then heated to the optimized temperature range and stirred for a period of 18 to 30 hours, allowing sufficient time for the catalytic cycle to reach completion. Detailed standardized synthetic steps, including specific molar ratios and workup procedures, are outlined in the guide below to assist process chemists in replicating these high-yielding results.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70°C to 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits by mitigating risks associated with raw material availability and process safety. The reliance on cheap and easily obtainable starting materials, such as aromatic amines and trifluoroacetic acid derivatives, ensures a stable supply chain that is less susceptible to market volatility compared to specialized diazo reagents. Furthermore, the elimination of hazardous explosives from the process flow drastically reduces the regulatory burden and insurance costs associated with manufacturing facilities, while the high reaction efficiency minimizes waste generation and solvent consumption. This translates directly into a more sustainable and cost-effective manufacturing model that aligns with modern green chemistry principles and corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of inexpensive catalysts like cuprous acetate and triethylamine, which replace costly noble metals or complex ligand systems often required in similar transformations. By avoiding the multi-step synthesis of unstable intermediates and reducing the need for extensive purification due to cleaner reaction profiles, the overall cost of goods sold (COGS) for the final triazole intermediate is substantially lowered. Additionally, the high atom economy of the cycloaddition reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal fees and maximizing resource utilization efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method contributes to a more resilient supply chain by utilizing commodity chemicals that are widely available from multiple global suppliers, thereby reducing the risk of single-source dependency. The mild reaction conditions allow for the use of standard stainless steel reactors without the need for specialized Hastelloy lining or extreme pressure ratings, enabling flexible production scheduling across diverse manufacturing assets. This operational flexibility ensures consistent delivery timelines and the ability to rapidly scale up production volumes in response to fluctuating market demand for downstream pharmaceutical products.
- Scalability and Environmental Compliance: The patent data confirms that the reaction can be successfully expanded to gram-level scales with maintained efficiency, indicating a clear pathway for commercial scale-up to kilogram and tonnage quantities without significant re-optimization. The simplified post-treatment procedure, involving basic filtration and chromatography, reduces the complexity of waste streams and facilitates easier compliance with environmental discharge regulations. Moreover, the avoidance of toxic hydrazine derivatives and explosive diazo compounds creates a safer working environment for plant operators and reduces the liability associated with handling dangerous substances in a large-scale industrial setting.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this trifluoromethyl triazole synthesis method, providing clarity for R&D teams evaluating its potential for their specific pipelines. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for process development decisions. Understanding these nuances is critical for assessing the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the primary advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods requiring harsh hydrazinolysis or dangerous diazonium salts, this patent (CN113307778A) utilizes mild conditions (70-90°C) and readily available starting materials, significantly improving operational safety and substrate tolerance.
Q: What is the substrate scope for the R group in this trifluoromethyl triazole synthesis?
A: The method demonstrates excellent versatility, accommodating phenethyl groups as well as substituted or unsubstituted aryl groups with various substituents including alkyl, alkoxy, halogens, and nitro groups at ortho, meta, or para positions.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent explicitly states that the reaction efficiency is high and the process can be easily expanded to gram-level reactions, providing a solid foundation for subsequent scale-up to kilogram or tonnage production for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics, and we possess the technical expertise to bring complex synthetic routes like this Mo/Cu co-catalyzed method to fruition. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required by global regulatory bodies, providing you with a reliable partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering superior value and innovation in pharmaceutical intermediate supply.
