Advanced Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly those containing nitrogen-rich rings like 1,2,4-triazoles which are ubiquitous in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a novel preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift from laborious multi-step syntheses to a streamlined, metal-free oxidative cyclization protocol. By leveraging a catalytic system composed of tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP) in the presence of diphenylphosphoric acid, this method achieves high conversion rates under relatively mild thermal conditions. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: the ability to access high-purity pharmaceutical intermediates with drastically reduced operational complexity and without the burden of removing toxic heavy metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiency and harsh reaction requirements. Traditional literature methods typically rely on quinoline-2-formic acid as the starting material, necessitating a cumbersome five-step reaction sequence to arrive at the target structure. This legacy approach suffers from a dismal total yield of approximately 17%, making it economically unviable for large-scale manufacturing. Furthermore, these conventional routes often demand severe reaction conditions, including strict anhydrous and oxygen-free environments, which require specialized equipment like gloveboxes or Schlenk lines. The reliance on such stringent parameters not only inflates capital expenditure but also introduces significant bottlenecks in supply chain continuity, as any breach in inert atmosphere can lead to batch failure. Additionally, the use of transition metal catalysts in older methodologies often necessitates complex downstream purification processes to meet regulatory limits on heavy metal impurities in final drug substances.
The Novel Approach
In stark contrast, the methodology described in patent CN113307790B utilizes readily available and inexpensive starting materials, specifically 2-methylquinoline and trifluoroethylimide hydrazide, to construct the triazole ring in a single operational step. This innovative route employs a TBAI/TBHP promoted oxidative cyclization that proceeds efficiently at temperatures between 80°C and 100°C. Crucially, the reaction does not require protection from air or moisture, allowing it to be conducted under ambient atmospheric conditions which dramatically simplifies reactor setup and operation. The absence of heavy metal catalysts is a major regulatory and environmental win, eliminating the need for expensive scavenging resins or complex extraction protocols to remove metal traces. This streamlined approach not only boosts the reaction yield significantly—demonstrating up to 97% in optimized examples—but also broadens the substrate scope, allowing for the introduction of diverse substituents on both the quinoline and phenyl rings to tune the physicochemical properties of the final intermediate.
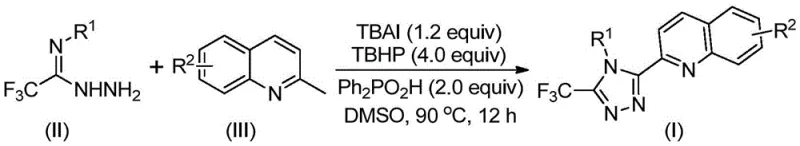
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological advancement lies in the synergistic interaction between the iodide salt and the peroxide oxidant. Mechanistically, the tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP) work in tandem to effect the oxidation of the methyl group on the 2-methylquinoline substrate. This initial oxidation converts the methyl group into a reactive 2-quinoline carbaldehyde intermediate in situ. Once formed, this aldehyde undergoes a condensation reaction with the trifluoroethylimide hydrazide to generate a dehydrated hydrazone intermediate. This transient species is then subjected to further oxidative iodination, followed by an intramolecular electrophilic substitution and subsequent aromatization to forge the final 1,2,4-triazole ring system. The patent suggests that the reaction may also proceed via a free radical pathway, facilitated by the homolytic cleavage of the peroxide bond, which generates radical species capable of abstracting hydrogen atoms from the methyl group. This dual possibility of ionic and radical mechanisms ensures robust reactivity across a wide range of electronic environments on the substrate.
From an impurity control perspective, the use of diphenylphosphoric acid as an additive plays a critical role in stabilizing the reaction intermediates and promoting the cyclization step. The specific choice of DMSO as the preferred solvent is also mechanistically significant; as a polar aprotic solvent, it effectively dissolves both the organic substrates and the ionic catalyst (TBAI), facilitating the necessary ion-pair interactions for the oxidation cycle. The high tolerance for functional groups observed in the patent examples—ranging from electron-donating methoxy groups to electron-withdrawing nitro and halogen substituents—indicates that the catalytic cycle is highly selective for the desired C-H activation and N-N bond formation, minimizing the formation of side products such as over-oxidized acids or polymerized tars. This selectivity is paramount for ensuring that the crude product profile is clean, thereby reducing the load on downstream purification units and ensuring consistent quality for the reliable pharmaceutical intermediate supplier.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow the generalized protocol established in the patent examples. The process begins by charging a reaction vessel with the requisite molar ratios of tetrabutylammonium iodide, aqueous tert-butyl peroxide, diphenylphosphoric acid, trifluoroethylimide hydrazide, and the specific 2-methylquinoline derivative. The mixture is suspended in an organic solvent, with DMSO being the optimal choice for maximizing conversion efficiency. The reaction is then heated to the specified temperature range and maintained for the duration required to reach full conversion, typically between 8 to 14 hours depending on the specific substituents. Detailed standardized synthetic steps for specific derivatives are outlined below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP), diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours under ambient atmospheric conditions without the need for inert gas protection.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The shift from a five-step sequence to a one-pot transformation fundamentally alters the cost structure of manufacturing these valuable heterocycles. By collapsing multiple unit operations into a single reactor charge, the process drastically reduces the consumption of solvents, energy, and labor hours associated with intermediate isolation and purification. This consolidation of steps directly translates to substantial cost savings in pharmaceutical intermediate manufacturing, allowing companies to offer more competitive pricing to their downstream clients while maintaining healthy margins. Furthermore, the elimination of expensive transition metal catalysts removes a significant line item from the bill of materials, as noble metals like palladium or rhodium are no longer required to drive the coupling reaction.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the utilization of commodity chemicals as starting materials. 2-methylquinoline and trifluoroacetic acid derivatives are widely available in the global market at low cost, unlike the specialized quinoline-2-formic acid required by older methods. Additionally, the avoidance of heavy metal catalysts means that manufacturers do not need to invest in costly metal scavenging technologies or endure the yield losses associated with rigorous metal removal processes. The simplified post-treatment, which involves basic filtration and standard column chromatography, further lowers the operational expenditure by reducing the complexity of the work-up phase. These factors combine to create a highly cost-effective production model that enhances the overall profitability of the supply chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this reaction protocol. Because the reaction does not require anhydrous or oxygen-free conditions, it can be performed in standard glass-lined or stainless steel reactors without the need for specialized inert gas blanketing systems. This flexibility reduces the risk of batch failures caused by equipment leaks or operator error regarding atmosphere control. Moreover, the starting materials are stable and commercially accessible, mitigating the risk of raw material shortages that often plague specialized reagent markets. The ability to run the reaction under ambient pressure and temperature ranges that are easily achievable in standard facilities ensures that production can be scaled up or shifted between different manufacturing sites with minimal requalification effort, ensuring continuous supply for critical drug development programs.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this process aligns well with modern green chemistry principles. The absence of toxic heavy metals simplifies waste stream management, as the effluent does not require complex treatment to remove hazardous metal ions before discharge. The use of TBHP as an oxidant generates tert-butanol as a byproduct, which is relatively benign and easier to handle than the stoichiometric waste generated by chromium or manganese-based oxidants used in traditional oxidations. The high atom economy of the cyclization step, combined with the high yields reported in the patent (often exceeding 80%), means that less raw material is wasted as unreacted starting material or side products. This efficiency not only reduces the environmental footprint but also facilitates easier regulatory approval for the manufacturing process, as the impurity profile is cleaner and more predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this technology into existing production workflows.
Q: What are the primary advantages of this synthesis method over traditional routes?
A: Unlike traditional methods requiring quinoline-2-formic acid and five reaction steps with a low total yield of 17%, this novel approach utilizes cheap 2-methylquinoline and trifluoroethylimide hydrazide in a single pot. It eliminates the need for toxic heavy metal catalysts and严苛 anhydrous/oxygen-free conditions, significantly simplifying operations and improving overall yield up to 97%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the reaction can be easily expanded to gram-scale and offers strong potential for future large-scale production. The use of commercially available, inexpensive reagents like TBAI and TBHP, along with simple post-treatment procedures like filtration and column chromatography, supports robust commercial scalability.
Q: What is the role of diphenylphosphoric acid in this reaction?
A: Diphenylphosphoric acid acts as a crucial additive in the catalytic system. Along with TBAI and TBHP, it promotes the oxidative cyclization reaction. The patent notes that using this specific additive contributes to relatively inexpensive processing and high reaction yields compared to other potential additives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to stay competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the methodology presented in patent CN113307790B and is fully prepared to leverage this metal-free oxidative cyclization strategy for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole intermediate we deliver meets the highest standards of quality and consistency required by top-tier drug developers.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss your project needs,索取 specific COA data for our reference standards, and obtain comprehensive route feasibility assessments. Let us help you navigate the complexities of heterocyclic synthesis and secure a reliable supply of high-quality intermediates for your next generation of therapeutics.
