Advanced Gem-Difluoroalkene Production: Scaling Metal-Free Catalysis for Pharmaceutical Intermediates with Unmatched Purity and Supply Chain Resilience
Patent CN114409515B introduces a groundbreaking methodology for synthesizing gem-difluoroalkene compounds through a metal-free hydrodefluorination process that represents a significant advancement in organic synthesis technology. This innovative approach addresses longstanding challenges in the production of fluorinated building blocks essential for pharmaceutical development, offering a more efficient pathway to these valuable intermediates. The patent details a reaction system utilizing diphenylphosphine oxide as a reducing agent and cesium carbonate as a base in ethyl acetate solvent under mild thermal conditions (70°C), which collectively enable superior functional group tolerance and regioselectivity compared to conventional methods. This development is particularly significant given the growing demand for fluorinated compounds in drug discovery pipelines where their unique properties enhance metabolic stability and bioavailability of therapeutic molecules. The process demonstrates remarkable versatility across diverse substrate classes while maintaining high purity standards (>98%) required for pharmaceutical applications, positioning it as a transformative solution for manufacturers seeking reliable access to these critical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
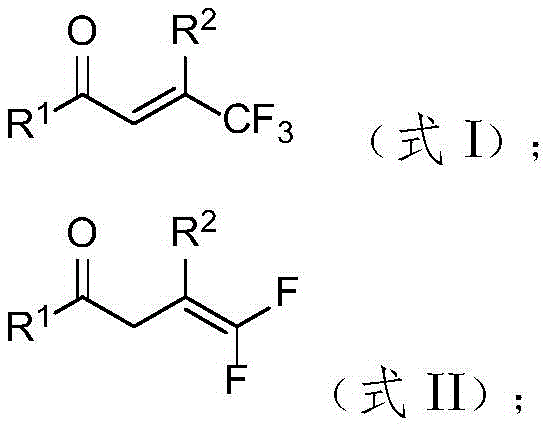
Traditional approaches to synthesizing gem-difluoroalkenes primarily rely on classic reactions such as Wittig and Horner-Wadsworth-Emmons methodologies, which typically require harsh alkaline conditions that severely limit substrate compatibility and functional group tolerance. These conventional methods often struggle with sensitive functional groups commonly found in complex pharmaceutical intermediates, necessitating extensive protection-deprotection strategies that increase both cost and processing time. The narrow substrate scope of existing techniques creates significant bottlenecks in developing new fluorinated compounds particularly when dealing with sterically hindered or electronically diverse molecular architectures. Furthermore many established processes involve transition metal catalysts that introduce contamination risks requiring additional purification steps to meet stringent pharmaceutical quality standards thereby complicating supply chain logistics and increasing overall production costs. The cumulative effect of these limitations has constrained widespread adoption of gem-difluoroalkenes despite their recognized value in enhancing drug properties such as metabolic stability and lipophilicity.
The Novel Approach
The patented methodology overcomes these limitations through an elegant metal-free hydrodefluorination process that operates under remarkably mild conditions (70°C in ethyl acetate) while maintaining exceptional functional group tolerance across diverse substrate classes. By utilizing diphenylphosphine oxide as a reducing agent and cesium carbonate as a base in the presence of water as an additive this innovative system achieves high regioselectivity without requiring strong alkaline environments or transition metal catalysts. The process demonstrates remarkable versatility with various substituents including halogenated methylated methoxylated cyanated nitrated trifluoromethylated ester-substituted phenyl groups naphthyl thiophene methyl biphenyl and styryl moieties significantly expanding accessible chemical space for fluorinated intermediates. Crucially elimination of metal catalysts removes need for costly metal removal steps while ensuring product purity exceeding 98% directly addressing key concerns for pharmaceutical manufacturers regarding trace metal contamination in final drug substances.
Mechanistic Insights into Diphenylphosphine Oxide-Mediated Hydrodefluorination
The reaction mechanism involves sophisticated interplay between diphenylphosphine oxide water and cesium carbonate facilitating selective defluorination while maintaining structural integrity of sensitive functional groups. Process begins with activation of trifluoromethyl group through coordination with diphenylphosphine oxide serving as both reducing agent and stabilizing ligand during defluorination sequence. Water plays critical role as proton source enabling hydrodefluorination step while suppressing unwanted side reactions through moderating effect on reaction environment. Cesium carbonate functions as mild base maintaining optimal pH conditions without causing decomposition of sensitive substrates or products. Precise stoichiometric balance between components creates synergistic effect driving high-yielding conversion to desired gem-difluoroalkene products while minimizing byproduct formation.
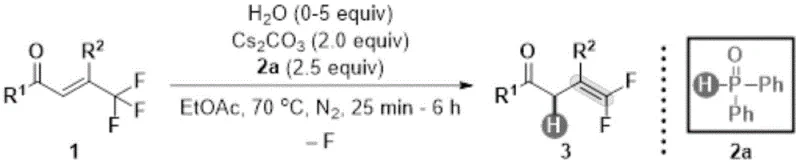
Exceptional purity (>98%) achieved stems from inherent selectivity and mild operating conditions preventing common side reactions such as over-reduction or isomerization plaguing conventional methods. Regioselectivity maintained through careful control of reaction parameters including temperature solvent choice and water content creating optimal environment for selective defluorination without affecting other functional groups present in molecule. This precise control over reaction pathways ensures consistent product quality across different substrate classes while eliminating need for extensive purification procedures otherwise required to remove metal catalysts or byproducts from traditional synthetic routes.
How to Synthesize Gem-Difluoroalkene Compounds Efficiently
This patented methodology represents significant advancement in synthesis of gem-difluoroalkene compounds offering pharmaceutical manufacturers reliable pathway to these valuable intermediates with superior purity yield characteristics compared to conventional approaches. Process leverages carefully optimized combination of diphenylphosphine oxide as reducing agent cesium carbonate as base water as additive in ethyl acetate solvent under mild thermal conditions achieving high regioselectivity functional group tolerance. Detailed standardized synthesis procedures developed based on extensive experimental validation across diverse substrate classes ensuring consistent results from laboratory scale through commercial production. Following step-by-step guide provides comprehensive instructions for implementing this innovative hydrodefluorination process in industrial settings.
- Prepare the reaction mixture by combining trifluoromethyl olefin compound (1 equiv), diphenylphosphine oxide (2.5 equiv), cesium carbonate (2 equiv), and water (5 equiv) in ethyl acetate solvent under nitrogen atmosphere.
- Stir the reaction mixture at precisely controlled temperature of 70°C for duration between 25 minutes to 6 hours to facilitate selective hydrodefluorination while maintaining optimal regioselectivity.
- Quench the reaction with saturated ammonium chloride solution, extract with ethyl acetate (3×20mL), wash organic phase with brine, dry over sodium sulfate, concentrate under vacuum, and purify by silica gel column chromatography using ethyl acetate/petroleum ether gradient elution.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative hydrodefluorination process addresses critical pain points in procurement supply chain management of fluorinated pharmaceutical intermediates delivering significant operational improvements without compromising quality reliability. Elimination of transition metal catalysts removes major supply chain vulnerabilities associated with metal price volatility availability constraints simultaneously reducing purification complexity associated costs. Mild reaction conditions enable seamless integration into existing manufacturing facilities without requiring specialized equipment extensive process modifications accelerating time-to-market for new drug candidates incorporating these valuable fluorinated building blocks. Furthermore exceptional functional group tolerance expands range accessible molecular architectures providing greater flexibility drug design while maintaining consistent high-quality output required regulatory compliance.
- Cost Reduction in Manufacturing: Metal-free nature eliminates expensive transition metal catalysts associated removal steps accounting significant portions production costs fluorinated intermediate synthesis. Utilizing readily available reagents like diphenylphosphine oxide cesium carbonate under mild conditions achieves substantial cost savings through reduced raw material expenses simplified purification requirements lower energy consumption compared conventional high-pressure high-temperature methods. Elimination metal contamination risks reduces quality control costs associated rigorous metal testing protocols required pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Use commercially available stable reagents long shelf lives significantly improves supply chain resilience compared processes requiring specialized unstable catalysts facing availability issues. Robustness reaction across diverse substrates ensures consistent product quality regardless minor variations raw material specifications reducing batch failures associated production delays. Reliability enhanced by process compatibility standard manufacturing equipment eliminating dependencies specialized infrastructure creating single points failure supply chain.
- Scalability and Environmental Compliance: Straightforward scale-up potential laboratory commercial production demonstrated consistent high yields different batch sizes without requiring significant process modifications. Elimination toxic metal catalysts reduces environmental impact simplifies waste treatment procedures aligning increasingly stringent environmental regulations lowering disposal costs. Mild operating conditions contribute improved workplace safety profiles compared traditional methods involving harsh reagents extreme reaction parameters.
Frequently Asked Questions (FAQ)
Following questions address common concerns regarding implementation benefits patented hydrodefluorination technology gem-difluoroalkene synthesis based extensive experimental data multiple substrate classes scale-up trials conducted industrial manufacturing conditions.
Q: What makes this hydrodefluorination process superior to conventional methods for gem-difluoroalkene synthesis?
A: Unlike traditional methods requiring strong alkaline conditions with limited substrate scope, this novel metal-free process operates under mild conditions (70°C in ethyl acetate) with excellent functional group tolerance across diverse substituents including halogenated phenyls, naphthyls, thiophenes, and styryl groups while maintaining >98% purity essential for pharmaceutical applications.
Q: How does the elimination of transition metals impact supply chain reliability for pharmaceutical intermediates?
A: The metal-free nature eliminates expensive catalyst procurement challenges and removes critical purification steps required to eliminate trace metals from final products. This simplification significantly enhances production reliability while ensuring consistent high-purity output that meets stringent pharmaceutical quality standards without additional quality control burdens.
Q: What is the scalability potential of this gem-difluoroalkene synthesis method?
A: The process demonstrates exceptional scalability due to its mild operating parameters (70°C), standard solvent system (ethyl acetate), and compatibility with conventional manufacturing equipment. Consistent high yields across diverse substrates confirm robustness from laboratory scale through commercial production volumes while maintaining stringent purity specifications required by regulatory authorities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Difluoroalkene Supplier
Our patented hydrodefluorination technology represents significant advancement production high-purity gem-difluoroalkene intermediates essential modern pharmaceutical development pipelines NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required global regulatory authorities Our state-of-the-art manufacturing facilities equipped rigorous QC labs capable meeting even most demanding analytical requirements fluorinated compounds used active pharmaceutical ingredients This combination innovative chemistry manufacturing excellence positions us ideal partner pharmaceutical companies seeking reliable access critical building blocks
We invite you contact our technical procurement team request specific COA data route feasibility assessments particular application needs Our experts provide Customized Cost-Saving Analysis demonstrating how patented technology optimize supply chain ensuring consistent high-quality output drug development programs
