Advanced Metal-Free Synthesis of Gem-Difluoroalkenes for High-Purity Pharmaceutical Intermediates
Introduction to Next-Generation Fluorination Technology
The landscape of fluorinated organic synthesis is undergoing a significant transformation, driven by the increasing demand for bioisosteres that enhance metabolic stability and lipophilicity in drug candidates. Patent CN114409515B introduces a groundbreaking methodology for the preparation of gem-difluoroalkene compounds, a class of molecules pivotal in modern medicinal chemistry and materials science. This technology leverages a unique hydrodefluorination strategy that converts readily available trifluoromethyl alkene precursors into valuable gem-difluoro products under remarkably mild conditions. By utilizing diphenylphosphine oxide as a reducing agent in conjunction with cesium carbonate and a catalytic amount of water, this process achieves high regioselectivity without the need for expensive transition metal catalysts. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more sustainable and cost-efficient manufacturing pathways for complex fluorinated intermediates.
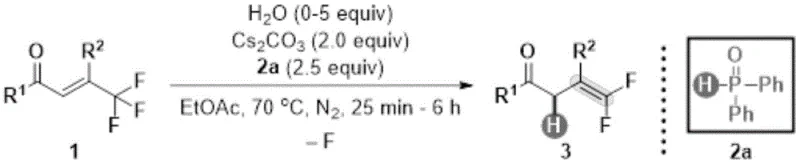
The significance of this invention lies not only in its chemical elegance but also in its practical applicability to industrial settings. Traditional methods for constructing carbon-fluorine bonds often involve hazardous reagents, extreme temperatures, or multi-step sequences that erode overall yield. In contrast, the disclosed method operates in common solvents like ethyl acetate at moderate temperatures around 70°C, making it inherently safer and easier to implement in existing production facilities. The broad substrate scope demonstrated in the patent, covering various substituted phenyl and heteroaryl groups, suggests that this technology can be adapted for a wide array of pharmaceutical and agrochemical building blocks. As we delve deeper into the technical specifics, it becomes clear that this approach addresses long-standing pain points regarding functional group tolerance and process scalability in fluorine chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of gem-difluoroalkenes has relied heavily on classical olefination reactions such as the Wittig or Horner-Wadsworth-Emmons protocols. While these methods are well-established in academic literature, they present substantial challenges when translated to commercial manufacturing environments. A primary limitation is the requirement for strong alkaline conditions, which can be incompatible with base-sensitive functional groups frequently found in advanced drug intermediates. Furthermore, these traditional routes often necessitate the preparation of specialized phosphorus ylides or phosphonates, adding extra synthetic steps and increasing the overall cost of goods. The use of strong bases also increases the risk of side reactions, such as polymerization or decomposition of the starting materials, leading to lower yields and difficult purification processes. Additionally, many conventional fluorination strategies rely on stoichiometric amounts of hazardous reagents or expensive transition metals, creating significant environmental and safety burdens that modern green chemistry initiatives seek to eliminate.
The Novel Approach
The methodology described in CN114409515B offers a compelling alternative by employing a direct hydrodefluorination mechanism that bypasses the limitations of classical olefination. Instead of building the double bond from scratch, this approach strategically modifies an existing trifluoromethyl alkene scaffold, effectively removing one fluorine atom and replacing it with a hydrogen atom. This transformation is mediated by diphenylphosphine oxide, a stable and non-toxic organophosphorus compound, acting as the terminal reductant. The reaction system is further optimized by the inclusion of cesium carbonate as a mild base and water as a critical additive, which together facilitate the selective cleavage of the carbon-fluorine bond. This metal-free protocol not only simplifies the reaction setup by avoiding air-sensitive catalysts but also ensures excellent functional group tolerance, allowing for the preservation of sensitive moieties like esters, nitriles, and halides. The result is a streamlined, one-pot procedure that delivers high-purity products with minimal waste generation.
Mechanistic Insights into Metal-Free Hydrodefluorination
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring consistent quality in large-scale production. The reaction proceeds through a concerted pathway where diphenylphosphine oxide serves as both a hydrogen source and an oxygen acceptor, driving the thermodynamic equilibrium towards the formation of the gem-difluoroalkene. The presence of cesium carbonate is essential for activating the phosphine oxide species, likely generating a reactive phosphinate intermediate that attacks the electron-deficient trifluoromethyl group. Interestingly, the role of water cannot be overstated; experimental optimization revealed that adding approximately 5 equivalents of water dramatically improves reaction efficiency compared to anhydrous conditions. This suggests that water molecules participate in the proton transfer network, stabilizing transition states and facilitating the departure of the fluoride ion as a cesium fluoride salt. The mild basicity of cesium carbonate ensures that the reaction environment remains neutral enough to prevent degradation of acid-sensitive substrates while being sufficiently basic to promote the redox cycle.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or transition-metal-catalyzed alternatives. The absence of free radical initiators minimizes the formation of oligomeric byproducts that often plague fluorination reactions. Moreover, the high regioselectivity observed ensures that the double bond geometry is preserved, typically yielding the E-isomer exclusively, which is critical for maintaining the biological activity of the final pharmaceutical agent. The byproduct of the reaction, diphenylphosphinic acid, is a solid that can be easily removed during the aqueous workup or silica gel chromatography, leaving the desired product with high purity levels exceeding 98%. This clean reaction profile reduces the burden on downstream purification units, directly translating to higher throughput and lower solvent consumption in the manufacturing plant. For process chemists, this level of predictability and cleanliness is invaluable when defining the design space for regulatory filings.
How to Synthesize Gem-Difluoroalkenes Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and minimize cycle time. The standard protocol involves charging a reactor with the trifluoromethyl alkene substrate, diphenylphosphine oxide, and cesium carbonate in ethyl acetate, followed by the controlled addition of water. The mixture is then heated to 70°C under an inert nitrogen atmosphere to prevent oxidation of the phosphine species. Reaction progress can be monitored via HPLC or GC, with typical completion times ranging from 25 minutes to 6 hours depending on the electronic nature of the substituents on the aromatic rings. Upon completion, the reaction is quenched with saturated ammonium chloride solution, and the product is extracted into the organic phase. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale are provided in the structured guide below, ensuring reproducibility across different manufacturing sites.
- Combine trifluoromethyl alkene substrate with diphenylphosphine oxide (2.5 equiv) and cesium carbonate (2.0 equiv) in ethyl acetate solvent.
- Add water (5.0 equiv) as a reaction promoter to facilitate the hydrodefluorination process under nitrogen atmosphere.
- Stir the reaction mixture at 70°C for 25 minutes to 6 hours, then quench and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible benefits that extend beyond simple chemical yield improvements. The most significant advantage is the drastic reduction in raw material costs associated with eliminating precious metal catalysts. Traditional cross-coupling or fluorination methods often rely on palladium, rhodium, or iridium complexes, which are not only expensive but also subject to volatile market pricing and supply constraints. By switching to a system based on cesium carbonate and diphenylphosphine oxide, manufacturers can secure a more stable and predictable cost structure for their key intermediates. Furthermore, the use of ethyl acetate as the primary solvent aligns with green chemistry principles, reducing the environmental compliance costs associated with disposing of chlorinated or aromatic solvents. This shift supports corporate sustainability goals while simultaneously lowering the total cost of ownership for the production process.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for expensive scavenging resins and rigorous heavy metal testing, which are mandatory for pharmaceutical grade materials. This simplification of the purification train significantly lowers operational expenditures and reduces the time required for quality control release. Additionally, the high atom economy of the hydrodefluorination reaction means less waste is generated per kilogram of product, further driving down disposal costs and improving the overall process mass intensity metrics.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, including cesium carbonate and diphenylphosphine oxide, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that can disrupt production schedules. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This resilience is critical for maintaining continuous supply to downstream customers in the highly regulated pharmaceutical industry.
- Scalability and Environmental Compliance: The mild thermal profile of the reaction, operating at just 70°C, allows for easy scale-up using standard stainless steel reactors without the need for specialized high-pressure or cryogenic equipment. This accessibility accelerates the timeline from pilot plant to commercial production. Moreover, the aqueous workup generates benign salt waste rather than toxic heavy metal sludge, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile enhances the marketability of the final product to environmentally conscious partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this synthesis route for their specific projects, we have compiled answers to common inquiries regarding reaction scope and operational parameters. The following insights are derived directly from the experimental data presented in the patent documentation, providing a realistic expectation of performance across diverse substrate classes. Understanding these nuances helps in making informed decisions about process integration and risk management.
Q: What are the advantages of this metal-free hydrodefluorination method over traditional Wittig reactions?
A: Unlike traditional Wittig or Horner-Wadsworth-Emmons reactions that require strong alkaline conditions and often suffer from limited substrate tolerance, this novel method operates under mild conditions (70°C) using cesium carbonate. It offers superior functional group compatibility and eliminates the need for harsh bases, reducing side reactions and simplifying downstream purification.
Q: Why is water added as an additive in this reaction system?
A: Water acts as a crucial reaction promoter in this hydrodefluorination protocol. Experimental data indicates that adding approximately 5 equivalents of water significantly enhances the reaction yield compared to anhydrous conditions. It likely facilitates proton transfer steps essential for the conversion of the trifluoromethyl group to the gem-difluoro moiety without degrading sensitive functional groups.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable due to its use of inexpensive, commercially available reagents like cesium carbonate and ethyl acetate. The absence of expensive transition metal catalysts removes the need for complex heavy metal removal steps, which is a significant bottleneck in GMP manufacturing. The mild temperature profile (70°C) also ensures safe and energy-efficient operation at large scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Difluoroalkene Supplier
As the demand for fluorinated building blocks continues to surge in the development of next-generation therapeutics, having a manufacturing partner with deep expertise in C-F bond formation is essential. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including dedicated containment systems and rigorous QC labs capable of detecting trace impurities at ppm levels. We are committed to delivering high-purity gem-difluoroalkene intermediates that meet the stringent purity specifications required by global regulatory agencies, ensuring your drug development programs proceed without delay.
We invite you to collaborate with our technical team to explore how this innovative hydrodefluorination technology can be integrated into your supply chain. By leveraging our process development capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize yield and reduce lead times. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Together, we can accelerate the delivery of life-saving medicines to patients worldwide through superior chemical manufacturing.
