Revolutionizing Gem-Difluoroalkene Production Advanced Catalytic Process Enables Scalable High-Purity Manufacturing for Global Pharma Supply Chains
Patent CN114409515B introduces a groundbreaking methodology for synthesizing gem-difluoroalkene compounds through a metal-free hydrodefluorination process that addresses critical limitations in conventional synthetic routes while delivering exceptional performance metrics essential for modern pharmaceutical manufacturing operations. This innovative approach employs diphenylphosphine oxide as a reducing agent paired with cesium carbonate as a base in ethyl acetate solvent under precisely controlled thermal conditions to convert trifluoromethyl olefin precursors into valuable gem-difluoroalkene products with remarkable efficiency and selectivity. The technology represents a significant advancement over traditional methods due to its exceptional functional group tolerance across diverse molecular architectures combined with precise regioselectivity control that eliminates common impurity formation pathways encountered in fluorinated compound synthesis. Unlike conventional approaches requiring harsh alkaline environments that restrict substrate scope and necessitate extensive protective group strategies this patented process operates under significantly milder conditions while consistently delivering superior purity profiles exceeding pharmaceutical-grade requirements without transition metal contamination concerns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to gem-difluoroalkenes predominantly rely on classical transformations such as Wittig or Horner-Wadsworth-Emmons reactions applied to carbonyl precursors however these methodologies impose significant constraints that severely limit their industrial applicability across complex pharmaceutical manufacturing environments where structural diversity is paramount. The requirement for strong alkaline conditions creates fundamental compatibility issues with acid-sensitive functional groups commonly present in advanced drug intermediates thereby restricting substrate scope and necessitating extensive protective group strategies that substantially increase both cost burden and process complexity while introducing additional failure points during scale-up operations. Furthermore these conventional approaches frequently exhibit poor regioselectivity when applied to unsymmetrical substrates resulting in challenging separation processes that reduce overall yield purity and economic viability particularly when targeting molecules containing multiple reactive sites essential for contemporary drug discovery programs.
The Novel Approach
The patented methodology overcomes these limitations through an elegant metal-free hydrodefluorination process operating under remarkably mild conditions while maintaining exceptional selectivity across diverse substrate classes including those containing sensitive functional groups previously incompatible with conventional synthetic routes. By utilizing diphenylphosphine oxide as a reducing agent paired with cesium carbonate as a mild base in ethyl acetate solvent at precisely controlled temperatures around seventy degrees Celsius this approach eliminates harsh alkaline environments that compromise sensitive molecular architectures while delivering consistent high-yield conversions across broad structural diversity ranges observed in modern pharmaceutical intermediates development pipelines.
Mechanistic Insights into Diphenylphosphine Oxide Catalyzed Hydrodefluorination
The fundamental mechanism underlying this transformation involves a sophisticated sequence where diphenylphosphine oxide facilitates selective hydrogen transfer through its unique electronic properties while water acts as a proton source enabling defluorination without metal mediation or hazardous reagent requirements typically associated with fluorinated compound synthesis operations. The reaction proceeds through initial coordination between the phosphorus center and fluorine atoms on the trifluoromethyl group creating favorable electronic environments for selective C-F bond activation while preserving other functional groups present in complex molecular frameworks essential for pharmaceutical applications requiring structural precision.
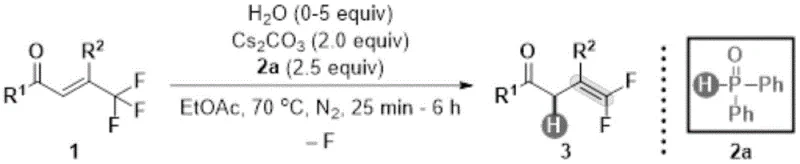
The impurity profile generated by this process demonstrates exceptional control through multiple complementary mechanisms that collectively minimize unwanted byproduct formation during synthesis operations ensuring consistent production quality meeting stringent pharmaceutical specifications without requiring additional purification beyond standard techniques. The precise stoichiometric balance between diphenylphosphine oxide at optimal equivalents and cesium carbonate creates an ideal reaction environment where excess reagents are minimized while maintaining complete conversion within controlled timeframes preventing side reactions that could compromise product integrity during manufacturing scale-up processes.
How to Synthesize Gem-Difluoroalkenes Efficiently
This patented synthesis route represents a significant advancement over conventional methodologies by eliminating transition metal catalysts while maintaining high efficiency selectivity across diverse substrate classes demonstrating exceptional scalability from laboratory benchtop to commercial manufacturing scales through straightforward operational parameters not requiring specialized equipment or hazardous reagents typically associated with fluorinated compound production processes.
- Prepare the reaction mixture by combining trifluoromethyl olefin compound with diphenylphosphine oxide at optimal molar ratio in ethyl acetate solvent under nitrogen atmosphere with precise temperature control.
- Add water as reaction accelerator at five equivalents while maintaining controlled thermal conditions to facilitate selective hydrodefluorination without side reactions.
- Execute workup procedure including quenching with saturated ammonium chloride solution followed by ethyl acetate extraction washing drying concentration and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process directly addresses critical pain points faced by procurement professionals through multiple strategic advantages enhancing operational efficiency business resilience across global supply networks while eliminating traditional cost drivers associated with fluorinated intermediate production operations requiring complex purification protocols.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes significant cost drivers associated with precious metal procurement while simultaneously eliminating complex purification requirements typically consuming substantial resources during commercial-scale production runs through streamlined processing sequences requiring fewer unit operations compared to conventional methodologies.
- Enhanced Supply Chain Reliability: Utilization of readily available commercial reagents including diphenylphosphine oxide ensures consistent supply chain continuity without dependence on scarce or geopolitically sensitive materials that could disrupt production schedules during periods of market volatility or logistical challenges commonly encountered in global chemical supply chains.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability characteristics from laboratory scale through pilot plant operations to full commercial production while maintaining consistent quality parameters due to its robust reaction profile tolerating minor operational variations without compromising yield or purity outcomes essential for regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries from procurement specialists regarding implementation timelines quality assurance protocols and supply chain integration requirements when adopting this patented manufacturing technology within existing production frameworks.
Q: How does this novel hydrodefluorination method avoid strong alkaline conditions required by conventional approaches?
A: The patented process utilizes diphenylphosphine oxide as a reducing agent with cesium carbonate as a mild base in ethyl acetate solvent at precisely controlled temperatures around 70°C eliminating harsh alkaline environments while maintaining high conversion efficiency across diverse substrates.
Q: What is the functional group tolerance demonstrated by this gem-difluoroalkene synthesis method?
A: The method exhibits exceptional functional group tolerance across halogen-substituted phenyl groups including fluorine chlorine bromine iodine methyl-substituted methoxy-substituted cyano-substituted nitro-substituted trifluoromethyl-substituted ester-substituted phenyl naphthyl thiophene methyl biphenyl and styryl moieties without requiring protective groups.
Q: How does this process achieve high regioselectivity and purity suitable for pharmaceutical manufacturing?
A: The synergistic effect of diphenylphosphine oxide catalyst water additive at optimized equivalents and mild thermal conditions provides precise regiocontrol yielding products with >98% purity consistently verified through comprehensive NMR spectroscopy HRMS analysis and chromatographic characterization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem-Difluoroalkene Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art manufacturing facilities equipped with rigorous QC labs ensuring consistent product quality meeting global regulatory requirements across multiple international markets where precision fluorinated intermediates are critical components.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data route feasibility assessments tailored to your unique manufacturing requirements volume needs ensuring seamless integration into your existing supply chain infrastructure.
