Scalable FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Supply Chains
Scalable FeCl3-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles, particularly those incorporating fluorine motifs which are critical for enhancing metabolic stability and bioavailability in drug candidates. A significant breakthrough in this domain is detailed in Chinese Patent CN111675662B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology addresses the longstanding challenges associated with introducing trifluoromethyl groups into complex heterocyclic scaffolds, offering a pathway that is not only chemically elegant but also commercially viable for large-scale manufacturing. The quinazolinone core is a privileged structure found in numerous bioactive molecules, including anticancer agents like Afloqualone and antimalarial drugs, making the development of streamlined access to these derivatives a high priority for R&D teams globally. By leveraging a cheap iron catalyst and readily available starting materials, this invention represents a paradigm shift away from expensive and hazardous traditional methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functionalities has been plagued by significant economic and operational hurdles that hinder their widespread adoption in process chemistry. Conventional literature methods typically rely on the cyclization of synthons such as anthranilamide or isatoic anhydride with expensive trifluoromethylating agents like trifluoroacetic anhydride or ethyl trifluoroacetate. These traditional pathways often suffer from severe reaction conditions, requiring stringent anhydrous environments, strong bases, or precious metal catalysts that drive up the cost of goods significantly. Furthermore, the substrate scope in these older methodologies is frequently narrow, limiting the ability of medicinal chemists to explore diverse chemical space around the quinazolinone core without encountering prohibitive yields or difficult purification challenges. The reliance on costly reagents and the generation of substantial chemical waste also pose environmental compliance issues that modern green chemistry initiatives strive to eliminate.
The Novel Approach
In stark contrast to these legacy processes, the novel approach outlined in the patent utilizes a strategic combination of trifluoroethylimidoyl chloride and isatin derivatives to construct the quinazolinone ring system with remarkable efficiency. This method employs ferric chloride (FeCl3) as a catalyst, which is not only abundant and inexpensive but also demonstrates exceptional catalytic activity under relatively mild aerobic conditions. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization, effectively assembling the target scaffold in a single pot. 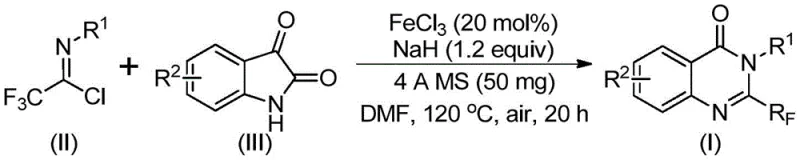 This streamlined protocol eliminates the need for multiple isolation steps and allows for excellent functional group tolerance, accommodating various substituents on the aromatic rings without compromising yield. The ability to use simple, commercially available building blocks transforms the economic landscape of producing these valuable intermediates.
This streamlined protocol eliminates the need for multiple isolation steps and allows for excellent functional group tolerance, accommodating various substituents on the aromatic rings without compromising yield. The ability to use simple, commercially available building blocks transforms the economic landscape of producing these valuable intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic attack and transition metal catalysis that ensures high selectivity and conversion. Initially, the sodium hydride acts as a base to deprotonate the isatin substrate, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a key trifluoroacetamidine intermediate, setting the stage for the subsequent ring closure. The presence of ferric chloride is crucial here, as it facilitates the decarbonylation step—a notoriously difficult transformation—by coordinating with the carbonyl oxygen and lowering the activation energy required for carbon monoxide extrusion. This iron-mediated process allows the reaction to proceed smoothly at 120°C in polar aprotic solvents like DMF, avoiding the extreme temperatures often needed for thermal decarbonylation. The use of 4A molecular sieves further drives the equilibrium forward by sequestering moisture, ensuring that the sensitive imidoyl chloride and the reactive intermediates remain stable throughout the 20-hour reaction window.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based trifluoromethylation strategies which often lead to complex mixtures of regioisomers and over-fluorinated byproducts. The concerted nature of the cyclization ensures that the trifluoromethyl group is installed specifically at the 2-position of the quinazolinone ring, as evidenced by the clean NMR spectra and high purity of the isolated products reported in the patent examples. The tolerance for electron-withdrawing and electron-donating groups on both the isatin and the imidoyl chloride components suggests that the electronic demands of the transition state are well-balanced by the iron catalyst. 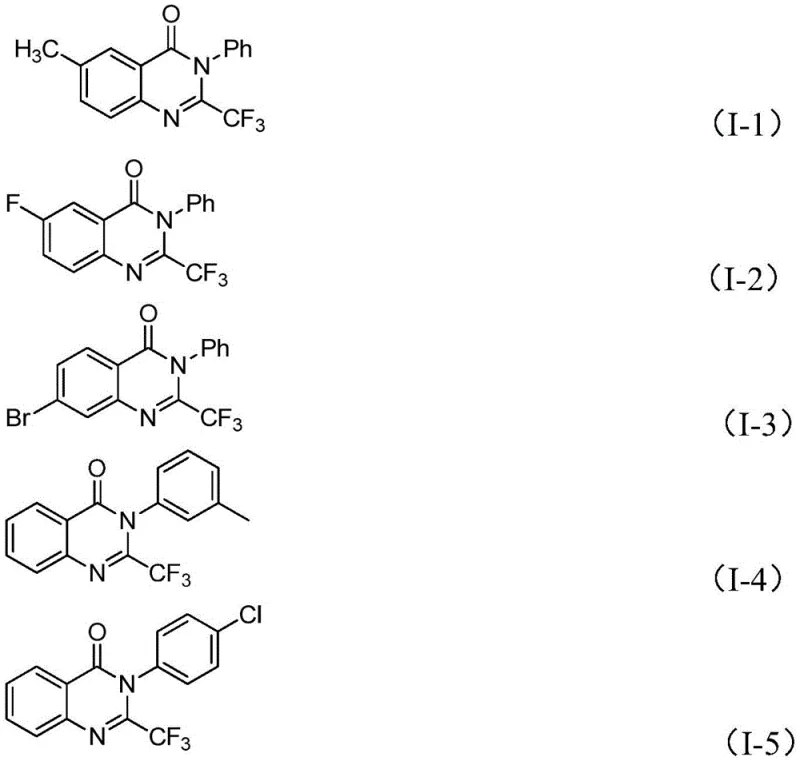 This robustness minimizes the formation of side products, thereby simplifying downstream purification and reducing the overall burden on quality control laboratories. For process chemists, understanding this mechanism provides confidence in scaling the reaction, as the rate-limiting steps are well-defined and manageable within standard batch reactor configurations.
This robustness minimizes the formation of side products, thereby simplifying downstream purification and reducing the overall burden on quality control laboratories. For process chemists, understanding this mechanism provides confidence in scaling the reaction, as the rate-limiting steps are well-defined and manageable within standard batch reactor configurations.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and temperature profiling to maximize yield and safety. The protocol dictates a two-stage heating profile, starting at a lower temperature of 40°C to allow for the initial coupling, followed by ramping to 120°C to drive the cyclization and decarbonylation to completion. The use of sodium hydride necessitates strict safety protocols regarding handling and quenching, while the selection of DMF as the solvent ensures optimal solubility for all polar intermediates involved in the cascade. Detailed standardized operating procedures for this specific transformation, including precise workup and purification guidelines, are essential for reproducibility.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed methodology presents a compelling value proposition centered on cost optimization and risk mitigation. The shift from precious metal catalysts to ferric chloride represents a drastic reduction in raw material costs, as iron salts are orders of magnitude cheaper than palladium or rhodium complexes. Moreover, the elimination of specialized fluorinating reagents in favor of commodity chemicals like isatin stabilizes the supply chain against market volatility. This resilience is critical for maintaining continuous production schedules for active pharmaceutical ingredients (APIs) where interruptions can have cascading effects on global drug availability. The simplified workflow also translates to reduced labor hours and lower utility consumption, contributing to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive catalytic systems and reagents with low-cost alternatives. By utilizing ferric chloride, a ubiquitous industrial chemical, manufacturers avoid the high procurement costs and recycling complexities associated with noble metals. Additionally, the high atom economy of the reaction, where the majority of the starting material mass is incorporated into the final product, minimizes waste disposal fees. The ability to run the reaction under air rather than inert gas further reduces infrastructure costs, as there is no need for extensive nitrogen purging systems or specialized glovebox equipment, leading to substantial overall cost savings in the production budget.
- Enhanced Supply Chain Reliability: Securing a reliable supply of critical intermediates is paramount for pharmaceutical manufacturing, and this route significantly de-risks the sourcing strategy. The starting materials, specifically isatin derivatives and aromatic amines used to make the imidoyl chlorides, are produced on a massive scale globally, ensuring consistent availability and competitive pricing. Unlike proprietary reagents that may be sourced from a single vendor, these commodities have a diversified supplier base, protecting the manufacturing process from single-point failures. This diversification allows procurement teams to negotiate better terms and maintain buffer stocks more easily, ensuring that production timelines are met even during periods of global supply chain disruption.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern sustainability goals, facilitating easier regulatory approval and community acceptance. The use of iron, a non-toxic metal, eliminates the need for rigorous heavy metal clearance steps that are mandatory when using palladium or copper, thus simplifying the purification train and reducing solvent usage. The reaction conditions are compatible with standard stainless steel reactors, meaning that scale-up from gram to kilogram quantities does not require capital-intensive equipment modifications. This ease of scale-up, combined with the generation of less hazardous waste, positions this technology as a sustainable choice for long-term commercial manufacturing of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what potential partners can expect regarding performance and feasibility. Understanding these details is crucial for evaluating the fit of this technology within existing manufacturing portfolios.
Q: What are the advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: Using ferric chloride (FeCl3) offers significant cost advantages as it is an abundant, non-precious metal compared to palladium or rhodium catalysts often used in cross-coupling. Furthermore, the patent data indicates high functional group tolerance and operational simplicity under aerobic conditions, eliminating the need for rigorous inert gas protection which simplifies reactor setup and reduces operational overhead.
Q: How does this method improve the supply chain stability for trifluoromethyl intermediates?
A: This methodology utilizes isatin and trifluoroethylimidoyl chloride as starting materials, both of which are commercially available and inexpensive commodity chemicals. By avoiding exotic or highly specialized fluorinating reagents that often suffer from supply bottlenecks, manufacturers can secure a more reliable raw material base, ensuring consistent production schedules and reducing the risk of project delays due to material shortages.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for scalability. The reaction conditions utilize standard solvents like DMF and operate at moderate temperatures (up to 120°C) without requiring high-pressure equipment. The patent documentation confirms successful expansion to gram-level synthesis with robust yields, indicating that the kinetics and thermodynamics are favorable for translation into multi-kilogram or ton-scale manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed synthesis for the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting the most stringent purity specifications required for clinical and commercial grade intermediates. We understand that consistency is key in the supply of critical building blocks, and our robust quality management systems guarantee that every batch of 2-trifluoromethyl quinazolinone delivered meets exacting standards for identity, purity, and impurity profiles.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this route can impact your bottom line. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your partner in driving efficiency and innovation in your supply chain.
