Advanced Metal-Free Synthesis of 3-Hydroxymethyl-4-phenyl-3,4-dihydroquinolinone Intermediates
Introduction to Patent CN112125843B Technology
The pharmaceutical industry continuously seeks robust and scalable methods for constructing complex heterocyclic scaffolds, particularly those found in bioactive natural products and synthetic drugs. Patent CN112125843B introduces a groundbreaking preparation method for 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone compounds, a structural motif known for enhancing metabolic stability and bioavailability in drug candidates. This technology leverages a unique epoxidation-intramolecular Friedel-Crafts alkylation tandem reaction promoted by potassium monopersulfate (Oxone). Unlike traditional methods that often rely on expensive transition metals or harsh acidic conditions, this approach operates under mild, metal-free conditions using acetonitrile as the reaction medium. The significance of this innovation lies in its ability to directly transform inexpensive N-methyl-N-aryl-2-phenylacrylamide derivatives into high-value quinolinone intermediates with excellent functional group tolerance.
For R&D directors and process chemists, the appeal of this methodology is rooted in its simplicity and environmental profile. The reaction utilizes readily available starting materials and avoids the generation of toxic heavy metal waste, aligning perfectly with modern green chemistry principles. By employing a dual-function reagent that acts as both an oxidant and an acid promoter, the process simplifies the reaction setup and downstream purification. This patent represents a significant leap forward in the synthesis of hydroxymethyl-functionalized heterocycles, offering a reliable pathway for the production of high-purity pharmaceutical intermediates that meet stringent regulatory standards for residual impurities.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3,4-dihydroquinolinone skeleton has often depended on transition metal catalysis or the use of strong stoichiometric acids, presenting significant challenges for large-scale manufacturing. Conventional routes frequently involve palladium, rhodium, or copper catalysts, which introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing these trace metals to meet ICH Q3D guidelines requires additional purification steps, such as scavenger treatments or extensive chromatography, which drastically increase production costs and reduce overall yield. Furthermore, many traditional cyclization protocols require harsh reaction conditions, including high temperatures or corrosive Lewis acids, which can lead to poor functional group compatibility and the formation of complex impurity profiles that are difficult to separate.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes potassium monopersulfate (Oxone) as a benign and efficient promoter for the cyclization of N-methyl-N-aryl-2-phenylacrylamides. This method eliminates the need for any transition metal catalysts or external acid additives, relying instead on the intrinsic properties of the oxidant system. The reaction proceeds smoothly in acetonitrile at moderate temperatures, typically around 90°C, achieving high conversion rates without the safety hazards associated with strong mineral acids. This metal-free strategy not only simplifies the workflow but also ensures that the resulting 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone compounds are free from metallic impurities, thereby reducing the burden on quality control laboratories and streamlining the regulatory approval process for downstream drug applications.
Mechanistic Insights into Oxone-Promoted Oxidative Cyclization
The mechanistic pathway of this transformation is a sophisticated cascade that begins with the epoxidation of the alkene moiety in the acrylamide substrate. Under heating conditions, potassium monopersulfate acts as the primary oxidant, transferring an oxygen atom to the double bond of the N-methyl-N-aryl-2-phenylacrylamide to generate an epoxy intermediate. Crucially, the reaction generates potassium bisulfate (KHSO4) as a byproduct, which serves as an in situ proton source. This acidic species activates the epoxide ring, facilitating its opening and generating a cationic intermediate that is primed for intramolecular attack. This elegant design allows the reaction to proceed without the addition of external acids, as the reagent itself provides the necessary acidic environment to drive the cyclization forward efficiently.
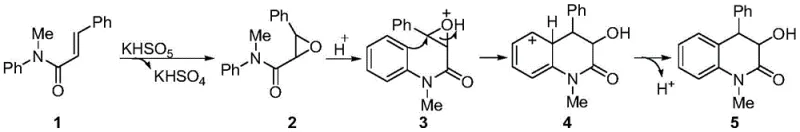
Following the activation of the epoxide, the system undergoes an intramolecular Friedel-Crafts alkylation, where the electron-rich aromatic ring attacks the electrophilic carbon of the opened epoxide. This step forms the core six-membered ring of the dihydroquinolinone structure. The final stage involves a dehydroaromatization process that restores aromaticity to the benzene ring, yielding the stable 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone product. From an impurity control perspective, this mechanism is highly advantageous because it avoids radical pathways often associated with metal catalysis, which can lead to polymerization or non-selective oxidation. The specificity of the ionic mechanism ensures a cleaner reaction profile, minimizing the formation of regioisomers or over-oxidized byproducts, which is critical for maintaining high purity in pharmaceutical intermediate manufacturing.
How to Synthesize 3-Hydroxymethyl-4-phenyl-3,4-dihydroquinolinone Efficiently
Implementing this synthesis requires careful attention to solvent selection and stoichiometry to maximize yield and reproducibility. The protocol dictates the use of acetonitrile as the exclusive reaction medium, as screening data indicates that other common organic solvents fail to support the reaction, highlighting the unique solvation properties required for this oxidative cascade. The concentration of the substrate should be maintained between 0.1 and 0.5 mol/L to ensure optimal interaction between the reactants and the oxidant. While the standard procedure utilizes 1.5 equivalents of Oxone, slight adjustments within the 1.2 to 2.0 equivalent range can be made depending on the specific electronic nature of the substrate substituents. The detailed standardized synthesis steps for laboratory and pilot scale execution are provided below.
- Dissolve N-methyl-N-aryl-2-phenylacrylamide in acetonitrile solvent to achieve a concentration between 0.1 and 0.5 mol/L.
- Add 1.5 equivalents of Potassium Monopersulfate (Oxone) to the reaction mixture under stirring.
- Heat the reaction mixture to 90°C and maintain for 24 hours to complete the epoxidation-intramolecular Friedel-Crafts alkylation cascade.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free oxidative cyclization offers substantial strategic advantages regarding cost structure and supply continuity. The elimination of precious metal catalysts removes a major variable cost component and mitigates the supply risk associated with fluctuating prices of commodities like palladium or rhodium. Furthermore, the use of Oxone, a commodity chemical produced on a massive scale for water treatment and cleaning applications, ensures a stable and low-cost supply of the key reagent. The simplified workup procedure, which avoids complex metal scavenging steps, translates directly into reduced processing time and lower consumption of auxiliary materials, contributing to a more lean and efficient manufacturing operation.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and external acid additives significantly lowers the raw material costs per kilogram of product. By avoiding the need for expensive ligands and specialized metal scavengers required for purification, the overall cost of goods sold (COGS) is drastically reduced. Additionally, the high atom economy of the tandem reaction minimizes waste generation, leading to lower disposal costs and a more sustainable production footprint that aligns with corporate environmental goals.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-hazardous starting materials such as acrylamide derivatives and Oxone enhances the resilience of the supply chain. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process are commoditized and easily sourced from multiple global suppliers. This diversification reduces the risk of production stoppages due to raw material shortages and allows for more flexible inventory management strategies.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process highly scalable from kilogram to multi-ton production without significant engineering hurdles. The aqueous byproducts are easier to treat compared to heavy metal waste streams, simplifying compliance with increasingly strict environmental regulations. This ease of scale-up ensures that the technology can seamlessly transition from R&D to commercial manufacturing, supporting rapid market entry for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on reaction parameters and substrate limitations. Understanding these details is essential for process engineers evaluating the feasibility of adopting this route for their specific intermediate requirements.
Q: Why is acetonitrile critical for this oxidative cyclization?
A: According to patent data, acetonitrile is the only solvent that supports the reaction; other common solvents like DCM, toluene, or DMF result in no reaction, likely due to specific solvation effects required for the Oxone activation.
Q: Does this method require transition metal catalysts?
A: No, the process is entirely metal-free, utilizing Potassium Monopersulfate as both the oxidant and the source of acidic protons (via KHSO4 byproduct), eliminating heavy metal contamination risks.
Q: What is the functional group tolerance of this synthesis?
A: The method demonstrates excellent compatibility with various substituents on the aryl ring, including alkyl (methyl), alkoxy (methoxy), and halogen (bromo) groups, yielding products with 65-80% isolated yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxymethyl-4-phenyl-3,4-dihydroquinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the metal-free oxidative cyclization technology described in Patent CN112125843B and confirmed its potential for high-efficiency manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-hydroxymethyl-4-phenyl-3,4-dihydroquinolinone intermediate meets the highest industry standards.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their pipeline projects. By optimizing this metal-free route, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating tangible economic benefits over traditional methods. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can accelerate your drug development timeline while optimizing your supply chain economics.
