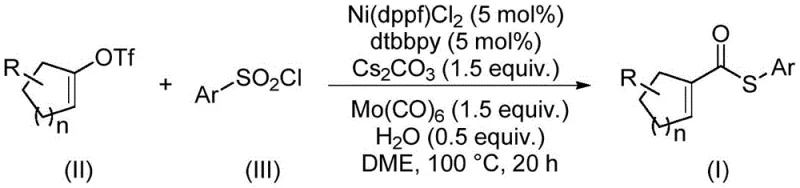
Advanced Nickel-Catalyzed Synthesis of Alpha Beta Unsaturated Thioesters for Commercial Scale-Up
The landscape of organic synthesis is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN114773242A, which discloses a novel preparation method for α,β-unsaturated thioester compounds. These compounds are pivotal building blocks in the construction of complex molecular architectures, serving as critical intermediates in Diels-Alder reactions, conjugate additions, and cascade cyclizations. Traditionally, the synthesis of such high-value intermediates has been plagued by the use of expensive precious metal catalysts and hazardous reagents. However, this new technology leverages a nickel-catalyzed thiocarbonylation strategy that fundamentally shifts the economic and safety paradigms of production. By utilizing readily available alkenyl trifluoromethanesulfonates and arylsulfonyl chlorides, this process offers a robust pathway for the reliable alpha,beta-unsaturated thioester supplier market, addressing long-standing inefficiencies in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α,β-unsaturated thioesters has relied heavily on transition metal-catalyzed thiocarbonylation reactions involving rhodium, platinum, or palladium complexes. While these precious metals exhibit high catalytic activity, their exorbitant cost presents a formidable barrier to large-scale commercial adoption, severely impacting the cost reduction in pharmaceutical intermediate manufacturing. Moreover, conventional protocols frequently necessitate the use of thiols as the sulfur source. Thiols are notoriously malodorous, toxic, and prone to causing catalyst poisoning, which leads to inconsistent reaction yields and complicates waste management. The reliance on gaseous carbon monoxide as a carbonyl source further introduces significant safety hazards, requiring specialized high-pressure equipment and rigorous containment protocols that are difficult to maintain in standard multipurpose chemical plants.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift by replacing these problematic components with safer, more economical alternatives. Instead of precious metals, the process employs a nickel catalyst system, specifically (1,1'-bis(diphenylphosphino)ferrocene)nickel dichloride, which is abundant and cost-effective. Crucially, the reaction utilizes arylsulfonyl chlorides as the sulfur source, completely eliminating the need for foul-smelling thiols and mitigating the risk of catalyst deactivation. Furthermore, molybdenum hexacarbonyl serves a dual function as both the carbonyl source and the reducing agent, obviating the need for dangerous CO gas cylinders. This integrated approach not only streamlines the operational workflow but also broadens the substrate scope, allowing for the synthesis of diverse derivatives with high efficiency and minimal environmental footprint.
Mechanistic Insights into Nickel-Catalyzed Thiocarbonylation
The core of this innovation lies in the intricate catalytic cycle facilitated by the nickel complex. The reaction initiates with the oxidative addition of the alkenyl trifluoromethanesulfonate to the active Ni(0) species generated in situ. Unlike traditional carbonylations where Ni(0) might form inactive Ni(CO)4 species with free CO, the use of Mo(CO)6 allows for a controlled release of carbonyl units. The nickel center subsequently undergoes migratory insertion of the carbonyl group, followed by the coordination and insertion of the sulfur moiety derived from the arylsulfonyl chloride. The presence of the bipyridine ligand, 4,4'-di-tert-butyl-2,2'-bipyridine, stabilizes the nickel center and modulates its electronic properties, ensuring high turnover numbers. The cycle concludes with a reductive elimination step that releases the desired α,β-unsaturated thioester product and regenerates the active catalyst. This mechanistic pathway is remarkably robust, tolerating a wide array of functional groups including halogens and alkyl chains, which is essential for the commercial scale-up of complex polymer additives and pharmaceutical precursors.
Impurity control is another critical aspect where this mechanism excels. The specific choice of cesium carbonate as the base and the precise stoichiometric balance of water (0.5 equivalents) play a vital role in suppressing side reactions such as hydrodehalogenation or homocoupling of the alkenyl species. The mild reaction conditions, typically maintained at 100°C in ethylene glycol dimethyl ether (DME), prevent thermal degradation of sensitive functional groups. This results in a cleaner crude reaction profile, significantly reducing the burden on downstream purification processes like column chromatography. For R&D teams, understanding these mechanistic nuances is key to optimizing the process for specific substrates, ensuring that the final high-purity OLED material or API intermediate meets stringent regulatory specifications without extensive recrystallization steps.
How to Synthesize Alpha,Beta-Unsaturated Thioesters Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and reproducibility. The protocol involves charging a sealed reaction vessel with the nickel catalyst, ligand, molybdenum carbonyl, base, and substrates under an inert atmosphere. The reaction mixture is then heated to facilitate the transformation over a defined period. While the general procedure is straightforward, attention to detail regarding reagent quality and mixing efficiency is paramount. The following guide outlines the standardized steps derived from the patent examples to assist technical teams in replicating this high-efficiency process.
- Combine nickel catalyst (Ni(dppf)Cl2), ligand (dtbbpy), molybdenum carbonyl, cesium carbonate, water, alkenyl trifluoromethanesulfonate, and arylsulfonyl chloride in a sealed tube.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture thoroughly to ensure homogeneity.
- Heat the reaction mixture to 100°C for 20 hours, then perform post-treatment including filtration and column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this nickel-catalyzed methodology offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material supply chain. By shifting away from scarce precious metals and hazardous gases, manufacturers can secure a more stable and predictable supply of inputs. The use of commodity chemicals like arylsulfonyl chlorides and alkenyl triflates ensures that production is not bottlenecked by the availability of exotic reagents. This stability is crucial for maintaining continuous manufacturing operations and meeting tight delivery schedules for global clients.
- Cost Reduction in Manufacturing: The substitution of expensive rhodium or palladium catalysts with nickel results in a substantial decrease in direct material costs. Nickel is an earth-abundant metal with a fraction of the price of precious metals, leading to significant savings on the bill of materials. Additionally, the elimination of thiols reduces the costs associated with specialized odor-control scrubbing systems and hazardous waste disposal. The dual role of molybdenum carbonyl further consolidates reagent costs, as a single compound replaces the need for separate carbonyl and reducing agents, streamlining inventory management and purchasing logistics.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against functional group variations means that a single platform technology can be adapted to produce a wide library of derivatives without requalifying entirely new processes. This flexibility allows suppliers to respond rapidly to changing market demands for specific intermediates. Furthermore, the avoidance of high-pressure CO gas removes a major regulatory and safety hurdle, facilitating easier permitting and faster ramp-up times in new production facilities. This agility ensures reducing lead time for high-purity pharmaceutical intermediates, providing a competitive edge in fast-paced markets.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior. The absence of volatile thiols and high-pressure carbon monoxide minimizes the risk of acute exposure incidents and lowers the facility's overall environmental impact. The reaction operates at atmospheric pressure in a sealed tube or standard reactor, simplifying the engineering requirements for scale-up from grams to tons. Waste streams are easier to treat due to the lack of sulfur-containing odors and heavy metal residues typical of precious metal catalysis, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries based on the patent specifications. These insights address the practical concerns of scaling this reaction and integrating it into existing production workflows. Understanding these details helps stakeholders make informed decisions regarding process adoption and supplier selection.
Q: Why is this nickel-catalyzed method superior to traditional rhodium or palladium methods?
A: Traditional methods often rely on expensive precious metals like rhodium and palladium, which significantly increase production costs. Furthermore, conventional thiocarbonylation frequently requires malodorous and toxic thiols that can poison catalysts. This novel nickel-based approach utilizes inexpensive nickel catalysts and avoids thiols entirely by using arylsulfonyl chlorides as the sulfur source.
Q: What serves as the carbonyl source in this reaction, and is it safe?
A: The reaction utilizes molybdenum hexacarbonyl (Mo(CO)6) which acts as both the carbonyl source and the reducing agent. This eliminates the need for handling hazardous carbon monoxide gas cylinders directly, thereby enhancing operational safety and simplifying the engineering controls required for commercial scale-up.
Q: Does this synthesis method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance. It successfully accommodates various substituents on the aryl ring, including alkyl groups (methyl, isopropyl, tert-butyl), halogens (chlorine), and electron-donating or withdrawing groups (methoxy, trifluoromethoxy), making it highly versatile for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed synthesis for the fine chemical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe handling of molybdenum reagents and efficient purification protocols. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha,beta-unsaturated thioester meets the highest quality standards required by the global pharmaceutical and agrochemical sectors.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply chain for your critical intermediates.
