Advanced Catalytic Cyanation: Scaling High-Purity Aryl Nitriles for Global Pharmaceutical Supply Chains
Advanced Catalytic Cyanation: Scaling High-Purity Aryl Nitriles for Global Pharmaceutical Supply Chains
The synthesis of substituted aromatic nitriles represents a critical bottleneck in the production of active pharmaceutical ingredients (APIs) and agrochemical intermediates, where traditional methods often compromise safety and efficiency. Patent CN101113138B introduces a transformative methodology utilizing a ferrocene imine cyclopalladium complex to catalyze the cyanation of aryl halides using potassium ferrocyanide as a non-toxic cyanide source. This technological breakthrough addresses the longstanding industry challenge of balancing high reaction specificity with environmental compliance, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. By replacing hazardous free cyanide salts with stable ferrocyanide and employing a highly active palladium catalyst, this process achieves exceptional yields under relatively mild thermal conditions, positioning it as a superior alternative for modern green chemistry initiatives in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aryl nitriles has relied heavily on nucleophilic substitution reactions using highly toxic cyanide sources such as potassium cyanide (KCN) or sodium cyanide (NaCN), which pose severe safety risks and require stringent containment protocols to prevent environmental contamination. Alternative methods utilizing copper(I) cyanide (CuCN) or zinc cyanide (Zn(CN)2) often suffer from stoichiometric metal waste generation and limited functional group tolerance, leading to complex downstream purification processes that inflate production costs. Furthermore, earlier palladium-catalyzed approaches frequently necessitated the use of expensive and air-sensitive bisphosphine ligands to stabilize the catalytic species, while still exhibiting narrow substrate scopes that were generally restricted to activated aryl bromides, thereby limiting their utility for diverse chemical libraries required in drug discovery.
The Novel Approach
The innovative strategy outlined in the patent data leverages a specialized ferrocene imine cyclopalladium complex to activate the carbon-halogen bond efficiently, enabling the use of the much safer potassium ferrocyanide trihydrate as the cyanating agent. This method eliminates the need for hazardous free cyanide handling and avoids the generation of equimolar heavy metal waste associated with copper or zinc reagents, significantly simplifying the waste treatment workflow. The reaction demonstrates remarkable versatility, successfully converting a wide array of substrates including electron-rich, electron-deficient, and sterically hindered aryl bromides and iodides, as well as heterocyclic compounds, into their corresponding nitriles with high purity. 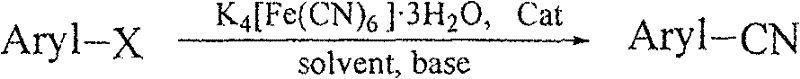
Mechanistic Insights into Ferrocene Imine Cyclopalladium Catalysis
The efficacy of this synthetic route is fundamentally rooted in the unique electronic and steric properties of the ferrocene imine ligand system coordinated to the palladium center, which facilitates a robust catalytic cycle capable of sustaining high turnover numbers. The ferrocene backbone provides a rigid yet electronically tunable framework that stabilizes the palladium species against aggregation and deactivation, a common failure mode in homogeneous catalysis involving simple palladium salts. During the catalytic cycle, the palladium complex undergoes oxidative addition with the aryl halide, followed by transmetallation with the ferrocyanide anion, and finally reductive elimination to release the aryl nitrile product while regenerating the active catalyst species. 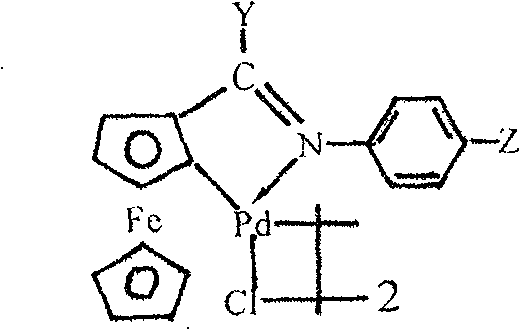
Impurity control is inherently enhanced by the high chemoselectivity of this catalyst system, which minimizes side reactions such as hydrodehalogenation or homocoupling that often plague less sophisticated catalytic systems. The specific coordination geometry imposed by the cyclometallated ferrocene ligand ensures that the cyanide transfer occurs with precision, resulting in product purities consistently exceeding 98% as demonstrated in the experimental examples provided within the patent documentation. This level of purity is critical for high-purity pharmaceutical intermediates where trace metal contaminants or organic byproducts can compromise the safety profile of the final drug substance, thereby reducing the burden on downstream crystallization and chromatography steps.
How to Synthesize Aryl Nitrile Derivatives Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety, beginning with the dissolution of the catalyst, potassium ferrocyanide, and a suitable inorganic base in a polar aprotic solvent such as N,N-dimethylacetamide or N,N-dimethylformamide. The reaction mixture is rigorously degassed and maintained under a nitrogen atmosphere to prevent oxidation of the catalyst or the reactants, after which the halogenated aromatic substrate is introduced and the system is heated to temperatures ranging between 100°C and 150°C. Detailed standardized synthesis steps, including specific molar ratios, reaction times, and workup procedures for various substrates, are provided in the technical guide below to ensure consistent results across different batches.
- Dissolve the ferrocene imine cyclopalladium catalyst, potassium ferrocyanide, and an inorganic base in a polar aprotic solvent such as DMF or DMAc.
- Add the halogenated aromatic compound (aryl bromide, iodide, or chloride) to the mixture under a nitrogen atmosphere.
- Heat the reaction mixture to 100-150°C for 1-20 hours, then cool, extract with organic solvents, and purify to obtain the aryl nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this ferrocene-catalyzed cyanation technology offers substantial strategic advantages by mitigating regulatory risks associated with toxic raw materials and streamlining the manufacturing workflow. The substitution of lethal cyanide salts with benign potassium ferrocyanide drastically reduces the safety infrastructure costs and insurance liabilities typically associated with handling Schedule 1 chemicals, while simultaneously simplifying the logistics of raw material sourcing and storage. This shift not only enhances the overall safety profile of the manufacturing facility but also aligns with increasingly stringent global environmental regulations regarding hazardous waste discharge, ensuring long-term operational continuity without the threat of regulatory shutdowns.
- Cost Reduction in Manufacturing: The elimination of expensive bisphosphine ligands and the use of a highly efficient catalyst loading significantly lowers the direct material costs associated with the cyanation process. By avoiding the generation of stoichiometric amounts of copper or zinc waste, the expense related to heavy metal scavenging and hazardous waste disposal is drastically reduced, contributing to a more lean and cost-effective production model. Furthermore, the high yields and selectivity minimize the loss of valuable starting materials, optimizing the overall atom economy and reducing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Utilizing potassium ferrocyanide as a cyanide source removes the supply chain vulnerabilities linked to the strict transportation and storage controls required for free cyanide salts. The robustness of the catalyst allows for the use of a broader range of commercially available aryl halides, including chlorides with appropriate activation, providing procurement teams with greater flexibility in sourcing raw materials and reducing dependency on specific, high-cost bromide precursors. This flexibility ensures a more resilient supply chain capable of adapting to market fluctuations in raw material availability.
- Scalability and Environmental Compliance: The mild reaction conditions and the stability of the catalyst system make this process highly amenable to scale-up from laboratory benchtop to multi-ton commercial production without significant re-engineering of the reactor setup. The reduced toxicity profile of the reagents simplifies the environmental impact assessment and permitting process for new manufacturing lines, accelerating the time-to-market for new products. Additionally, the simplified workup procedure involving standard aqueous extraction and crystallization facilitates easier integration into existing multipurpose manufacturing suites.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this catalytic system, focusing on substrate compatibility, catalyst recovery, and safety protocols to assist R&D and process engineering teams in evaluating feasibility. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a reliable foundation for process development decisions.
Q: Why is potassium ferrocyanide preferred over traditional cyanide sources like KCN?
A: Potassium ferrocyanide is significantly less toxic and environmentally hazardous compared to free cyanide salts like KCN or NaCN, reducing safety risks and waste disposal costs in large-scale manufacturing.
Q: What represents the key advantage of the ferrocene imine ligand in this catalyst?
A: The ferrocene imine ligand provides superior stability and electronic tuning for the palladium center, enabling high yields across a broad range of substrates including electron-rich and electron-deficient aryl halides without requiring expensive bisphosphine ligands.
Q: Can this method be applied to aryl chlorides?
A: Yes, while aryl bromides and iodides are preferred, the patent indicates that aryl chlorides can also be converted effectively, particularly when assisted by additives like triphenylphosphine, expanding the scope of accessible starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Nitrile Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced catalytic technologies to deliver superior quality chemical intermediates to the global marketplace. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of aryl nitrile meets the exacting standards required by top-tier pharmaceutical clients.
We invite potential partners to engage with our technical procurement team to discuss how this innovative cyanation technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments for your target molecules, ensuring a secure and competitive supply chain for your critical drug development programs.
